Pharmacology
Notes
Drug Development in the US
* The FDA oversees studies, they don’t participate in them
Basic research
1.1. identify target
Drug design
2.1. high throughput screening
2.2. rational drug screening
Preclinical studies
3.1. in vitro
3.2. cell culture
3.3. animals
3.4. toxicity studies
3.5. therapeutic index
3.5.1. want a high index
Clinical trials
4.1. Phase I
4.1.1. Safety & tolerability
4.1.2. ~20-100 healthy volunteers
4.1.3. 66% success rates
4.2. Phase II
4.2.1. efficacy
4.2.2. ~100-300 participants with target disease/disorder
4.2.3. 49% success rate
4.3. Phase III
4.3.1. safety & side-effects
4.3.2. ~1,000-5,000 patients
4.3.3. 59% success rate
FDA approval/denial
5.1. an independent panel of reviewers
5.2. sometimes the FDA approves drugs despite the review panel not approving it
Insurance
6.1. The insurance company can then determine if they’ll cover the drug & how much they’ll cover
10% success rate of drugs reaching the market
Can take ~15 years & millions of dollars
hence the high cost
patent for ~20 years
then off-brand copies can be made
Receptor Families
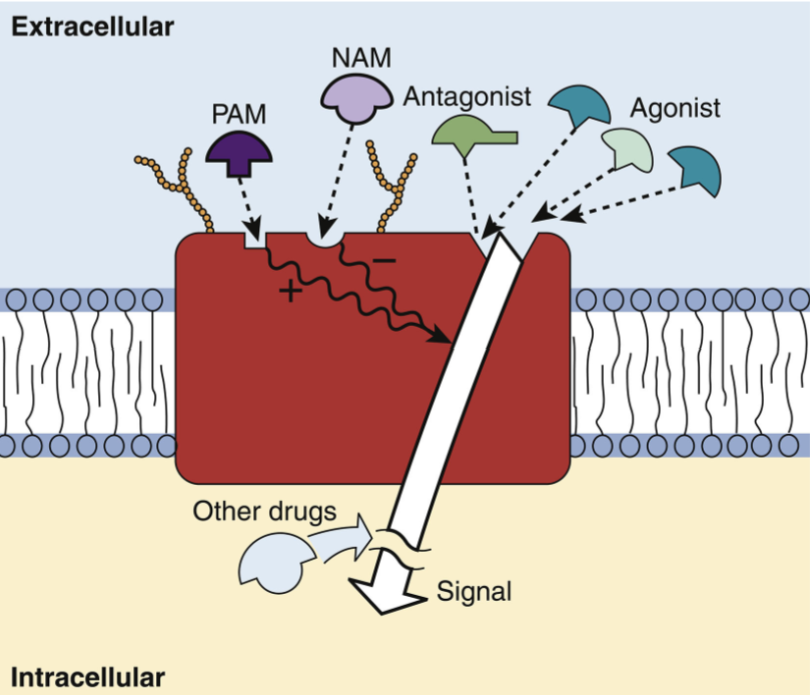
Potency
the amount of drug required for a response
Efficacy
how good a drug is at getting a desired response
Therapeutic index
margin of safety measured in a ratio of—
toxic dose-effect/therapeutic dose effect
the higher the index the better
the lower, the more controlled it needs to be
Ligand-Gated Ion Channels (LGIC)
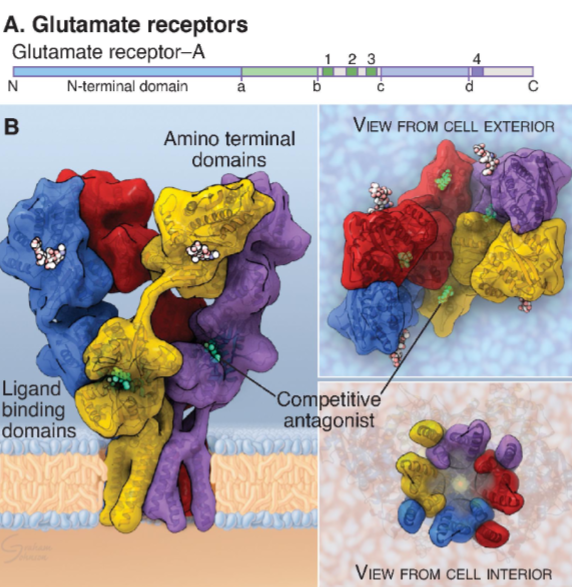
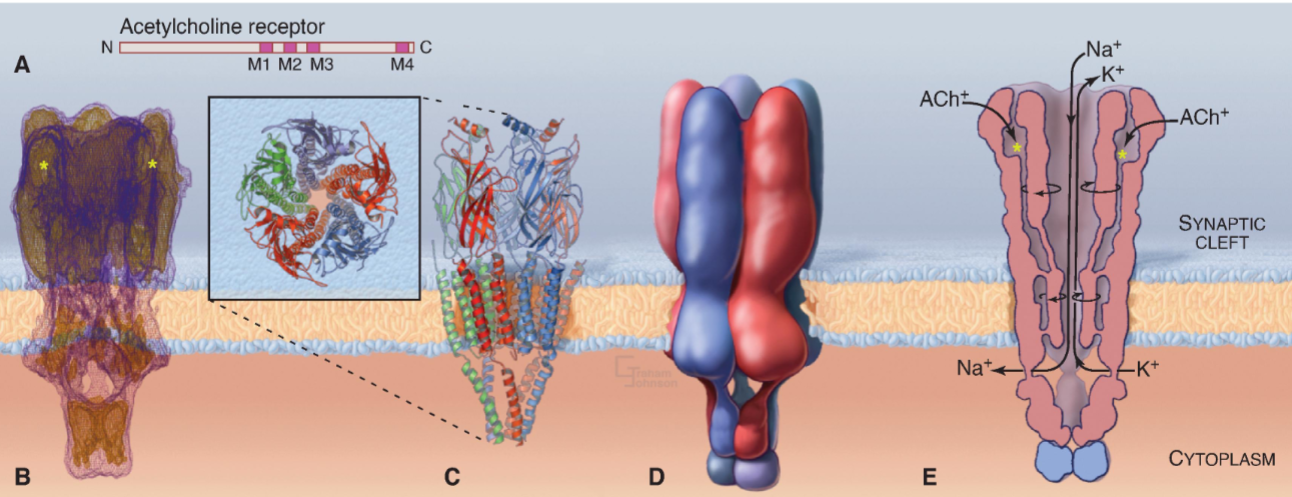
Nicotine is an agonist
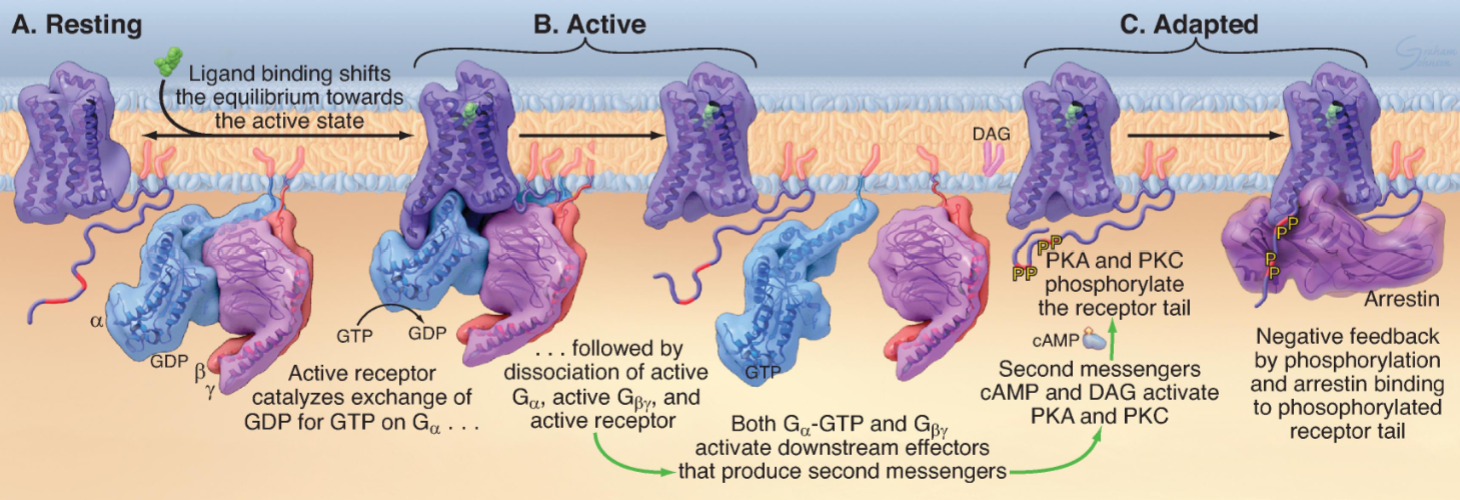
G protein-coupled receptors (GPCR)
aka: Seven-helix receptor
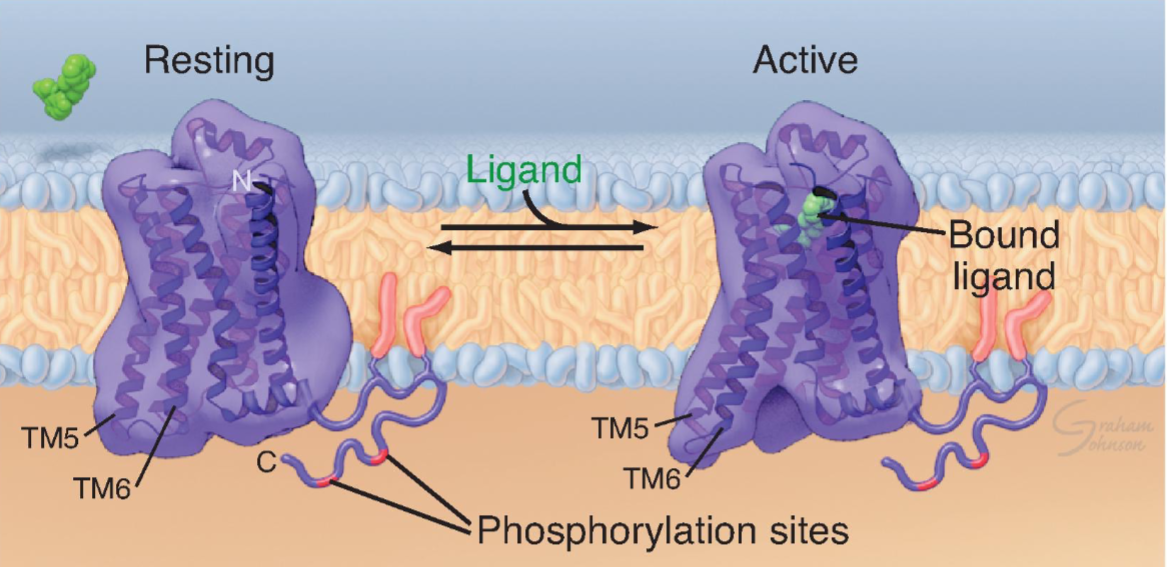
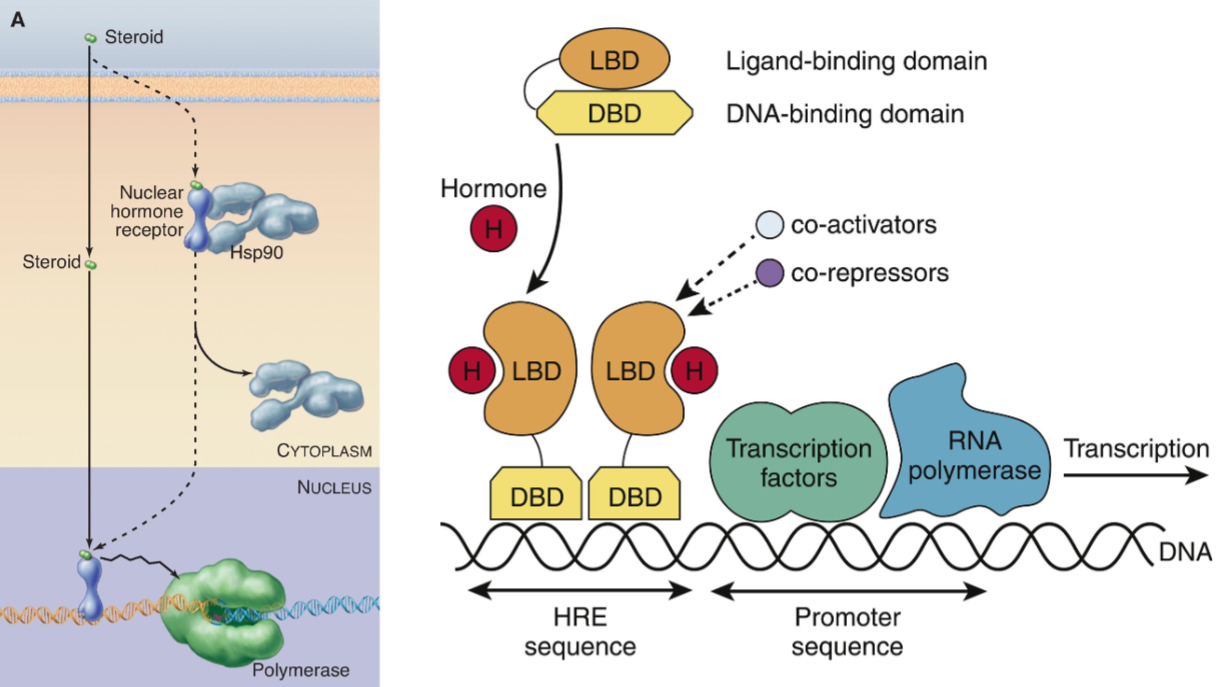
Nuclear Hormone Receptors (NHR)
