Fluid
About two-thirds of the body’s fluid is located in cells and is called intracellular fluid (ICF). The other third is called extracellular fluid (ECF).
Body fluid is categorized into two main compartments: intracellular fluid (ICF) and extracellular fluid (ECF). The ICF accounts for approximately two-thirds of the body's total fluid, as it is the fluid contained within cells, while the remaining third is the ECF.
Regulation of Body Water Gain
The body regulates water balance mainly by adjusting fluid intake. Thirst, the primary control mechanism, is triggered by dehydration (water loss > water gain). The renin-angiotensin II pathway stimulates the hypothalamic thirst center in response to decreased blood volume and pressure. Metabolic water production is determined by the rate of aerobic cellular respiration, which reflects ATP demand.
The term body fluid refers to body water and its dissolved substances.
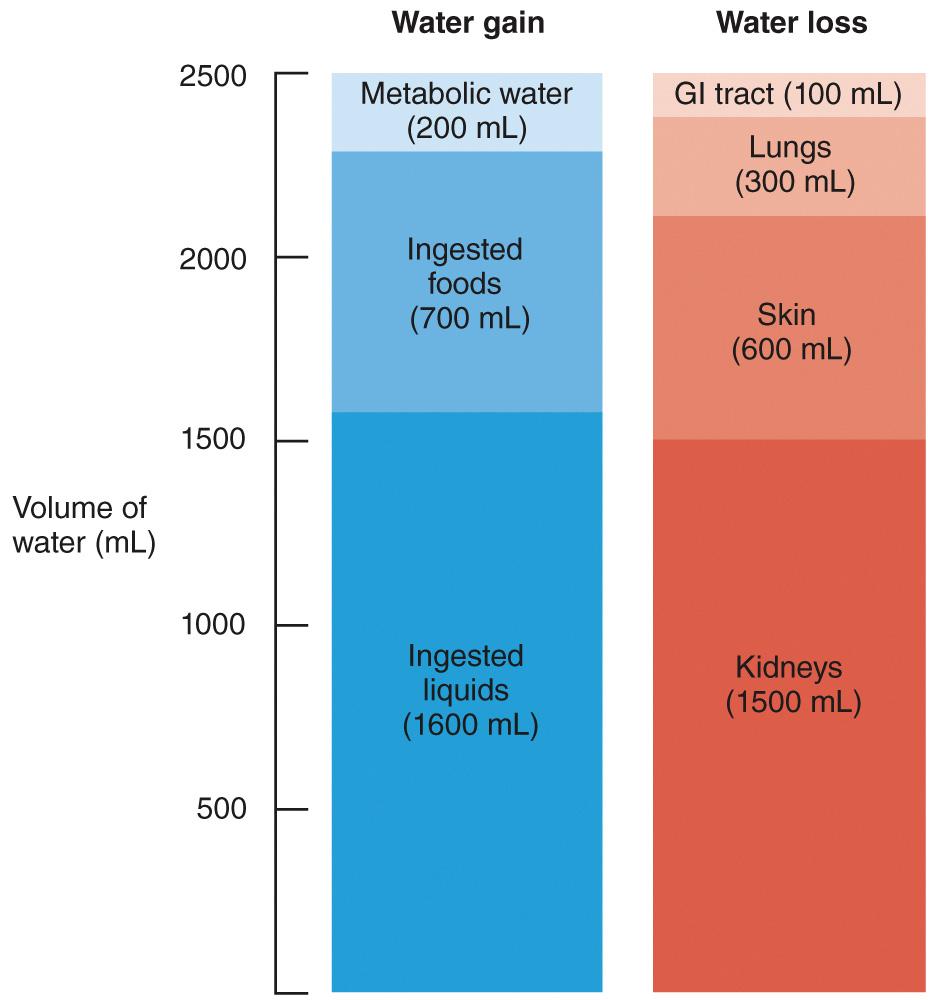
Normally, daily water loss equals daily water gain.
