L7 - Phyllosilicates and Framework Silicates
Sheet Silicates (Phyllosilicates)
Phyllosilicates are characterized by infinite sheets of Si tetrahedra. Their structure is soft and flexible due to weak van der Waals forces or hydrogen bonds between layers, allowing them to easily slide over each other; resulting in easy hydration via incorporation of water (OH- groups) within the tetrahedral sheet's hexagonal patterns. The tetrahedral sheets overlay corresponding octahedra, the charge balance is achieved differently based on the valence of the ion at the center of the octahedra:
Divalent ions (e.g., , ) require every octahedral site to be occupied.
Trivalent ions (e.g., , ) require two out of every three octahedral sites to be occupied.
Crystal structures vary:
1:1 layer silicates: balanced by a single layer of octahedra
Examples: kaolinite , and serpentine .
2:1 layer silicates: one layer of octahedra balanced by two layers of tetrahedra.
Charge misbalance and large spaces allow large mono- or divalent ions (e.g., , ) to fit between layers.
Examples: talc, muscovite, phlogopite, and montmorillonite.
2:1:1 layer silicates: an additional octahedral layer is sandwiched between each 2:1 T-O-T layer.
Can also carry large cations and include chlorite group minerals.
Key Phyllosilicates:
Clay minerals - Consist of small-grained phyllosilicates (<2 µm) that readily carry water and are products of weathering of anhydrous aluminosilicates.
Examples include
Kaolinites -
Muscovite -
Serpentine -
Framework Silicates (Tectosilicates)
Framework silicates constitute the majority of crustal rocks. They involve interconnected networks of Si tetrahedra bonded at all vertices to four other tetrahedra. Complete bonding leads to charge balance, exemplified by quartz .
Quartz can undergo phase transitions, where its structure changes with temperature, pressure, or composition. An example is the melting of ice into water around .
Low quartz (-quartz): Trigonal crystal structure at room temperature and pressure.
High quartz (-quartz): Hexagonal structure at higher temperatures.
The transition between low and high quartz is a displacive phase transition involving minor atomic shifts via slight tetrahedra rotations, occurring rapidly with little energy input. Quenching cannot prevent it, as no bonds are broken and minimal atomic movement occurs, meaning the process involves very little energy.
Tridymite: High temperature form of quartz.
Cristobalite: Even higher temperature form of quartz.
Transitions to tridymite and cristobalite are reconstructive phase transitions. These breaks/reforms bonds, requires significant energy and time. Quenching this form of forms preserves the structure, trapping tetrahedra in their cristobalite form.
Thermodynamics vs. kinetics: mineral phases depend on both energy considerations and the time available.
Feldspars
Feldspars () are other framework silicates. Al substitutes into tetrahedra, balanced by large monovalent cations (e.g., , , ).
Alkali feldspars: solid solution between orthoclase and albite .
Plagioclase feldspars: solid solution between albite and anorthite .
Together, feldspars make up 50-60% of all igneous rocks
Interlocking tetrahedral chains create void spaces for accommodating large cations (crankshaft structure), where interconnected 4-member rings of tetrahedra form this structure. Solid solutions have different mineral names (e.g., labradorite, anorthoclase, sanidine).
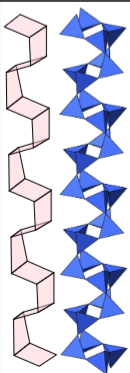
Feldspar crystal structures have a kinetic component; the ordering of Al and Si can be random at high temperatures, meaning there is symmetry, (preserved by quenching) or periodic during slow cooling, resulting in no symmetry. Feldspar mineralogy traces a rock's thermal and kinetic history.