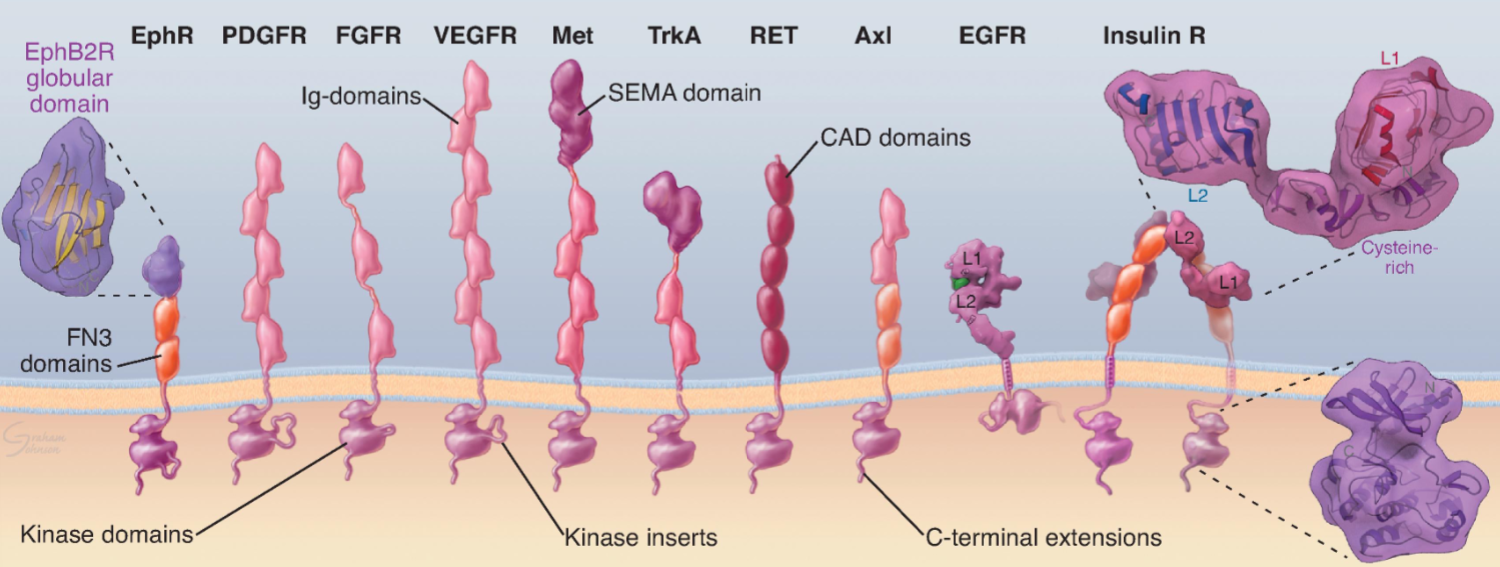
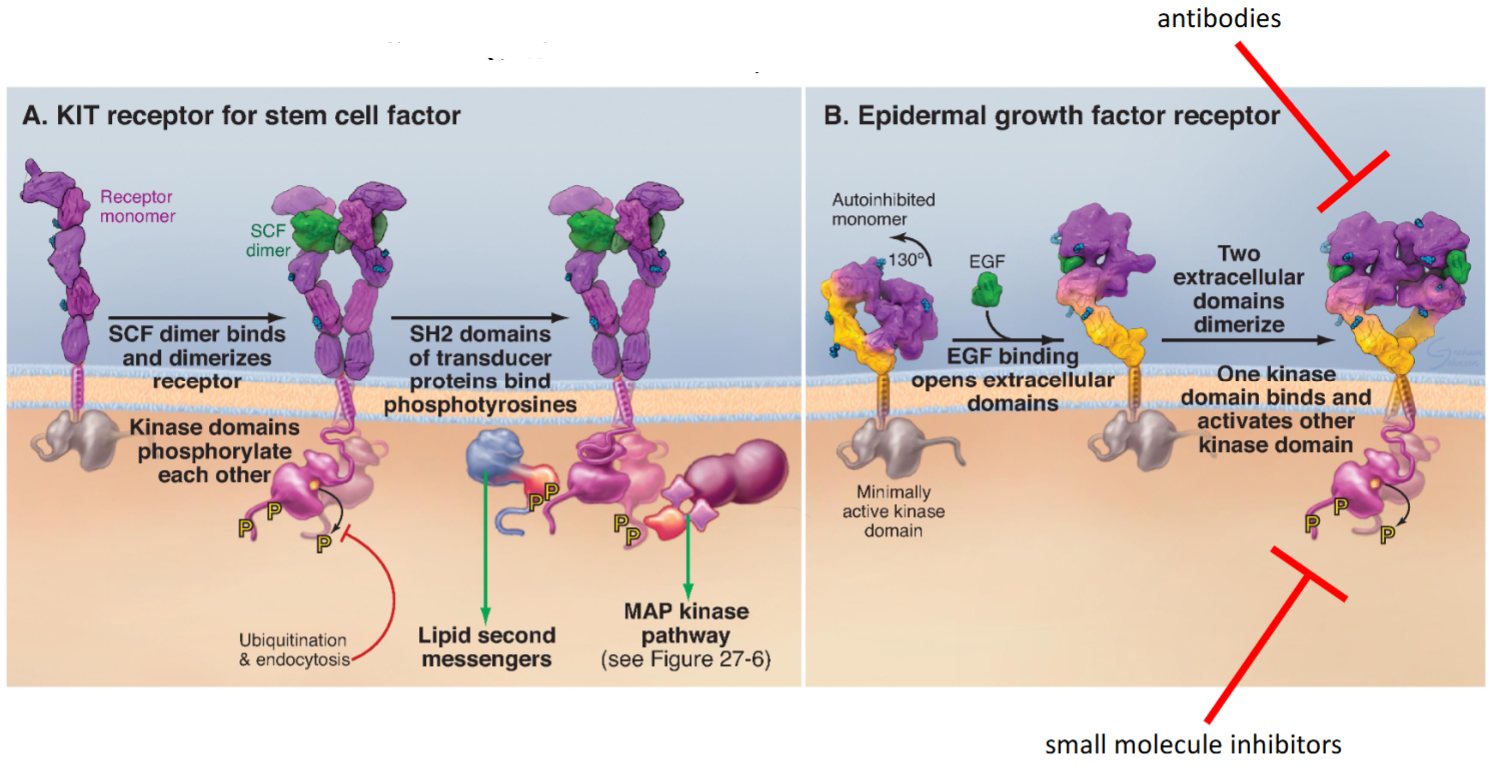
Receptor Tyrosine Kinases (RTKs)
Adrenergic Receptors
Autonomic nervous system is part of the PNS
Catecholamines
Dopamine
Norepinephrine
Epinephrine
Receptors
α1
α1A
α1B
α1D
coupled to “Gq” alpha subunit
activates PLC → hydrolyzes PIP2 to DAG + IP3 → Ca2+ release into cytoplasm
located in blood vessels → vasoconstriction
agonists
Phenylephrine
promotes vasoconstriction
useful for decreasing nasal congestion
antagonists
Prazosin
promotes vasodilation
can be used to treat high blood pressure
α2
α2A
α2B
α2C
coupled to “Gi” alpha subunit
inhibits adenylyl cyclase → reduces cAMP levels in the cytoplasm
located in presynaptic nerve endings → inhibit NE release
agonists
Clonidine
reduces the activity of the sympathetic nervous system by inhibiting the release of NE
reduces heart rate & promotes vasodilation