Regulation of Body Water Gain
The body primarily controls its water balance by adjusting the amount of water consumed.
Sources of Water Gain:
Metabolic Water: The volume of water produced through aerobic cellular respiration is directly proportional to the body's demand for ATP.
Water Intake: Adjusting the volume of fluid intake is the main mechanism for regulating the overall body water balance.
Stimulus for Water Intake (Thirst):
Dehydration, which occurs when water loss surpasses water gain (Figure 27.3), triggers thirst sensations.
The renin-angiotensin II pathway is a key mechanism that stimulates the thirst center in the hypothalamus. This pathway is activated in response to a decrease in blood volume, and consequently, a drop in blood pressure (Figure 27.3).
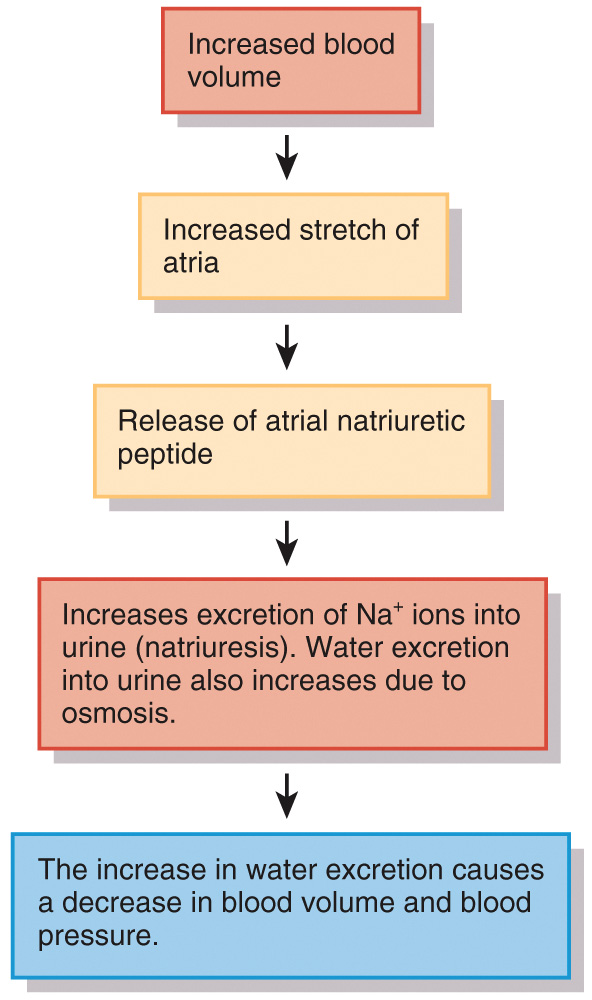
ANP increases the excretion of Na+ ions into urine (natriuresis)
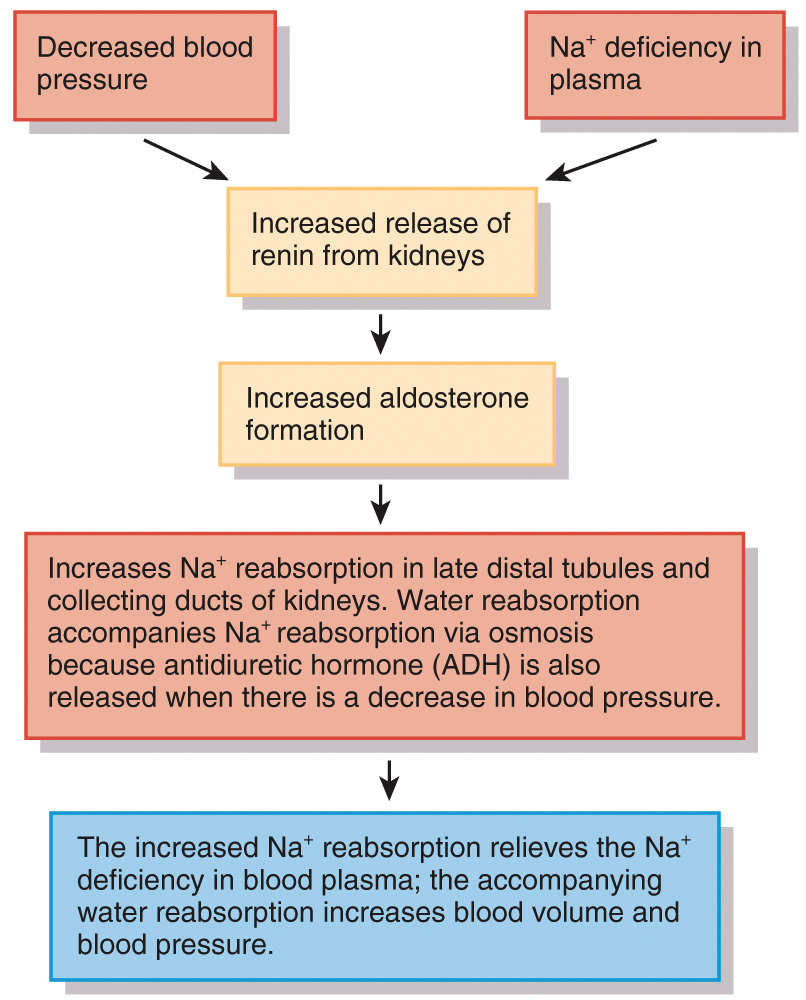
Aldosterone increases the amount of Na+ reabsorption in the kidneys.
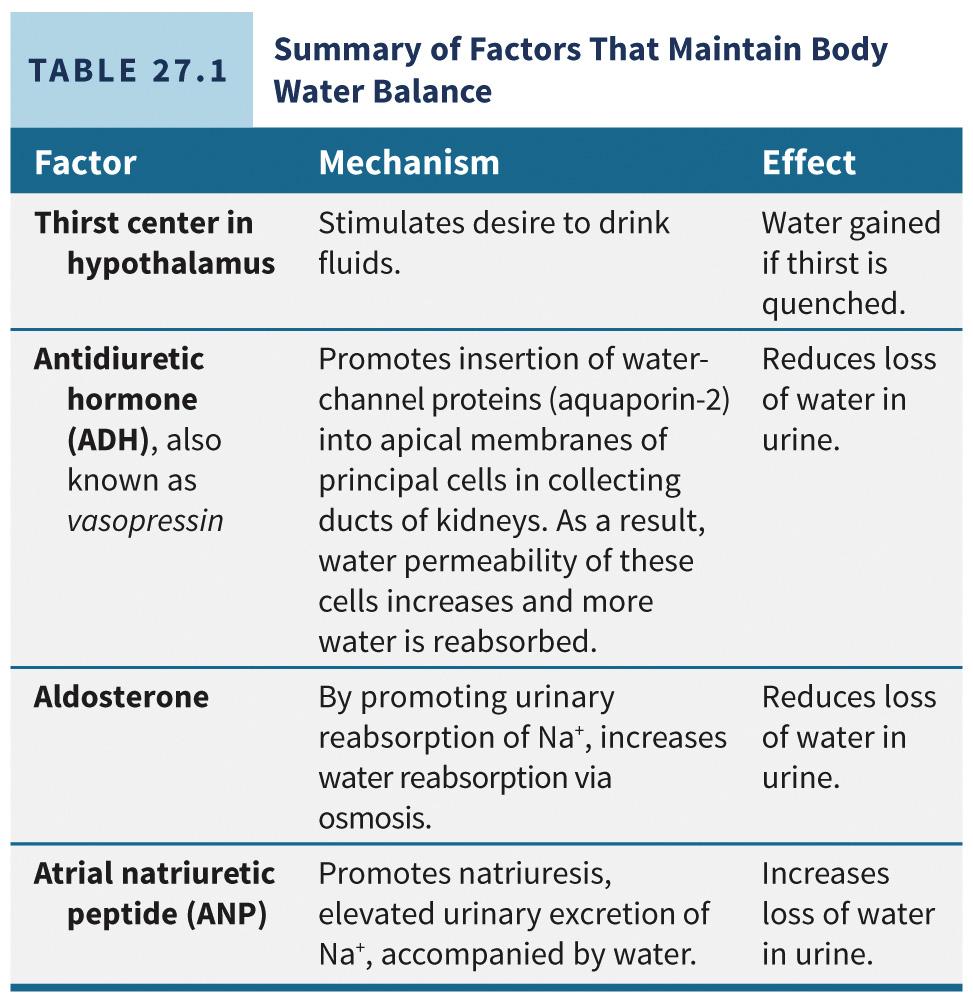
Changes in the osmolarity of intracellular and interstitial fluids can cause a fluid imbalance between them.
