C3
Types of chemical bonds (Overview)
Ionic → Involves oppositely charged ions and occurs between a metal and a non metal
Covalent → Involves sharing a pair of electrons between two non metals
Metallic → Involves sharing delocalised electrons between two metals/alloys
Ionic bonding:
Metals lose electrons to become a positively charged ion and non metals gain electrons to become a negatively charged ion. Held together by electrostatic forces
For example, Group 1 metals react with Group 7 elements where the G1 metals lose 1 electron and give it to the G7 element to both achieve their respective stable electronic structures
G1 metal → Lose 1 electron to have a full electron shell
G7 metal → Gain 1 electron to complete its electron shell
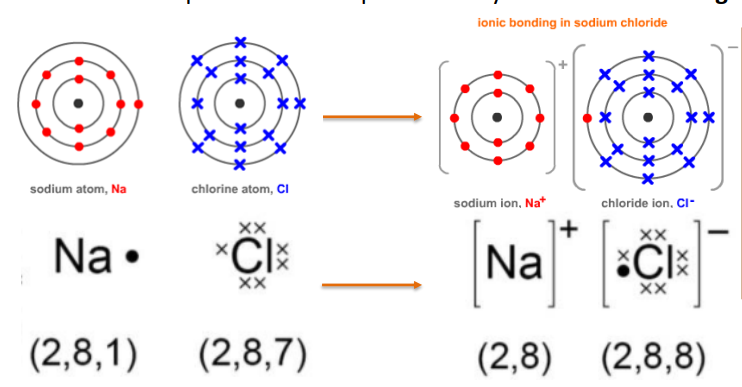
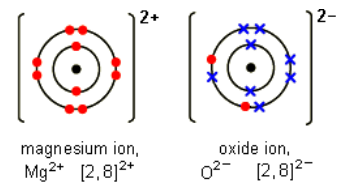
Bonding in Magnesium Oxide → Each magnesium loses two electrons and each oxygen gains two electrons. Magnesium ions have the formula Mg2+, while oxide ions have the formula O2-. Therefore, it gives out the formula MgO as both of the “2” charges cancel each other out
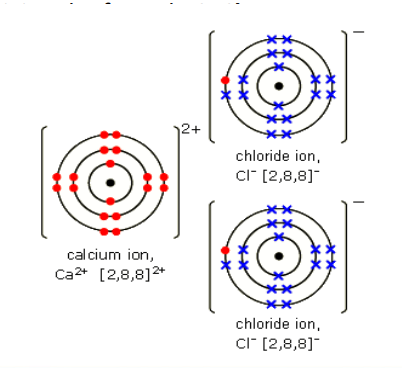
Bonding in Calcium Chloride → Each calcium atom needs to lose two electrons but each chlorine atom needs to gain only one electron. This means that two chlorine atoms react with every one calcium atom
 Ionic compound - Giant structure of ions. They are held together by strong electrostatic forces of attraction between oppositely charges ions. These forces act in all directions in the lattice.
Ionic compound - Giant structure of ions. They are held together by strong electrostatic forces of attraction between oppositely charges ions. These forces act in all directions in the lattice.
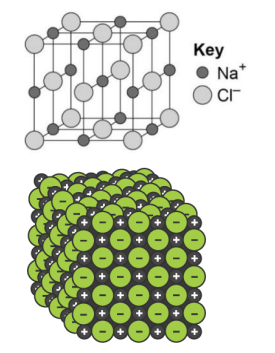 Giant ionic lattice - Ionic compounds with regular structures
Giant ionic lattice - Ionic compounds with regular structures
Properties of ionic compounds:
High melting and boiling points → Large amounts of energy needed to break the many strong bonds and overcome the electrostatic attraction
Conduct electricity → Only when compound is in molten form or dissolved in water where the ions are free to move and carry charge
Covalent Bonding:
When atoms share pairs of electrons, they form covalent bonds which are strong bonds.
Examples of covalently bonded substances are:
Small molecules
Very large molecules
Giant covalent structures
Polymers → Made up of many small reactive molecules that bond to each other covalently to form long chains. It is solid at room temperature as it has strong intermolecular forces
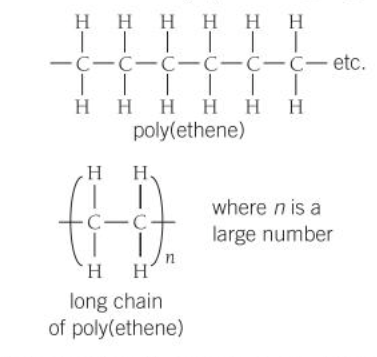
Ways to show covalent bonded substances:
Dot and cross with outer shells as circles
Good → Show which atom the electrons in the bonds come from
Bad → All electrons are identical
2D with bonds
Good → Show which atoms are bonded together
Bad → It shows the bond incorrectly at 90°
3D ball and stick model
Good → Attempts to show the correct angle of the bond
Bad → Shows the impact of the lone pair
Small molecules:
Structure → Weak forces between the molecules and they are overcome when they change state
Properties → Low melting and boiling points so only small amounts of energy are needed to break the intermolecular forces. Most small molecules are liquids or gases. They also do not conduct electricity as the particles don’t have an overall electrical charge
Examples include → Water, Amino acids, Glucose, Sugars, Lipids
Giant Covalent Structures:
Structure → Linked by strong covalent bonds and these bonds must be broken for a solid to melt or boil
Properties → Very high melting and boiling points so large amounts of energy needed to overcome these covalent bonds. They do not conduct electricity as the particles don’t have an overall electrical charge
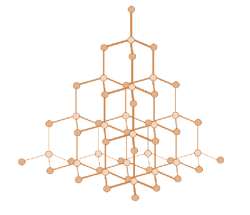
Diamond:
Structure → Each carbon atom forms four covalent bonds with other carbon atoms
Properties → Because of its structure, it is very hard so it is used for jewellery and cutting tools. Very high melting and boiling point so lots of energy is needed to break the covalent bonds. It can’t conduct electricity because there are no free electrons or ions to carry a charge.
