Chapter 20
Charges and Forces
neutral: no special electrical properties to undisturbed rods; no overall net charge
neutral object contains equal amounts of positive and negative charge
charging: rubbing a rod with wool or silk; the charge is transferred between objects
positively charged: if amount of positive charge is greater then negative charge
negatively charged: if amount of negative charge is greater than positive charge
long range repulsive force: (force requiring no contact) happens when two identical objects are charges in same way
electric force: the force between charged objects
decreases with distance between charged objects
increases with increasing charge of the objects
like charges: repel each other (positive/positive or negative/negative)
opposite charges: exert attractive forces (positive/negative)
if something is attracting the other object it has a positive charge
law of conservation of charge: charges are transferred from one place to another, if positive charge appears then negative charge must also appear elsewhere
discharging: removing charge from a charged object by touching it to another object
conductors: materials through/along which charge easily moves
metal
insulators: materials in which charge remains immobile
glass and plastic
both conductors and insulators can be charged but they differ in ability to move a charge
electrostatic equilibrium: state where excess charges on conductor have redistributed and stopped moving resulting in no net charge motion
the charge movement happens very fast
charge polarization: slight separation of positive and negative charge in neutral object when charged object is brought near
polarization force: attractive force that arises because charges in metal are slightly separated no because the rod and metal are oppositely charged; is always attractive between a charged object and neutral one
Charge Model
frictional forces, rubbing, add or remove charge from object, the process is called charging, vigorous rubbing produces large quantity of charge
two kinds of charges, positive and negative
2 objects of like charge repel & 2 objects of opposite charge attract
called electric forces
force between 2 charged objects is long range force, magnitude of force increases as quantity of charge increases and decreases as distance increases
neutral objects have equal mixture of positive & negative charge
rubbing process charges objects by transferring charge, objects acquire equal but opposite charges
Charge is conserved: cannot be created or destroyed'
two types of materials
conductors through which charge easily moves
insulators in which charges remain fixed
charge can be transferred from one object to another by contact
Charges, Atoms, and Molecules
electron cloud: negatively charged electrons surrounding the nucleus of an atom
charge like mass inherent property of electrons and protons
electrons and protons are the only sources of charge in matter
object is charged if it has unequal number of electrons and protons
electrically neutral: object has no net charge
objects gain positive charge by losing electrons not gaining protons
ionization: process of removing a electron from electron cloud
forces of friction cause molecular bonds to break resulting in molecular ions when bonds are broken
charge symbol q or Q
charge SI unit is coulomb (C)
fundamental charge (e): magnitude of charge of an electron or proton 1.60\cdot10^{-19}C
electrons and protons are neither created nor destroyed in ordinary process so their associated charge is conserved to
insulator electrons are tightly bound to positive nuclei so not free to move around
charging this by friction leaves patches of molecular ions but they are immobile
conductors, in metals, outer atomic electrons (valence electrons) are weakly bound to nuclei, so when atoms come together to form solid, outer electrons become detached from nuclei and roam the solid