This change in osmolarity is most frequently caused by a shift in the concentration of sodium (Na+) and chloride (Cl-).
Consuming water faster than the kidneys can eliminate it may result in water intoxication.
The repeated use of enemas poses a risk for developing fluid and electrolyte imbalances.
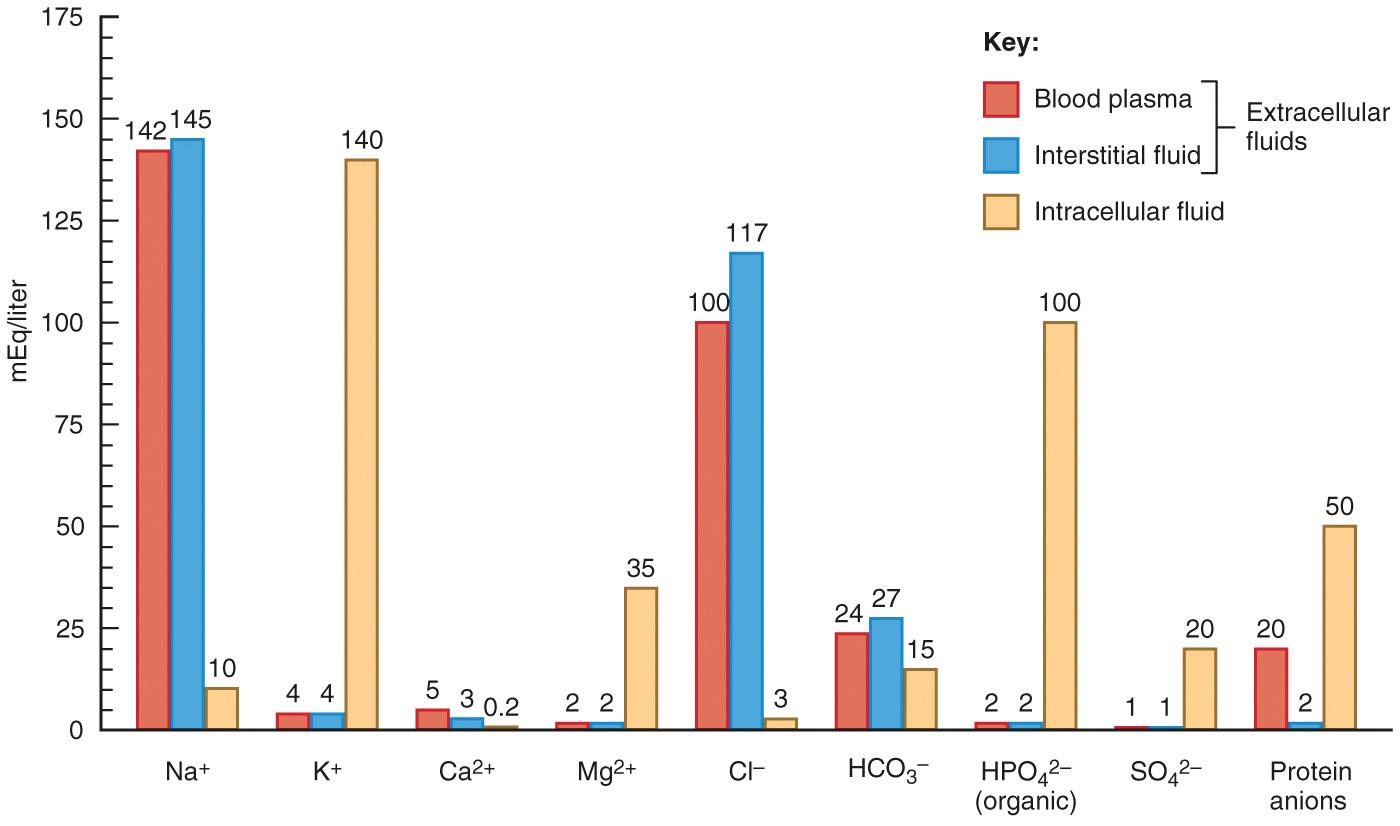
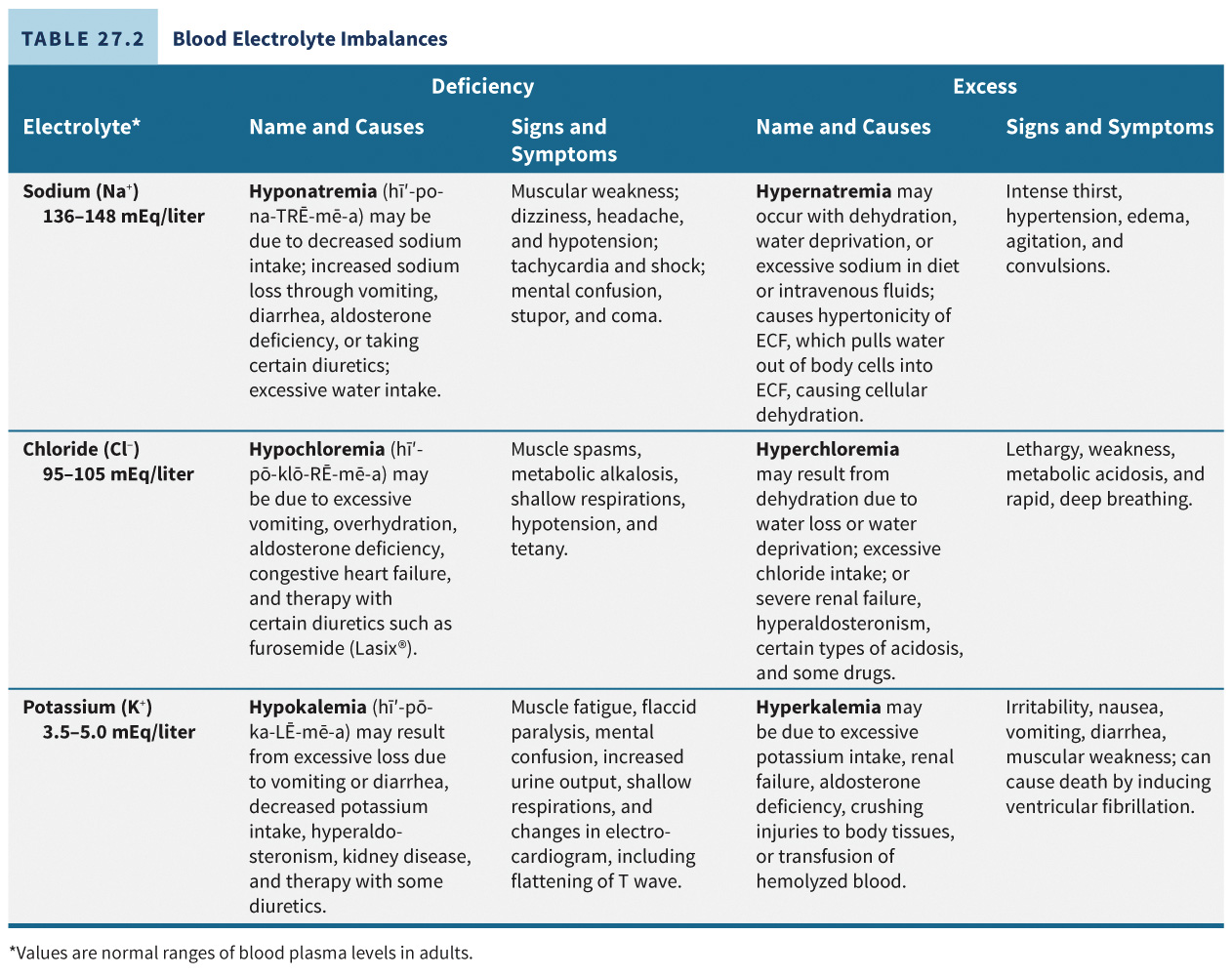
