ASVAB Chemystery
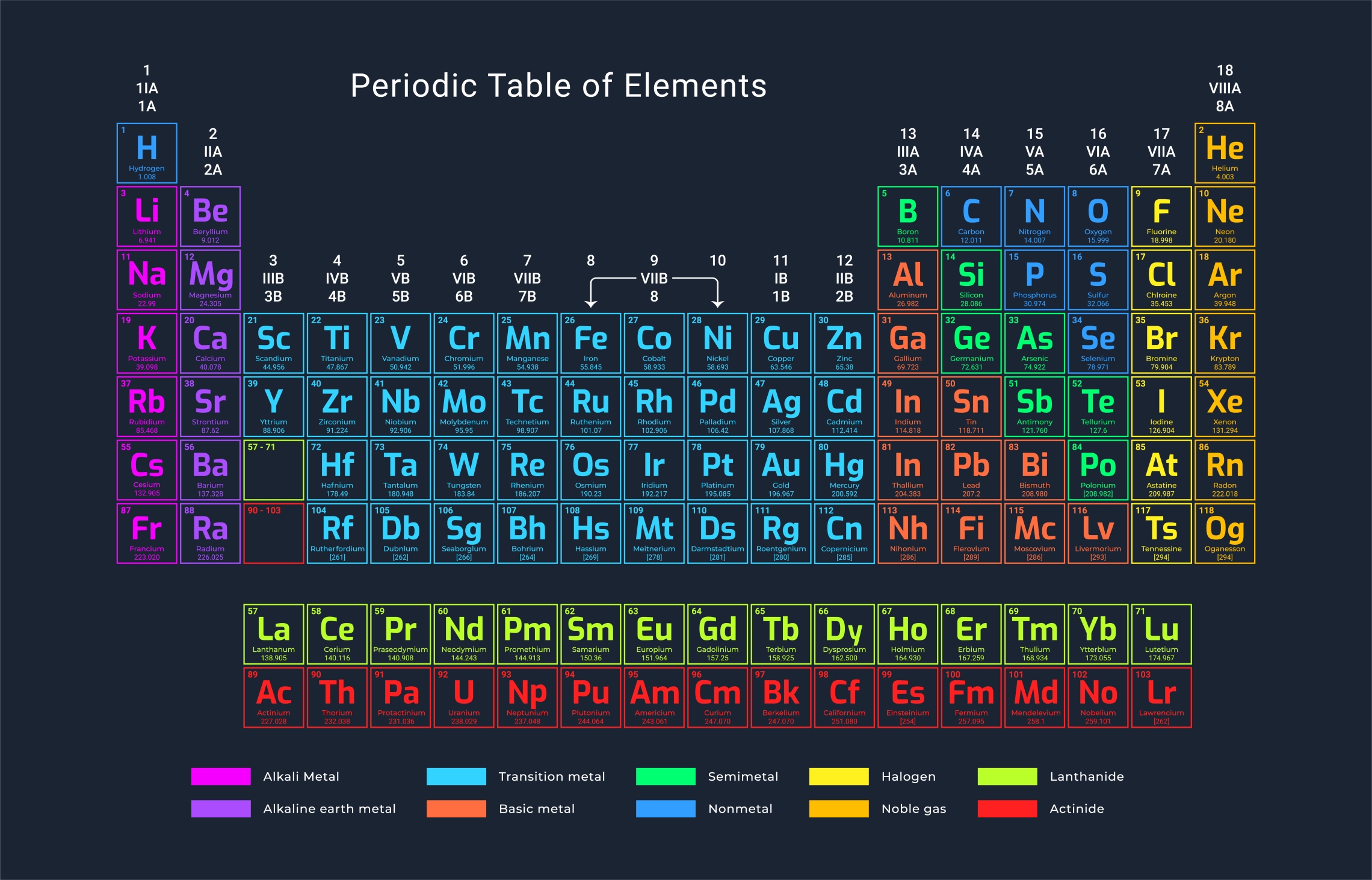
The rows of the periodic table are called periods and correspond to the number of occupied electron shells in an atom. The 1st period has 1 occupied shell, the second period has 2 occupied shells, and so on.
