Calcium-induced calcium release — CICR — in a cardiac/muscle cell
One-line summary
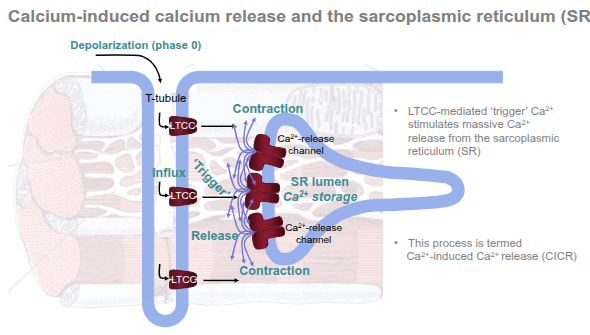
A small Ca²⁺ influx through L-type Ca²⁺ channels (LTCC) on the T-tubule membrane triggers a much larger Ca²⁺ release from the sarcoplasmic reticulum (SR) via ryanodine receptors (RyR); that cytosolic Ca²⁺ rise produces contraction and is then rapidly cleared so the cell can relax.
Step-by-step
Resting state
Cytosolic free Ca²⁺ is very low (~100 nM).
The SR lumen stores Ca²⁺ at a much higher concentration (order of 0.5–1 mM, bound to buffering proteins such as calsequestrin).
The T-tubule system sits close to junctional SR to form dyads (small signalling microdomains).
Electrical trigger (depolarisation — phase 0)
An action potential travels along the sarcolemma and into the T-tubules, depolarising the membrane locally.
Opening of L-type Ca²⁺ channels (LTCC / DHPR)
Depolarisation opens voltage-gated LTCCs in the T-tubule membrane.
A small, local inward Ca²⁺ current flows from the extracellular space into the narrow dyadic cleft. This is called the trigger Ca²⁺.
Activation of ryanodine receptors (RyR2) on the SR
The small trigger Ca²⁺ rapidly reaches the RyR channels on the junctional SR.
RyR2 are Ca²⁺-sensitive: binding of trigger Ca²⁺ increases their open probability.
Massive SR Ca²⁺ release — CICR
Once RyR2 open, a large volume of Ca²⁺ floods from the SR lumen into the cytosol (many local “Ca²⁺ sparks” sum to produce the global Ca²⁺ transient).
This amplification is the essence of calcium-induced calcium release (CICR).
Contraction
Cytosolic Ca²⁺ concentration rises (typically to ~0.5–1 μM).
Ca²⁺ binds troponin C, causing tropomyosin to move and allowing actin–myosin cross-bridge cycling → muscle contraction.
Termination of release and relaxation
RyR channels close (release diminishes).
Cytosolic Ca²⁺ is lowered by:
SERCA (SR Ca²⁺-ATPase) pumping Ca²⁺ back into the SR (major route).
Na⁺/Ca²⁺ exchanger (NCX) on the sarcolemma extruding Ca²⁺ in exchange for Na⁺.
Plasma membrane Ca²⁺-ATPase (PMCA) contributes a minor amount.
As cytosolic Ca²⁺ falls, troponin releases Ca²⁺ and the muscle relaxes.
Refilling the SR
SERCA activity refills SR stores; SR proteins (calsequestrin) help buffer high luminal Ca²⁺.
The cell is then ready for the next action potential.
Key components and terms
LTCC (L-type Ca²⁺ channel / dihydropyridine receptor) — located in T-tubules; provides trigger Ca²⁺.
RyR (ryanodine receptor, mainly RyR2 in heart) — SR release channel; mediates CICR.
Sarcoplasmic reticulum (SR) — intracellular Ca²⁺ store.
Dyad / junctional SR — very small cleft where LTCC and RyR face each other for fast signalling.
SERCA — SR Ca²⁺-ATPase that pumps Ca²⁺ back into SR (regulated by phospholamban).
NCX (Na⁺/Ca²⁺ exchanger) — extrudes Ca²⁺ across the cell membrane.
Modulation & physiological relevance
β-adrenergic stimulation (fight/flight) → PKA phosphorylation increases LTCC open probability and relieves phospholamban inhibition of SERCA → larger, faster Ca²⁺ transients and stronger, quicker contractions.
Dysregulation (excessive RyR leak or impaired SERCA) contributes to arrhythmias and heart failure because Ca²⁺ handling is central to both contraction and electrical stability.
Drugs that affect LTCC (calcium-channel blockers) alter the trigger current and thus contractility and rhythm.
Quick numeric flavour
Resting cytosolic Ca²⁺ ≈ 100 nM → peak during systole ≈ 0.5–1 μM (rough figures).
SR luminal Ca²⁺ ≈ ~0.5–1 mM (buffered).
Short final summary
CICR is a local amplification mechanism: a small extracellular Ca²⁺ entry (via LTCC) triggers large SR Ca²⁺ release (via RyR), producing the cytosolic Ca²⁺ transient that drives contraction — after which pumps and exchangers restore basal Ca²⁺ levels so the cell can relax and repeat the cycle.