The electrolyte compositions of extracellular and intracellular fluids are distinct.
Calcium is the body's most abundant mineral, primarily serving as an extracellular ion.
Structural Role: Forms bones and teeth.
Physiological Processes:
Blood coagulation (clotting)
Neurotransmitter release
Maintenance of muscle tone
Excitability of nervous and muscle tissue
Regulation: Plasma concentration is tightly regulated, mainly by parathyroid hormone and calcitonin.
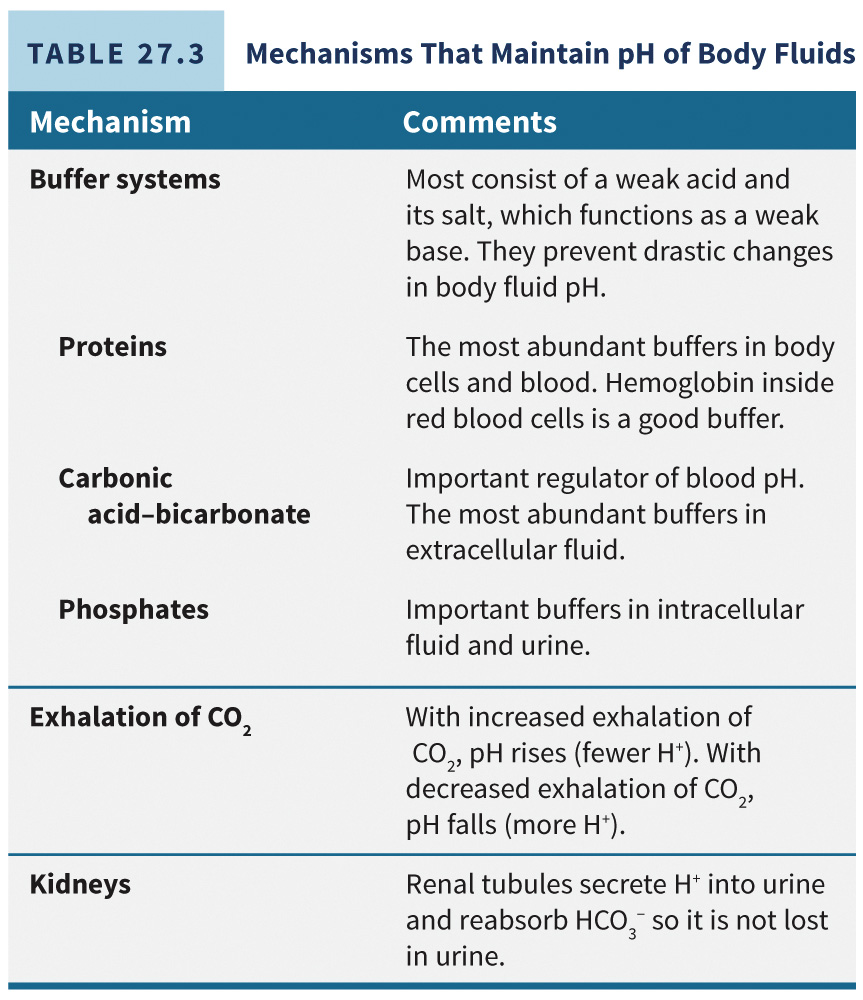
Buffer systems play a crucial role in preventing rapid and drastic shifts in the $\text{pH}$ of body fluids. They achieve this by converting strong acids and bases into weak ones, with this action occurring almost instantaneously (within fractions of a second).