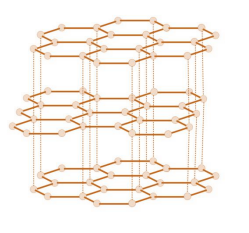
Graphite:
Structure → Carbon atom forms three covalent bonds with three other carbon atoms, forming layers of hexagonal rings which have no covalent bonds between the layers.
Properties → Soft and slippery as layers can easily slide over each other because the weak forces of attraction between the layers are easily broken making it useful as a lubricant. It can conduct electricity as one electron from each carbon atom is delocalised
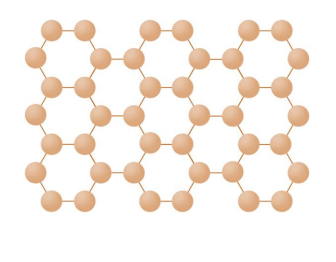
Very Large molecules:
Graphene:
Structure → single layer of graphite ,a layer of inter-locking hexagonal rings of carbon atoms one atom thick.
Properties → Excellent conductor of thermal energy and electricity. Low density. Strong. Used in electronics industry
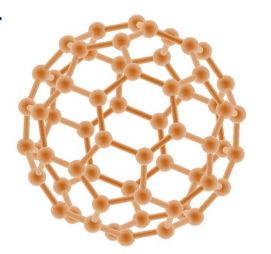
 Fullerenes:
Fullerenes:Structure → molecules of carbon with hollow shapes. The structure is based on hexagonal rings of carbon atoms, but may have 5 or 7 carbon rings
1st discovered was called buckminsterfullerene
Properties → Carbon nanotubes are cylindrical fullerenes with very high length compared to their diameter. This makes them useful for nanotechnology, electronics and materials, good conductors of electricity and excellent lubricant, drug delivery ,
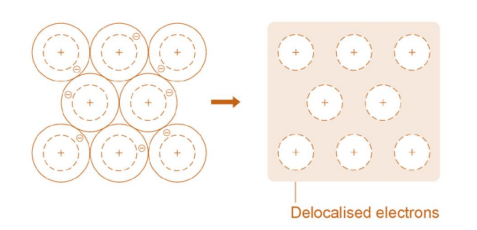
Metallic Bonding:
Sharing of delocalised electrons between two metals. Electrons in the outer shell of metal atoms are delocalised and are free to move throughout the structure. Sharing delocalised electrons leads to strong metallic bonds
Properties:
High melting and boiling points → Giant structure of atoms with strong metallic bonding
Malleable → layers of atoms (or ions) in a giant metallic structure can slide over each other
Ductile → layers of atoms (or ions) in a giant metallic structure can slide over each other
Conductors of heat and electricity → Delocalized electrons
Alloys:
Metal mixed with other elements
Harder than pure metals because there are different sized atoms of the metals distort the layers in the structure, making it more difficult for them to slide over each other
Particle Theory
All matter is made up of tiny particles and describes the movement and distance between particles
Solids:
Particles packed together closely
In a regular pattern
Vibrate in fixed positions
Liquids:
Particles are close together
Random arrangement
Move around each other
Gases:
Particles are far apart
Random arrangement
Move quickly because it has more kinetic energy
Melting and freezing occurs at the melting point
Boiling and condensing occurs at the boiling point
The stronger the intermolecular forces between the particles the higher the melting and boiling point of the substance
Problems with the particle model of matter:
There are no forces shown
All particles are shown as spheres
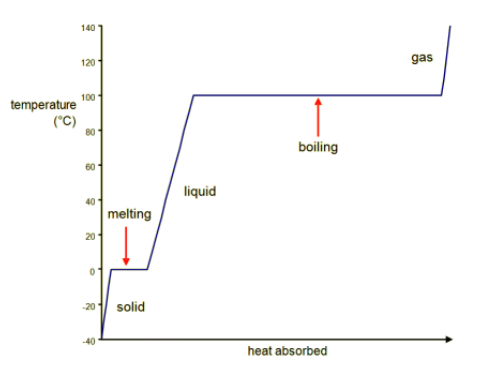
a heating curve of a solid, which shows the temperature of a substance plotted against the amount of energy it has absorbed
A substance must absorb heat energy so that it can melt or boil.
The temperature of the substance does not change while changing state, even though energy is still being transferred.
Nanoscience:
The study of small particles that are between 1 and 100 nanometres in size. Particles consisting of fewer than 100 atoms are often called nanoclusters
1Nanometre → 1×10^-9m
As the side of cube decreases by a factor of 10 the surface area to volume ratio increases by a factor of 10.
Nanoparticles have a high surface area to volume ratio so it is more reactive than materials with normal sized particles resulting in a more sustainable approach to industry as less resources are being used up
Uses of nanoparticles are: medicine, in electronics, in cosmetics and sun creams, as deodorants, and as catalysts.,
Nanoparticles may be harmful as they can diffuse into the bloodstream via the skin and cause harmful reactions inside the body