solid as a whole remains neutral as no electrons have been added or removed
but their is sea of electrons moving about the positive ion cores
while electrons are mobile within metal they are still weakly bonded to ion cores and won’t leave metal
polarization of one atom is very small effect but their are a lot of atoms in object resulting in a polarization force
electric dipole: 2 equal but opposite charges with separation between them
induced electric dipole: when the polarization is caused by an external charge
permanent electric dipoles: molecules with asymmetry in charge distribution
like water, due to unequal sharing of electrons
hydrogen bond: attractive electric force between a positive H atom bonded to N,F, or O towards the negative N,F,or O of second molecule
a weak bond is formed, results in stickiness, giving water special properties
nucleotides form hydrogen bonds
Coulomb’s Law
Coulomb’s Law: electric force increases for objects with more charge and decreases as charged objects are moved farther apart
F_{1on2}=F_{2on1}=\frac{K\left|q_1\Vert q_2\right|}{r^2}
charge is a magnitude the sign doesn’t matter
K is the electrostatic constant 8.99 × 10^9 N * m²/C²
if multiple charges are present the net electric force is the sum of all the forces due to charge
The Concept of the Electric Field
field model: alteration of space around charge A is the agent that exerts a force on charge B
postulates:
group of charges called source charges, alter space around them by creating electric field
another charge is placed in this electric field it experiences a force exerted by the field
field: alteration of space
electric field: alteration of space around a charge
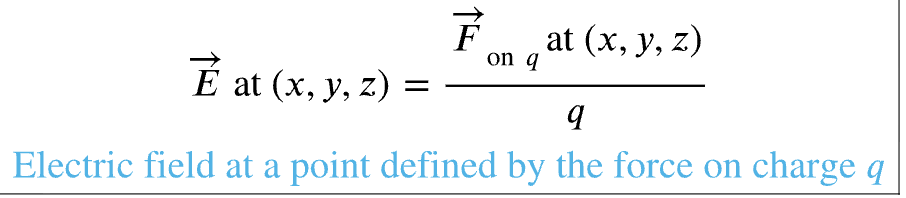
created by charge separation
exist in all points of space around charges
force to charge ratio so units are N/C
if charge is positive electric field vector points in same direction as force on the charge and if negative point opposite of the force
electric field strength: magnitude of electric field

electric field diagram
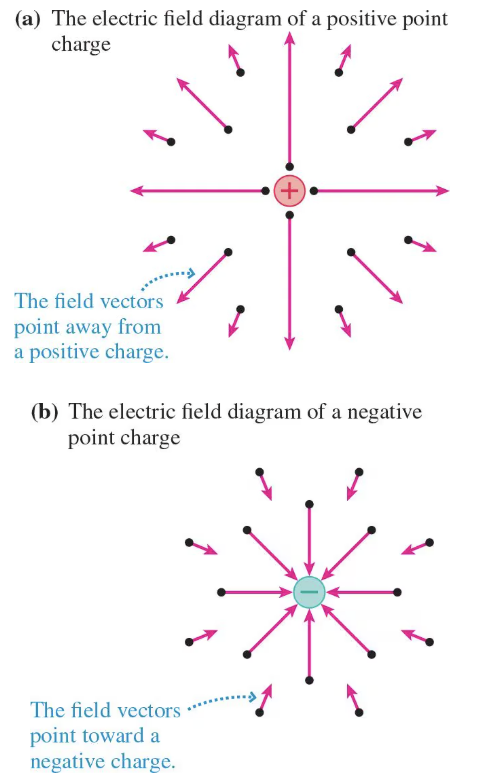
arrows indicate direction and strength of electric field
The Electric Field of Multiple Charges
electric field due to multiple charges is vector sum of electric field due to each of the charges
parallel-plate capacitor: arrangement of 2 electrodes, closely spaces, and charged equally but oppositely
uniform electric field: field inside a parallel-plate capacitor is same in strength and direction
depends on charge to area ratio Q/A (charge density), charges packed closely field is larger
spacing between plates doesn’t affect electric field
shape of electrodes not relevant as long as they are close together
permittivity constant: epsilon naught; = 1/4piK= 8.85 × 10^-12 C²/N x m²
electric field lines: imaginary lines drawn through a region of space so
tangent to a field line at any point is in the direction of electric field
filed lines are closer together where electric field strength is greater
cannot cross
start on positive charge and end on negative charge
Conductors and Electric Fields
electric field is zero at all point inside a conductor in electrostatic equilibrium
so any extra charge is on the surface
electric field right at surface of charged conductor is perpendicular to surface
electric field within a conducting enclosure is zero
the sharper the point the more closely packed charges so the stronger field
Forces and Torques in Electric Fields
place a charge q at a point where the electric field is E then the charge experinces a electric force
q is positive then force on q is in the direction of E
q is negative then force on q is opposite of E
electric force is small for ordinary objects, large for small objects that have been charges, and dominant at atomic scale
electric dipole moment: a vector that points from negative to positive charge of a dipole
in a uniform electric field experiences a torque causing it to rotate