useful for the management of hypertension
β
β1
located in the heart → increase heart rate and force of contraction
agonists
Dobutamine
increases heart rate & force of contraction
used during the treatment of heart failure
antagonists
Atenolol
decreases heart rate & force of contraction
can be used to treat high blood pressure
β2
located in the smooth muscles (relaxation) → bronchodilation and urinary retention
agonists
Albuterol
causes bronchodilation
administered by inhalation
used to treat asthma & chronic obstructive pulmonary disease (COPD)
β3
located in detrusor muscles of the bladder → urinary retention
agonists
causes relaxation of the detrusor muscle in the bladder
increases bladder capacity
used to treat urinary incontinence
coupled to a “Gs” alpha subunit
activates adenylyl cyclase → increases cAMP levels in cytoplasm → activates Protein Kinase A (PKA)
Differently distributed amongst organs in the body
drugs that target a single receptor type can act on a specific organ system
Ways to activate adrenergic receptors
Application of adrenergic agonists
Direct-acting agonists
Elevating endogenous catecholamines
Indirect-acting agents
Both mechanisms simultaneously
Mixed-action agents
Drugs that activate a1, B1, B2, & B3 are sympathomimetic
Drugs that activate a2 receptors are sympatholytic
Nociception & General Anesthetics
Somatic Nervous System
sensory system
senses noxious stimuli
Primary afferents
neurons that relay information to the brain
Nociceptors
primary afferents that relay pain signals
Aδ
afferent fibers that are myelinated
fast signaling
C
fibers are smaller & unmyelinated
slower signaling
Tissue damage → release of ATP & glutamate → activation of Aδ and C fibers
Axons of Aδ and C fibers terminate in dorsal horn of the spinal cord
What is anesthesia?