Most buffer systems are composed of a weak acid and the salt of that acid, which functions as a weak base.
The body employs several important buffer systems:
Protein Buffer System: This is the most prevalent buffer found in both intracellular fluid and plasma. Within red blood cells, the protein hemoglobin is a particularly effective buffer for carbonic acid.
Carbonic Acid-Bicarbonate Buffer System: This system, which relies on the bicarbonate ion, is a key regulator of blood $\text{pH}$.
Phosphate Buffer System: This system is significant in regulating $\text{pH}$ within red blood cells as well as in the kidney tubular fluids.
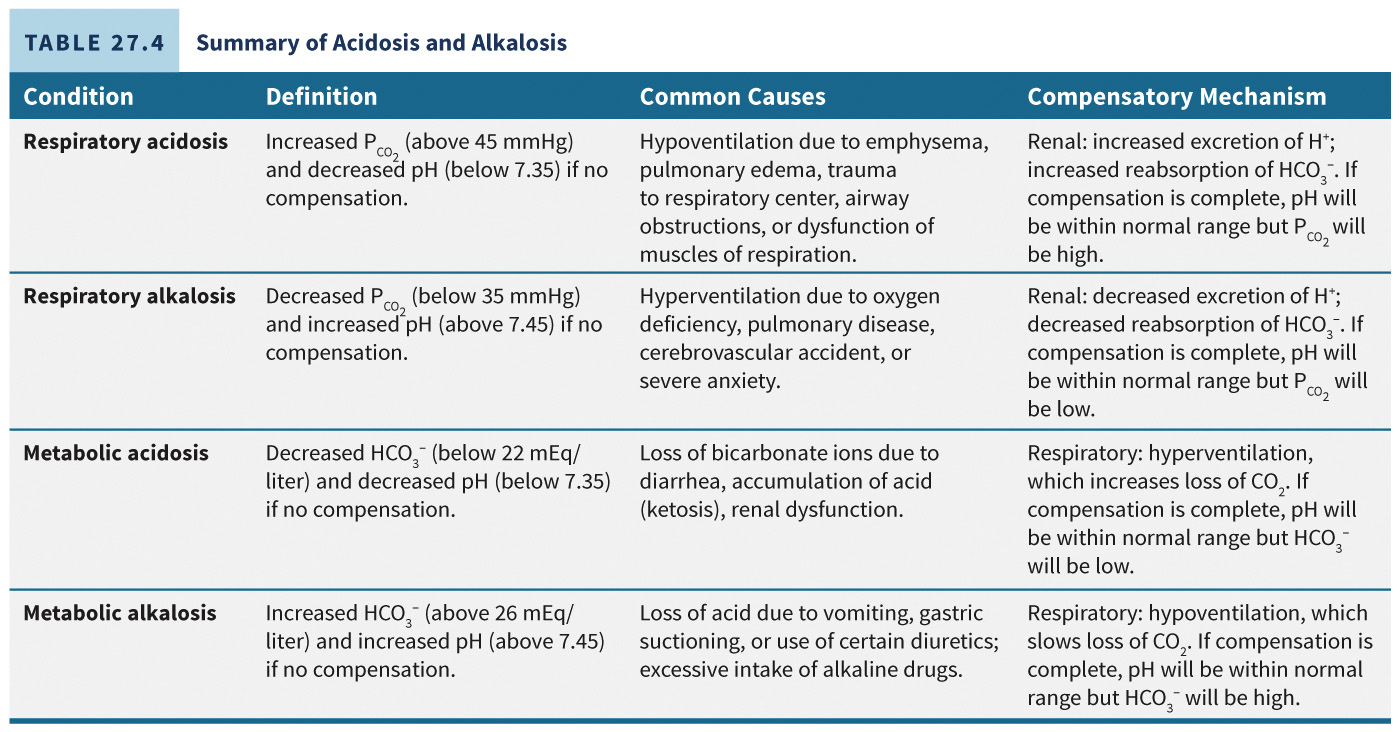
Metabolic alkalosis is characterized by high bicarbonate, caused by nonrespiratory acid depletion (e.g., severe vomiting) or excessive alkaline medication intake. Table 27.4 summarizes both acidosis and alkalosis.
Body fluids make up about 55–60% of the total body weight in lean adults.
Water is the main component of all bodily fluids.
These fluids are divided into two main categories:
Intracellular fluid (ICF): Accounts for roughly two-thirds of the total and is located inside the cells.
Extracellular fluid (ECF): Makes up the remaining one-third and is found outside the cells.
Fluid balance is achieved when the different body compartments hold the correct quantity of water, distributed appropriately based on the unique requirements of each compartment.
-
Normally, daily water loss equals daily water gain.
Movement of Water Between Body Fluid Compartments
Changes in osmolarity, most often caused by a shift in the concentration of Na+ and Cl-.3, can lead to a fluid imbalance between the intracellular and interstitial compartments.
For example, consuming water more rapidly than the kidneys can excrete it may result in water intoxication (see Figure 27.7). Additionally, the repeated use of enemas carries a risk of causing fluid and electrolyte imbalances (Clinical Connection).