Electrons generally occupy the lowest shell they can and only move to higher shells when there’s no room left in the lower ones. This minimizes the energy of an atom (think about gravity, objects fall to occupy the lowest position relative to the Earth’s surface, minimizing their potential energy).
A column is called a group. All the elements in a group have the same number of electrons in their outer (valence) shell. This means they have similar chemical properties. The first group has one electron in its outer shell, the second group has two, etc. However the column number is not always the number of valence electrons, the number of valence electrons in a given element can be found by the number at the bottom of each column label on the picture above (e.g. 1A has 1 valence electron, 7B has 7).
The first group contains alkali metals, which are highly reactive and tend to lose their single valence electron easily during chemical reactions.
The 18th (far right) group contains the noble gases which are chemically nonreactive because their valence shells are full.
Atoms of the same element can have different numbers of neutrons, these different varieties are called isotopes.
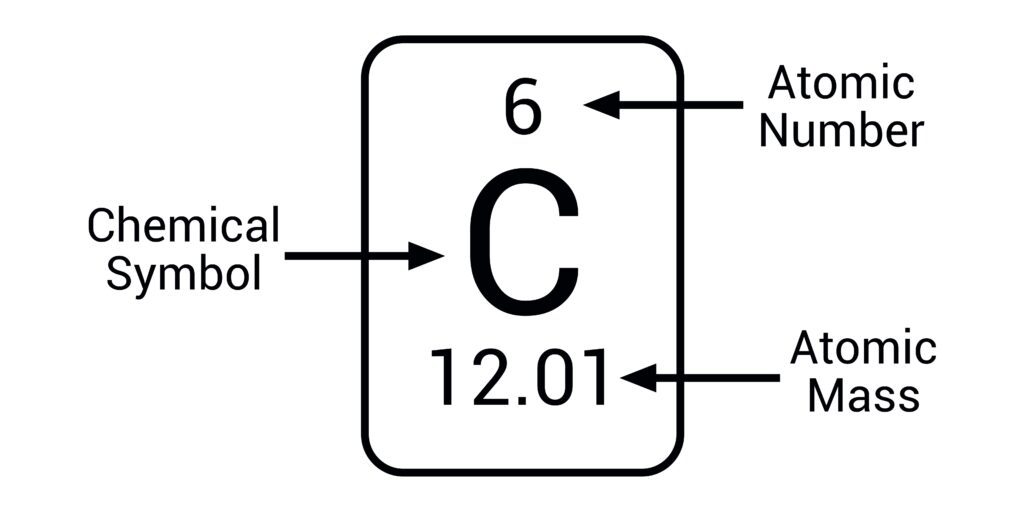
Atomic number = number of protons in the nucleus, this defines the element.
Atomic mass = approximately the number of protons + the number of neutrons. Different isotopes have different atomic mass, the number listed on the periodic table is the average atomic mass for an element based on the relative masses and concentrations of the different isotopes.
The number of electrons in an element is usually the same as the number of protons, in which case it’s electrically neutral. Otherwise it’s an ion which has a positive or negative charge.
Compounds
Ionic compound: one atom borrows electrons from another. These can separate into a positively charged ion and a negatively charged ion in solution (e.g. water). An example is sodium chloride (table salt)
Covalent compound: Atoms share electrons in pairs (a covalent bond). These don’t separate like ionic compound. An example is table sugar.
Acids and Bases
Acid: A substance that gives up H+ ions when dissolved in water.
Corrode metals
Have a sour taste
Not all safe to drink! (lol)
Examples:
vinegar (contains acetic acid)
lemon juice (contains citric acid)
hydrochloric acid
nitric acid
sulfuric acid (Found in lead batteries. I love drinking this one, it goes great with crayon cigars!)
Base: A substance that gives up OH- (hydroxyl) ions when dissolved in water. Also called alkaline.