loss of sensation & response to painful stimuli
general anesthesia
global anesthesia that includes loss of movement/awareness/consciousness
important for surgery
localized anesthesia
numbs a specific part of the body without causing amnesia & loss of consciousness
Stages of general anesthesia
Induction/sedation
period of time when patient transitions from consciousness to unconsciousness
still responsive to verbal & painful stimuli, no amnesia
Excitement
inhibition of inhibiting neurons leads to involuntary muscles movements, enhances reflexes, abnormal breathing patterns, & heart rate irregularities
General/surgical anesthesia
unconscious & amnesic to events, loss of muscle tone
may require mechanical breathing support
Deep general anesthesia/overdose
risk of respiratory & cardiovascular failure
2 routes of administration for general anesthetics
Inhalation anesthetics
gasses
molecular mechanisms for causing anesthesia are poorly understood
Halogenated ethers
Sevoflurane, Isoflurane, & Desflurane
cause loss of consciousness, analgesia, & muscle relaxation
used during induction & maintenance phases of general anesthesia during surgery
Inorganic gases
Nitrous Oxide (N2O) & Xenon
very weak ability to produce consciousness & immobility
produce significant analesia (good at blocking pain)
Typically used during maintenance phase of anesthesia
nitrous oxide is also known as “laughing gas”
Xenon has properties of ideal anesthetic, but is too rare & expensive to obtain for regular clinical use
Intravenous anesthetics
act on ligand-gated ion channels & seven-helix receptors
most contain substituted benzene rings
GABAa Allosteric Modulators
Propofol, etomidate, barbituric acid (parent structure of all barbiturates), & generic structure of benzodiazepines
Propofol, etomidate, barbiturates, & benzodiazepines all act on GABAa receptors
Act as positive allosteric modulators, cause GABAa receptors to remain open for longer in response to GAGA → more Cl- influx into neurons → hyperpolarizes neuron, making it hard for neuron to fire
Suppress neuronal activity to produce unconsciousness
Much better at producing unconsciousness than immobility or analgesia
Commonly used during the induction phase of anesthesia
Due to risk of overdose, bezos have largely replaced barbiturates
Ketamine
Acts on NMDA receptors → Blocks Ca2+ conductance through channel
Decreases glutamate-mediated neuronal excitation
Has sleep-producing, pain-relieving, and short-term memory loss effects
Used in induction and maintenance of anesthesia
One of the few drugs approved for the induction of anesthesia in c-sections; also considered drug of choice for children and burn victims
Dexmedetomidine
Agonizes α2 receptors (similar to clonidine)
Acts in CNS to inhibit the sympathetic nervous system
Useful for rapid onset of sedation, but not unconsciousness
Used drug maintenance for anesthesia as an adjunct to supplement analgesia
Local Anesthetics
Act on voltage-gated sodium channels (VGSC)
Can be closed (“resting”), open, or inactivated
Local anesthetics bind to the intracellular (cytoplasmic) side of the channel to block Na+ ion conductance
Local anesthetics preferentially bind to open or inactivated channels
Neurons with more frequent activity are inactivated first
Often weak bases
Need to cross the plasma membrane
Cross in unprotonated (uncharged) form
Only bind to VGSC in protonated (charged) form
Many are derivatives of cocaine
Cross plasma membrane & bind cytoplamic opening of VGSCs
Block propagation of action potentials
Used to produce transient loss of sensory perception without producing unconsciousness
Drugs
My Awful And Scary Iguana Took Tom’s Dog
Muscarinic Antagonists: Atropine, Scopolamine, Ipratropium, Tropicamide, Tolterodine, Darifenacin
Mechanism of action
acetylcholine (ACh)