Osmosis and Osmolarity: The movement of water between the cell's interior (intracellular fluid) and the surrounding fluid (extracellular fluid) is driven by osmosis, which is caused by differences in solute concentration (osmolarity).
Hypertonic Extracellular Fluid:
Condition: Extracellular fluid has a higher solute concentration (increased osmolarity) than the intracellular fluid.
Result: Water moves out of the cells, causing them to shrink.
Hypotonic Extracellular Fluid:
Condition: Extracellular fluid has a lower solute concentration (decreased osmolarity) than the intracellular fluid.
Result: Water moves into the cells, causing them to swell.
Causes of Osmolarity Changes: Fluctuations in extracellular fluid osmolarity are most often due to changes in the concentrations of sodium ($\text{Na}^+$) and chloride ($\text{Cl}^-$) ions, which are the main contributors to its total osmolarity.
Functions of Electrolytes in Body Fluids
Water Osmosis: Regulate water movement (osmosis) between fluid compartments due to higher concentration than nonelectrolytes.
Acid-Base Balance: Essential for maintaining the acid-base balance needed for normal cellular function.
Electrical Activity: Carry electrical currents for:
Generating action and graded potentials.
Controlling hormone and neurotransmitter secretion.
Development.
Enzyme Cofactors: Several ions act as cofactors necessary for optimal enzyme activity.
These cells are crucial for regulating the pH of body fluids.
They maintain balance by excreting:
Excess hydrogen ions H+ when the pH is too low (acidic).
Excess bicarbonate ions HCO3– when the pH is too high (alkaline).
The specific mechanisms for maintaining body fluid pH are detailed in Table 27.3.
Acid-Base Imbalances: The normal pH range of systemic arterial blood is between 7.35-7.45.