Bitter taste
Examples:
baking soda (sodium bicarbonate)
liquid soap (contains potassium hydroxide)
lye (sodium hydroxide)
bleach (contains sodium hypochlorite)
When acids and bases react they can neutralize each other, producing water and a salt (not necessarily sodium chloride).
pH Scale
pH is a number from 0 to 14 that shows how acidic or basic something is.
pH < 7 = acidic
pH = 7 = neutral
pH > 7 = basic
+1 in pH = 10x as basic, -1 in pH = 10x as acidic
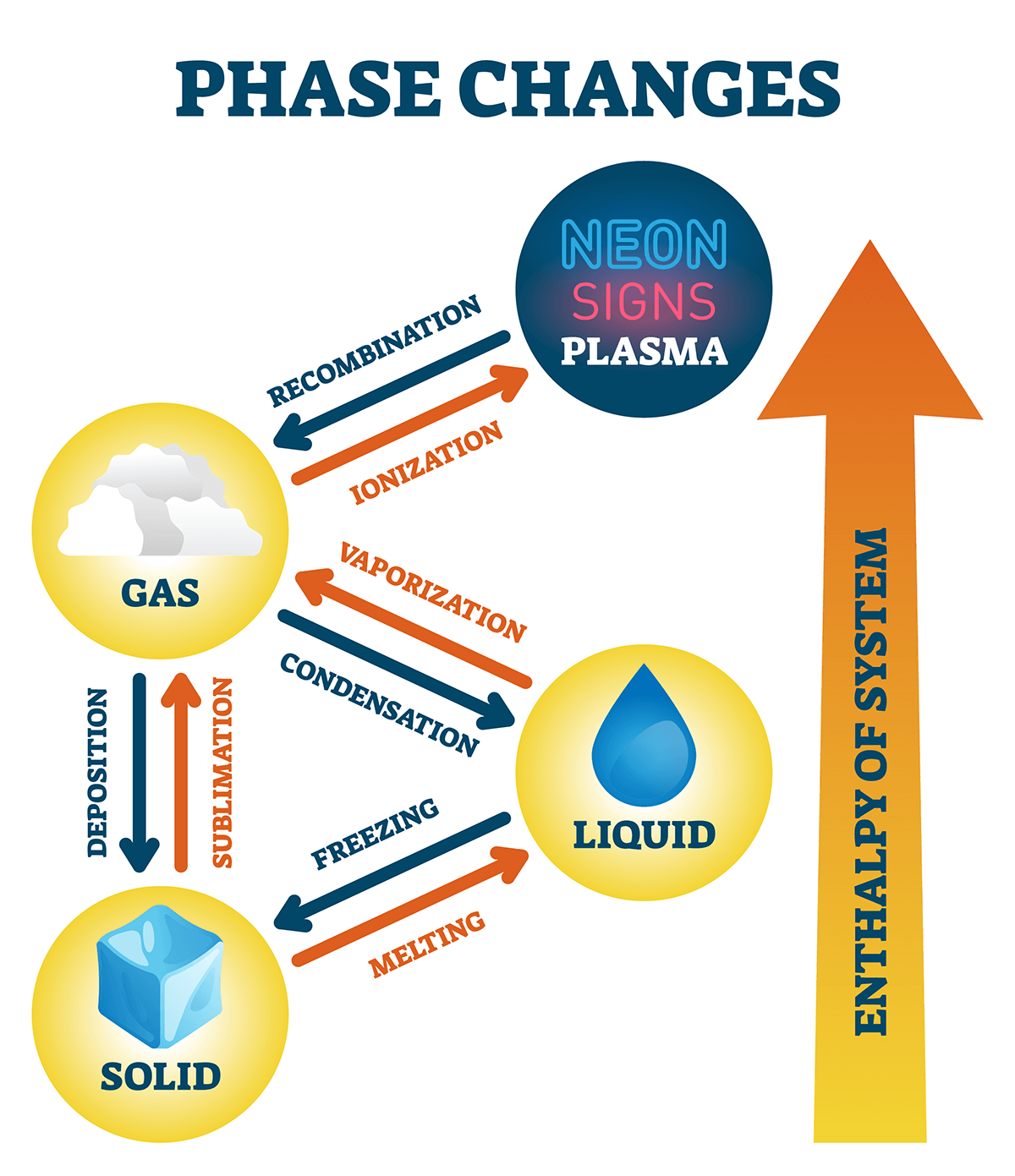
States of Matter and Phase Changes
Solid: constant shape and volume
Liquid: variable shape, constant volume
Gas: variable shape and volume