neurotransmitter released by the parasympathetic nervous system
Binding
ACh binds to muscarinic receptors on the postsynaptic cell membrane
G-protein activation
the receptor activates a G-protein,
excitatory (Gq)
inhibitory (Gi)
Second messenger generation
the G-protein generates a second messenger
diacylglycerol (DAG)
cyclic adenosine monophosphate (cAMP)
Cellular response
the second messenger activates a protein kinase, which phosphorylates key proteins
Cellular function
the phosphorylated proteins produce a cellular response
muscle contraction
inhibition of adenylyl cyclase
Receptor subtypes
M1, M3, M5
stimulatory receptors that use the Gq protein
M2, M4
inhibitory receptors that use the Gi protein
Effects
slows the heart rate
contracts muscles in the intestines, bronchioles, and bladder
increases secretions from the salivary, gastric acid, and airway mucosal glads
causes bronchoconstriction
constricts the pupils & increases the outflow of aqueous humor
triggers glucose storage to reduce blood glucose levels
Drugs
Atropine
can cross BBB
causes CNS side effects
mechanisms of binding
blocks ACh binding through antagonizing muscarinic receptors
blocks the parasympathetic nervous system
allows the sympathetic nervous system to dominate
uses
reduce saliva & fluid in the respiratory tract
treat insecticide/mushroom poisoning
side effects
dry mouth
blurry vision
constipation
slow heartrate
Scopolamine
less basic than atropine
crosses BBB more readily
competes with ACh & other muscarinic agonists for a common binding site on the muscarinic receptor
uses
prevent nausea & vomiting
motion sickness
side effects
CNS
dizziness
blurred vision
drowsiness
Ipratropium
low lipid solubility
an ACh antagonist via the blockade of muscarinic cholinergic receptors
uses
COPD
asthma
side effects
dry mouth
throat irritation
bitter taste
nasal congestion
headache
dizziness
constipation
Tropicamide
short-acting
blocks the effects of ACh
inhibits parasympathetic NS
relaxes constrictor muscles in eye causing pupil dilation
uses
topically applied in eye exams
dilate the pupil
paralyze the eye muscle
side effects
blurred vision
light sensitivity
burning sensation in eyes
Tolterodine
antimuscarinic
competitively binds to M3 receptors in the bladder
decreases bladder contraction by decreasing detrusor muscle tone & increasing interal urethral sphincter tone
uses
urinary incontinence
side effects
dry eyes
dry mouth
drowsiness
headache
constipation
blurred vision
Darifenacin
blocks M3 muscarinic ACh receptors in the bladder
relaxes the bladder muscles to prevent urgen, frequent, or uncontrolled urination
uses
urinary incontinence
side effects
constipation
dry mouth
blurred vision
No Animals Look Very Calm
Nicotinic Agonists: Lobeline, Varenicline, Cytisine
Receptor
nicotinic cholinergic receptors in both sympathetic & parasympathetic
Mechanism of action
binds to nicotinic cholinergic receptors at the effector junction
Drugs
Lobeline
plant alkaloid
uses
sold as unregulated herbal supplement
not useful for smoking cessation
side effects
nausea
vomiting
coughing
tremor
dizziness
Varenicline
synthetic compound
elimination half-life of 24 hrs
more than 90% of the drug is eliminated from the body chemically unchanged through the glomerular filtration system & active tubular secretion
partial agonist of nicotinic receptor
functions to antagonize ability of nicotine to bind to receptors
low plasma protein binding
uses
useful for smoking cessation
side effects
irregular heartbeat
chest pain
trouble breathing
abdominal discomfort
Cytisine
plant alkaloid
elimination half-life of 5 hrs
poor bioavailability
doesn’t easily penetrate BBB
uses
smoking cessation
side effects
nausea
chest-pain
constipation
shortness of breath
News Break: No-dodging: Dogs-take Paid Attacks. Dogs, Stop!
