Acid bases and salts
^ PH
* Ph tells us how acidic or basic a substance is. The acidity or basicity of a solution can be described in terms of the hydrogen ion concentration.
[ H+] → concentration of hydrogen is represented in [] - square brackets.
* Acids have Phs lower than 7 and bases higher than
1. [ H+] ( In all acids) is = to the [OH-] (base/alkali) making the solution neutral since none is greater than the other. If the acid is equal to the base you have a neutral solution.
2. The [H+] (acid) is > - greater than the [OH-] (base) making the solution acidic. When the acid is greater than the base the solution is acidic.
3. [H+] (acid) is < - less than the [OH-] (base) making the solution basic. When the base is greater than the acid, or the acid is less than the base the solution is basic.
* [H+] ( The hydrogen ion concentration ) is the acid. [OH-] ( the hydroxide ion concentration) is the base or alkali.
* The litmus paper tells if the solution is acidic or basic.
* Bases and acids react with each other to produce salt.
- Finding the Ph of substances
* The universal indicator ( universal paper) is the test used to find Ph.
1. Universal indicator/paper:
A mixture of acid-base indicators that show changes in colour at different Ph values. It is matched on the Ph scale.

2. Ph meter:
It translates the concentration of H+ ion into an electric signal that is converted into a digital display or deflection on a meter. The Ph meter would give a more accurate reading because it is able to give decimals.

* The universal and litmus paper have indicators which is why they can change colours.
- Indicators
* Liquid indicators exist.
Indicator | Colour in acid | Colour in base |
Litmus | red | blue |
Phenolphthalein | colourless | pink |
Methyl orange | green | yellow |
Bromothymol blue | yellow | blue |
Screened Methyl orange | light red | green |
^ ACIDS
- Properties of acids
Corrosive
Sour taste
Ph less than 7
change blue litmus to red
conduct electricity when in solution (aqueous)
- Definition of acid
* Acids are substances that produce a greater concentration of hydrogen ions than hydroxide ions when dissolved in water or aqueous solutions.
- The states acids exist as
Gases, liquids & solids
* Gases:
CO 2 Carbon dioxide
SO2 Sulpher dioxide
SO3 Sulphite trioxide
NO2 Nitrogen oxide
* Liquids:
HCL
Nitric acid
Sulphuric acid
*Solids:
Citric acid [ from citric fruits eg lime, orange, lemon] C6 H8 O7
Ascorbic acid ( vitamin C) C6 H8 O6
Tartaric acid ( from grapes)
- Types of Acids
Mineral acids
Organic acids
1. Mineral Acids:
* Obtained from minerals
H2SO4 Sulphuric acid
HCL hydrochloric acid
Carbonic acid H2CO3
Nitric Acid HNO3
2. Organic Acids
* Obtain from plants and animals
CH3COOH ethanoic acid
C6 H8 O7 Cirtric acid
C6 H8 O6 Ascorbic Acid
C4H6O6 Tartarc acid
* A dilute solution for ethanoic acid is vinegar. Vinegar is made from apples - apple cider vinegar.
- Concentrated & Dilute
* The concentrated solution contains more moles of solute per volume of solution than dilute ones. To go from a dilute to a concentrated solution you heat it.
{ BASICITY in acid}
* This is the number of H+ ions produced per molecule of acid when the acid dissolves in water.
* A hydrogen ion can also be called a proton because after ionization only one proton is left.
- The Three Types
Monobasic or Monoprotic
Dibasic or Diprotic
Tribasic or Tri protic
* Examples
Monobasic or Monoprotic : Hydrochloric acid (HCL), Nitric acid ( HNO3 ), Acetic acid
Dibasic or Diprotic: Sulphuric acid ( H2SO4), Carbonic acid ( H2SO3)
Tribasic or Tri protic: Phosphoric acid ( H3PO4)
- monobasic
* These produce 1 Hydrogen H+ per molecule when in solution.
- Dibasic
* Produces 2 Hydrogen H+ molecules when in solution
Tribasic
* Produce 3 Hydrogen H+ molecules when in solution.
* Monobasic acids can only form 1 salt which is normal salt. Dibasic acid can form 2 salts. Tribasic acid can from 3 salts.
- Strong & Weak Acids
* Weak Acids:
A weak acid is only partially ionized when dissolved in water.
* Strong Acids:
A strong acid is fully ionized (dissociated) when dissolved in water.
Examples of Strong Acid:
HCl - hydrochloric acid.
