6.4 - transport of carbon dioxide
a small amount of carbon dioxide dissolves in water, reacts with water
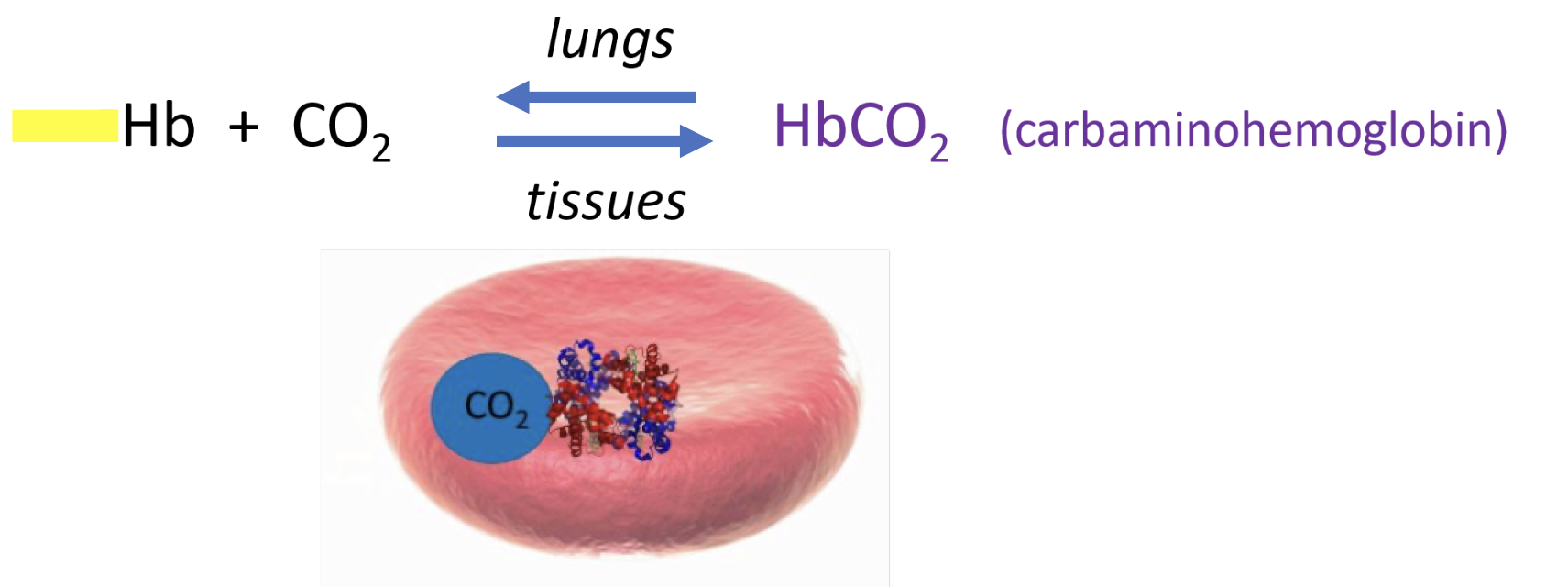
some Co2 is carried away by hemoglobin (Co2 binds to the amino group on the globin part of hemoglobin)
most Co2 combines with H2o and is transported to blood plasma and dissolves as bicarbonate ions
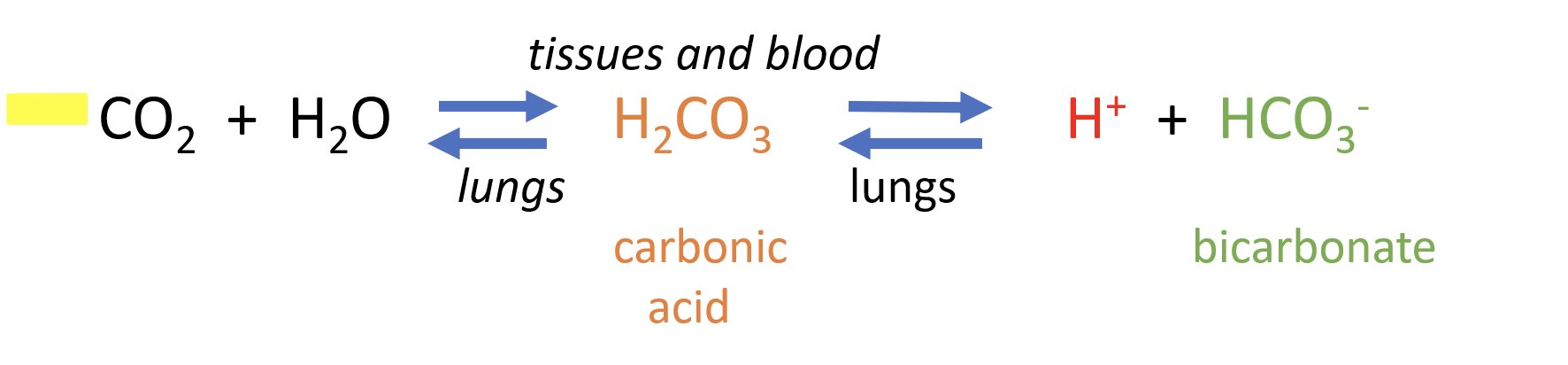
carbonic anhydrase speeds up the reaction above, ensuring a movement of Co2 from tissues to blood
BUT H+ WOULD CHANGE BLOOD PH TOO MUCH!
H+ helps dislodge O2 from Hb so it can move up into the tissues. H+ then attaches to Hb to form reduced hemoglobin
H + Hb → HHb
therefore, Hb acts as a buffer (O2 released lowers blood pH and Hb soaks up excess Hydrogen ions)
at the lungs, H+ and Co2 are both dislodged from the Hb by O2 (O2 loading into the RBC from the alveolus causes H+ to be released)
H+ recombines with the HCO3 (bicarbonate in the blood plasma) to form Co2 and H2O (Co2 is exhaled and H2O remains in the blood)
H + HCO3 → H2HCO3 (carbonic acid) → CO2 + H2O