(LM25) Valence Bond Theory
Valence Bond Theory (VB) describes a covalent bond as the overlap of half-filled atomic orbitals, where a shared pair of electrons forms a bond between two atoms
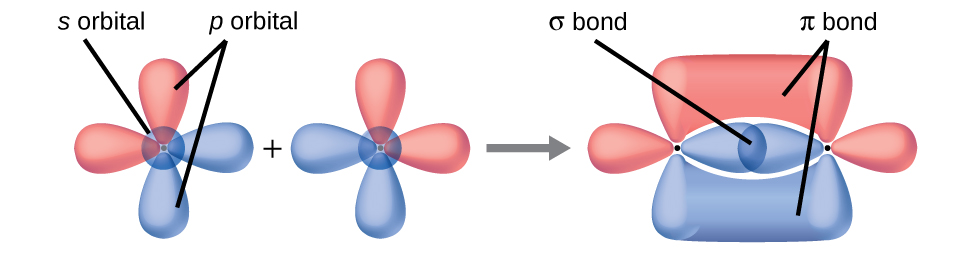
Sigma Bonds - bonds occur when the electron density is along the axis between the two atoms in the bond.
Pi Bonds - bonds occur when the electron density is off the axis formed between the two atoms in the bond.
single bond = 1 sigma
double bond = 1 sigma & 1 pi
triple bond = 1 sigma & 2 pi
hybrid orbit - mixture of orbits
sp , sp³, sp² etc.
can only form sigma bonds
will contain s orbitals
Hybridization is the mixing of atomic orbitals to create new hybrid orbitals that can form bonds, providing a better representation of molecular structure.