HNO3 - nitric acid.
H2SO4 - sulfuric acid.
Examples of Weak Acids:
Formic acid HCOOH
Acetic acid CH3COOH
Benzoic acid C6H5COOH
Oxalic acid C2H2O4
Hydrofluoric acid HF
Nitrous acid HNO2
Sulfurous acid H2SO3
Phosphoric acid (chemical formula: H3PO4)
- Reactions of Acids
1. Acids & Carbonates
Acid + Carbonate → salt + water + carbon dioxide
* In reaction A, all of the sodium carbonate will dissolve once enough H2SO4 is added to it, however, in creation B all of the calcium carbonate will not dissolve because a layer of calcium sulphate will form around the calcium carbonate preventing more H2SO4 from reaching the calcium carbonate hence stopping the reaction.
2. Acid & Metal
Acid + metal → salt + Hydrogen gas
* All metals except Cu, Ag and Au will displace the H+ from the acid.
Reactivity series:

3. Acid & Metal Oxide
* Acid + Metal oxide → salt + water

4. Acid & Alkali
* Acid + Alkali → salt + water
* All alkalis are soluble except CaH2
^ BASES
* Bases are proton acceptors.
* It is an oxide or hydroxide of metal.
- Examples of Bases

* Alkalis are soluble bases.
Examples: KOH & NaOH (NH4OH is weak)
- Properties of bases
Soapy slippery feel
Bitter
Ph greater than 7
Change red litmus paper to blue
Conduct electricity when dissolved in water
corrosive
- Strong & Weak Alkalis
* Strong Alkalis are fully ionized in water. Weak Alkalis are not fully ionized in water.

* Once there is OH- it means it's basic or Alkali but it does not make it strong or weak. The
majority don't dissolve so they're weak.
Examples of strong bases:
Potassium hydroxide KOH
Sodium hydroxide NaOH
Barium hydroxide Ba(OH)2
Strontium hydroxide Sr(OH)2
Calcium hydroxide Ca(OH)2
Lithium hydroxide LiOH
Rubidium hydroxide RbOH
Examples of weak bases:
Al(OH)3 aluminium hydroxide.
Pb(OH)2 lead hydroxide.
Fe(OH)3 iron hydroxide.
Cu(OH)2 copper hydroxide.
Zn(OH)2 zinc hydroxide.
NH3 Ammonium
- Amphoteric Oxides
* They can react with both acids and strong Alkalis to form salt and water.
amphoteric oxide or hydroxide + acid → salt + water
amphoteric oxide or hydroxide + strong alkali → salt + water
* Alkalis can react with other alkalis
Examples of Amphoteric oxides:




- Acidic Oxide
* Acidic oxide reacts with water and produces an acid, it is the oxide of non-metals.
Acidic oxide + water → acid
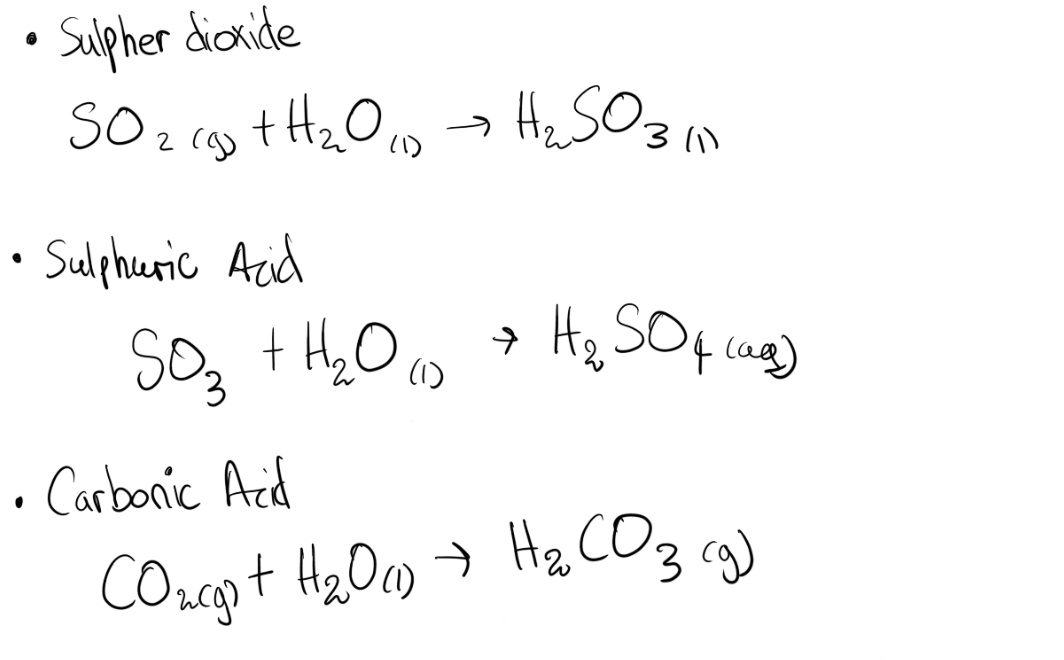
- Neutral Oxide
* Neutral oxide show neither basic nor acidic properties and they do not form salts when reacted with acids or bases.
