Topic 6 Regents Chemistry Review: Bond Polarity
Topic Overview
Chemical bonds provide the “glue” that holds all compounds together. There are different types of chemical bonds, and these different bond types account for the different properties of substances. In this section you will learn how the electron structure of atoms helps explain many aspects of chemical bonding. You will also use the Lewis Dot Diagrams to aid in your understanding of electronic structure and its role in bonding.
Energy and Chemical Bonds
Chemical bonds are the forces that hold atoms together in a compound. Energy is required to overcome these attractive forces and separate the atoms in a compound. Thus, the breaking of a chemical bond is an endothermic process. If energy is required to break a bond, then the opposite process of forming a bond must release energy. The formation of a bond is an endothermic process.
When a chemical bond is formed, the resulting compound has less potential energy than the substances from which it was formed. Energy is always released when a bond is formed. The greater the energy released during the formation of a bond, the greater is stability. Consider the following two reactions.
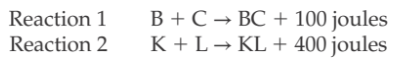
The bond formed in Reaction 2 is more stable than the bond formed in Reaction 1. It takes 400 joules to break apart Compound KL, but only 100 joules to break apart compound BC.'
Lewis Dot Diagram
In the next sections you will study the two types of bonds that commonly form. These two bond types result from the transfer of electrons from one atom to another and from the sharing of valence electrons from one atom to another and from the sharing of valence electrons between atoms. A simple modeling technique known as a Lewis Dot Diagram provides an easy method for showing how electrons are transferred or shared during bond formation. Learning to draw and interpret these diagrams is an important skill.
A Lewis Dot Diagram, also called an electron dot diagram, consists of a chemical symbol surrounded by one to eight dots representing valence electrons
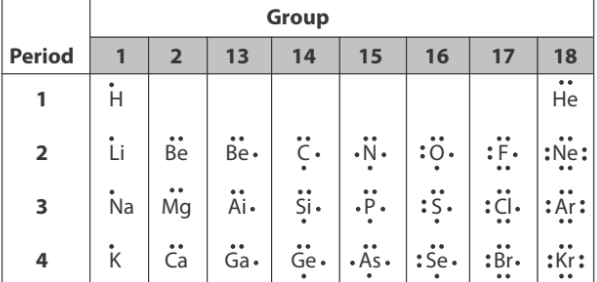
There are slight variations in how electrons are arranged around the kernel in an electron dot diagram. Often, the first two electrons are placed at the 12 o'clock position. As the next three electrons are added, they are placed singly at the 3, 6, and 9 o'clock positions. The last three electrons are added to make pairs in the same order, at the 3, 6, and 9 o'clock positions.
When atoms gain or lose electrons they become charged particles called ions. When metals react, they do so by losing their valence electrons. Thus, there aren't any dots in the electron dot diagrams of the ions for sodium (Na+), magnesium (Mg2+), or aluminum (Al3+). The square brackets around the kernel and the ionic charge written as a superscript outside the brackets indicate the diagram is that of an ion. Whereas metals lose electrons to form ions, nonmetals gain electrons to form ions. The valence electrons gained when a nonmetal ion forms are shown in its electron dot diagram. It is common to show the added valence electrons with a different symbol than that used to represent the atom's originally present valence electrons.
The number of valence electrons to use in an electron dot diagram can also be determined by consulting the periodic table. The periodic table often lists the electron configuration for the elements using a modified Bohr model. For example, the electron configuration for sodium (Na) is listed as 2-8-1. This notation indicates that there is only one outermost, or valence electron for a sodium atom. The other 10 electrons belong to the kernel of the atom. The corresponding electron dot diagram for the sodium atom would show this one valence electron as a single dot, outside the kernel.
Vocabulary
Asymmetrical Molecule - A molecule with an uneven distribution of electrons.
Covalent Bond - A bond formed by the sharing of electrons between atoms.
Dipole-Dipole Forces - Attractive forces between polar molecules.
Double Covalent Bond - A bond where two pairs of electrons are shared.
Hydrogen Bond - A strong attraction between a hydrogen atom and an electronegative atom.
Ion - An atom or molecule with a net electric charge due to the loss or gain of electrons.
Ionic Bond - A bond formed by the transfer of electrons from one atom to another.
Lewis Dot Diagram - A diagram showing the bonding between atoms and the lone pairs of electrons.
London Dispersion Forces - Weak intermolecular forces due to temporary dipoles.
Malleability - The ability of a substance to be deformed or molded.
Metallic Bond - A bond where free electrons are shared among a lattice of metal atoms.
Multiple Covalent Bond - A bond where more than one pair of electrons is shared.
Nonpolar Covalent Bond - A bond where electrons are shared equally between atoms.
Octet - A stable configuration with eight electrons in the valence shell.
Octet Rule - The principle that atoms tend to bond to achieve eight valence electrons.
Polar Covalent Bond - A bond where electrons are shared unequally between atoms.
Symmetrical Molecule - A molecule with an even distribution of electrons.
Triple Covalent Bond - A bond where three pairs of electrons are shared.