Neuromuscular Blockers: Non-depolarizing: d-Tubocurarine, Pancuronium, Atracurium. Depolarizing, Succinylcholine
2 types
non-depolarizing
antagonists
bind to the muscle type nicotinic receptors & black ACh from activating the channels, leaving them unable to depolarize
channels are more permeable to Na+ when depolarized
specifically, the motor endplate is unable to depolarize
leads to flaccid paralysis
Depolarizing
agonist
binds to muscle-type nicotinic receptor
leads to muscle depolarization & contraction
resistant to hydrolysis by AChE → remains bound to receptor
Phase I block
sustained receptor activation prevents muscle re-polatization, leading to flaccid paralysis
Phase II block
nicotinic receptors become inactivated by prolonged activation → muscle can re-polarize, but still unresponsive to ACh
leads to flaccid paralysis similar to nondepolarizing blockers
rapid onset of action
short-lasing effects
Drugs
d-Tubocurarine
natural plant alkaloid
uses
arrow poisoning
rarely used in the clinical setting
relaxation of skeletal muscle
side effects
hypotension
asthma
muscle weakness
Pancuronium
clinically used to provide skeletal muscle relaxation
uses
during tracheal intubation
mechanical ventilation
during surgery
side effects
flushing
increased saliva
rash
muscle weakness
rapid heartbeat
low/high blood pressure
Atracurium
clinically used to provide skeletal muscle relaxation
uses
during tracheal intubation
mechanical ventilation
during surgery
side effects
skin flushing/redness
hives
itching
wheezing
shortness of breath
Succinylcholine
depolarizes the muscle & contracts it
resistant to ACh
uses
short term relaxant for intubation
side effects
rash
fainting
lightheadedness
twitching
malignant hyperthermia
Go! Meet Trey!
Ganglionic blockers: Mecamylamine, Trimethaphan
function to block activity of post-ganglionic neurons
specific effects depend on which ANS component is dominant at a particular organ
physiological state can change which system is dominant
“fight or flight” response → dominant sympathetic tone
at rest, the parasympathetic system of often the predominant tone
Drugs
Mecamylamine
voltage-depended non-competitive antagonist
blocks open nicotinic channel at ganglionic neuron
uses
occasionally used to treat hypertensive emergencies
for producing controlled hypotension during surgery
side effects
drowsiness
dizziness
lightheadedness
tiredness
blurred vision
decreased sexual bility/interest
nausea
constipuation
dry mouth
Trimethaphan
competes with ACh for receptor binding
non-polarizing competitive antagonist
uses
occasionally used to treat hypertensive emergencies
for producing controlled hypotension during surgery
side effects
diarrhea
headache
confusion
nausea
rash
dizziness
Opioid Analgesics
3 main opioid receptors
μ (MOP), δ (DOP), κ (KOP)
7-helix receptors (GPCRs) named after their ability to bind to opioids
Endogenous agonists
Neuropeptides known as Enkephalins, Dynorphins, & Endorphins
Opiate
A natural opioid derived from the poppy plant
Morphine, Codeine, Thebaine, Papaverine, Noscapine, Narceine
Opioid
Any drug that interacts with an opioid receptor
Semi-synthetic opioids are synthesized from poppy compounds
Morphine → Heroin
Thebaine → Oxycodone
Thebaine → Hydrocodone
Synthetic opioids
Fentanyl
100x more potent than morphine & 50x more potent than heroin
Methadone
a long-acting agonist that can be used to treat opioid withdrawal symptoms
Naloxone
Opioid receptor antagonists
Lifesaving application → treatment of opioid overdose,
overdose normally results in respiratory depression
Brand name: “Narcan”
Buprenorphine
Partial agonist
Can be used to treat opioid use disorder
NSAIDs
Inflammation pathway
Phospholipase A2 is hydrolyzed to arachidonic acid which acts on either cox-1 or cox-2
Cox-1
ubiquitous
consistently active
prostaglandins
thromboxane
Cox-2
upregulated in response to inflammation & injury
prostaglandins
inflammation, pain & fever
NSAIDs inhibit cox enzymes
fewer prostaglandins
reduced inflammation, pain, & fever
Asprin
non-selective NSAID
inhibit both cox-1 & cox-2
irreversibly inhibits cox-1 & cox-2
cox-1 in platelets → thromboxane a → platelet aggregation inhibition of cox-1 → loss of thromboxane A → decreased platelet aggregation → platelets lack a nucleus → effect os aspirin lasts until platelet turnover → 8-10 days
Ibuprofen
non-selective NSAID
acts on both cox-1 & cox-2
chronic inhibition of cox-1 can lead to stomach upset, ulcers, and bleeding
useful for the reduction of inflammation, pain, & fever
Naproxen
non-selective NSAID
useful for the reduction of inflammation, pain & fever
Acetaminophen
Not an NSAID
doesn’t reduce inflammation
through to reduce prostaglandin synthesis in CNS
useful for the reduction of pain & fever
lacks the cox-1 mediated side effects in the GI tract
metabolized in liver
at high concentrations, it can be metabolized into toxic by-products
liver toxicity