Examples:
Carbon Monoxide CO
Nitrus Oxide N2O
Nitric Oxide NO
^ SALTS
What is a salt?
* A salt is a compound formed when some or all of the hydrogen ions in an acid are replaced by metal ammonium ions.

* Ionic compounds is another name for salts.
* Zinc hydroxide, and sulphuric acid have positive & negative charges but are not salts. Has to be a metal or ammonium ions.
- Normal Salts
* Normal salts are formed when all of the H+ ions in an acid are replaced by metal or ammonium ions.
Monobasic acids form normal salts.
Examples:
KCl Potassium chloride
NaCl Sodium chloride
FeS04 Iron sulphate
Na2S04 Sodium sulphate
FeCl2 Iron chloride
- Acid Salts
* Acid salts are formed when H+ ions in an acid are only partially replaced by a metal or ammonium ions.
Examples:
Ammonium Chloride NH4Cl
Ammonium Sulphate (NH4)2SO4
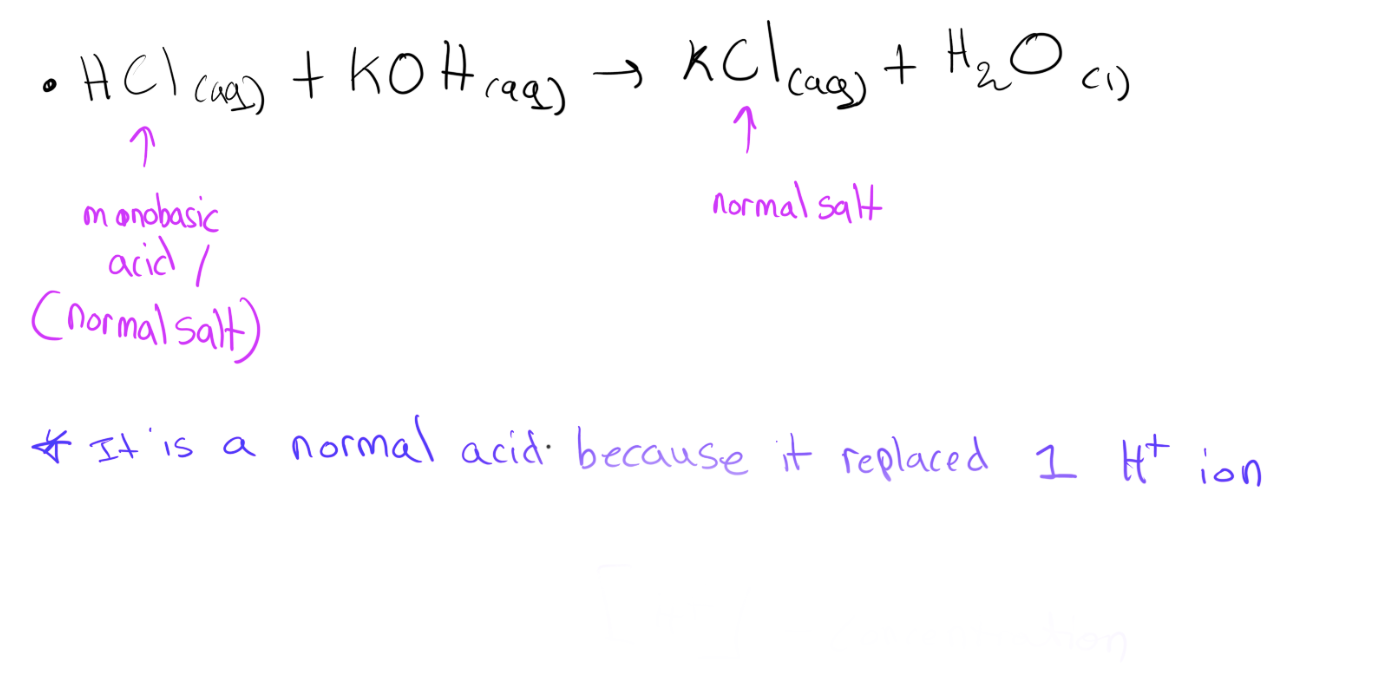
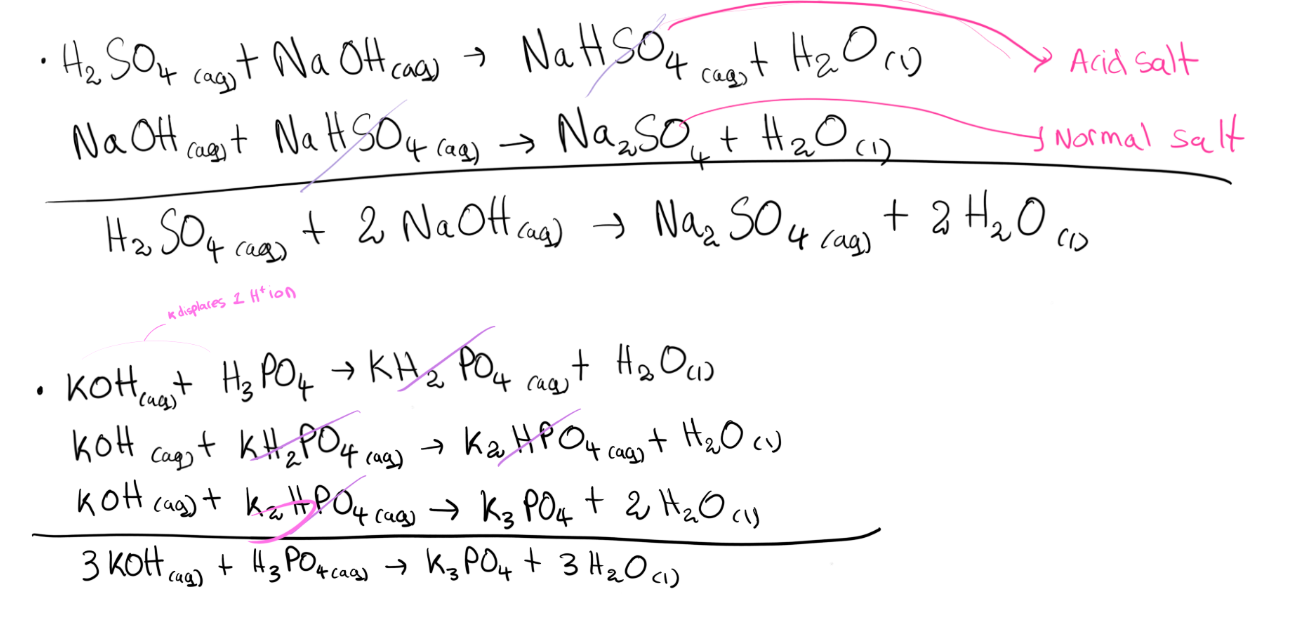
- Preparation of Salts
* When preparing salts you have to determine if the salt is soluble or insoluble.
Preparation of Soluble Salts
A) Acid & Metal carbonate
Acid + metal carbonate → salt + water + carbon dioxide
Measure 15cm3 of an acid and pour it into a beaker
Add excess metal carbonate to the acid
[ This is to use up all of the acids to replace all of theH+ ions. Forms normal salts. You know when to stop adding when it stops dissolving or effervescence stops. ]
Filter the suspension
Collect the filter & discard the residue
Evapourate the filtrate/solution to about 1/2 the volume
[ This is to make the solution more concentrated by removing water.
Leave or allow to cool
[ So crystallization can take place. ]
Filter to collect crystals
Wash crystals with small amounts of cold distilled water & leave crystals to dry.
B) Acid + Metal
Acid + metal → salt + hydrogen gas
Measure 15cm3 of an acid and pour it into a beaker
Add excess metal to the acid
[ This is to use up all of the acids to replace all of theH+ ions. Forms normal salts. You know when to stop adding when it stops dissolving or effervescence stops. ]
Filter the suspension
Collect the filter & discard the residue
Evapourate the filtrate/solution to about 1/2 the volume
[ This is to make the solution more concentrated by removing water.
Leave or allow to cool
[ So crystallization can take place. ]
Filter to collect crystals
Wash crystals with small amounts of cold distilled water & leave crystals to dry.
C) Acid + Metal Oxide
Acid + Metal Oxide → salt + water
Measure 15cm3 of an acid and pour it into a beaker
Add excess metal oxide to the acid
[ This is to use up all of the acids to replace all of theH+ ions. Forms normal salts. You know when to stop adding when it stops dissolving or effervescence stops. ]
Filter the suspension
Collect the filter & discard the residue
Evapourate the filtrate/solution to about 1/2 the volume
[ This is to make the solution more concentrated by removing water.
Leave or allow to cool
[ So crystallization can take place. ]
Filter to collect crystals
Wash crystals with small amounts of cold distilled water & leave crystals to dry.
D) Acid + Alkali
Acid + Alkali → salt + water
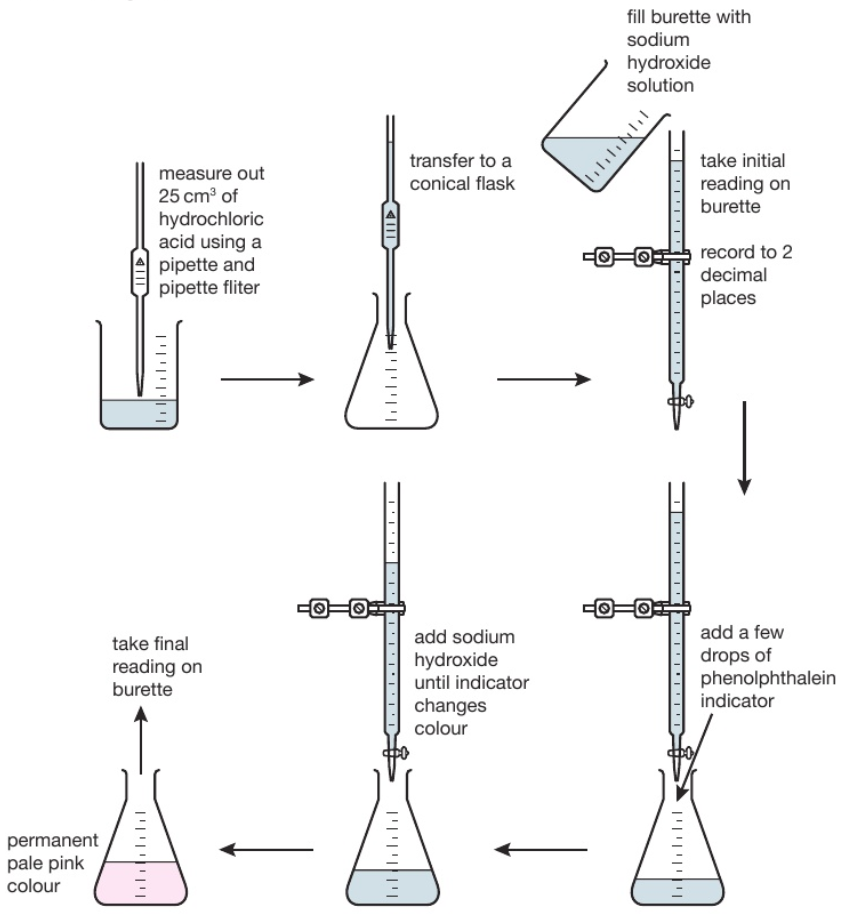
Measure 15cm3 of Alkali and use a pipette to add the alkali to a conical flask
Add a few drops of indicator to the alkali
Fill a burette with acid and record the starting volume
Slowly add the acid from the burette to the alkali in the conical flask
Stop adding the acid when the indicator changes the colour of the solution permanently
Record the volume of acid
fill up a burette with acid
Pour alkali into a conical flask
Slowly add the acid to the alkali until the end point volume is reached
filter the suspension
Collect the filter & discard the residue
Evapourate the filtrate/solution to about 1/2 the volume [ This is to make the solution more concentrated by removing water.
Leave or allow to cool [ So crystallization can take place. ]
Filter to collect crystals
Wash crystals with small amounts of cold distilled water & leave crystals to dry.
* You can't have the indicator in the salt so you make over the salt without the indicator. The acid & alkali are colourless solutions so you need the indicator to see a noticeable pH change occurring near the end point of acid-base titrations, an indicator can be used to signal the end of a titration.





