Biochemistry
Biochemistry -branch of science that deals with the chemical processes related to living organisms
Matter
Pure substance
Element - Basic building block of all matter. Cannot be broken down into simpler substance. Combination of atoms forms element. E.g. letters in the alphabet; combination of the letter (atoms) in the alphabet creates word (element)
Compound - Combination of two or more elements that can be broken down into simpler substances by means of chemical reaction. E.g. combination of words (elements) that forms sentences (compound)
Organic compound - with carbon atom (carbohydrates, lipids, proteins, nucleic acid)
Inorganic compound - without carbon atom (water, acid, bases and salt)
Mixture - combination of 2 or more pure substances
Homogeneous mixture - uniform, consistent, equally distributed.
Heterogeneous mixture - non-uniform, inconsistent and not equally distributed.
MACROMOLECULE
Large molecules made up of smaller elements connected to one another.
Carbohydrates - CH2O. Main source of energy for living organisms. The most abundant macromolecule on earth because this is a product of photosynthesis. Can be classified into:
Monosaccharides - single sugar unit. Building block of sugar. Common forms are:
Glucose - dextrose blood or blood glucose. Primary sugar found in the bloodstream and acts as the source of energy for the blood cells.
Fructose - corn sugar. Sugar found in fruits. 10x sweeter than lactose.
Galactose - component of milk sugar, lactose.
Disaccharides - C12H22O11. Two monosaccaride unit bond together. Most common forms are:
Sucrose - Table sugar. Sugar that sweetens our foods and drinks. Combination of glucose and fructose.
Lactose - Milk sugar. Combination of glucose and galactose.
Maltose - Raw material for beer production. Combination of two glucose.
Hydrolysis - process of breakdown of disaccharide into monosaccaride units to be utilized by living things.
Polysaccharides - formed from the branching of 3 or more monosaccaride.
Starch - storage carbohydrates for plants made with several glucose units.
Glycogen - “animal starch” storage of carbohydrates in animal's liver, therefore preventing them from eating every minute.
Cellulose - found in plant cell wall giving them protection and strength and also making them hard to digest. Provides rich supply of glucose if digested.
Chitin - found in mushrooms and insects exoskeleton inflexible than cellulose and use for support and does not swell in water.
Protein
Composed of carbon, hydrogen, oxygen and nitrogen and sulfur and phosphorus may also be present.
Most abundant organic compound in living things because most of our body like muscle tissues, hair, collagen are made up of protein.
Essential to the structure and function of living cells because it serves as the building blocks for cell membrane, organelles and many more.
Amino acid - building block of protein. Living organisms uses 20 different amino acid to make proteins.
Amino acids
Amino acids
Three letter code
One letter code
Alanine
Ala
A
Arginine
Arg
R
Aspartic acid
Asp
D
Asparagine
Asn
N
Cysteine
Cys
C
Glutamic acid
Glu
E
Glutamine
Gln
Q
Glycine
Gly
G
Histidine
His
H
Isoleucine
Ile
I
Leucine
Leu
L
Lysine
Lys
K
Methionine
Met
M
Phenylalanine
Phe
F
Proline
Pro
P
Serine
Ser
S
Threonine
Thr
T
Tryptophan
Trp
W
Tyrosine
Tyr
Y
Valine
Val
V
Essential for building and repairing body cells - proteins makes up most tissues in our body like muscle and organ tissues. By synthesizing proteins from protein synthesis of RNA, the newly synthesized proteins contribute to the repair of the damage. Synthesizing synthesized of synthesis
Aid in speeding up chemical reactions in the body - protein serves as enzymes and enzymes act as catalyst facilitating and lowering the energy needed for chemical reaction to occur.
It brings individual differences - protein contribute to unique characteristics of individual.
It serves as protection in form of antibodies - by serving as antibodies, proteins recognize and neutralize harmful substances such as pathogens.
Functions | Example | Explanation |
|---|---|---|
Movement | Myosin and actin - the muscle proteins responsible for contraction of muscles | Muscle contraction rely on the interaction of proteins |
Structure | Collagen - most abundant protein in the human body. It forms bones, skin, tendons and cartilage. | These type of proteins (collagen and keratine) makes up the framework of various body components. |
Regulation | Enzyme - acts as catalysts that regulate the speed of chemical reactions | It can transmit signals within cells to regulate functions such as metabolism, growth and response to external stimuli |
Transport | Hemoglobin - the iron-protein compound in RBC. It carries oxygen through bloodstreams. | Oxygen can bind with the hemoglobin inside the lungs. Heart then pumps the oxygenated blood (oxyhemoglobin) to parts of the body that needed oxygen. |
Nutrition | Casein - constitutes about 80% of proteins in cows milk | |
Defense | Antibodies - “Immunoglobulin”, a defense agaisnt foreign substances | Immunoglobulin produced by b cells, is a response to specific pathogens. It recognizes self to non-self and neutralize harmful threats. |
Lipids
Has a chemical composition of Carbon, Hydrogen and lesser oxygen than carbohydrates.
The larger hydrogen gives more energy per molecule than carbohydrates.
Can store fats providing a long term energy source
Highest energy-giving foods
Include fats that are insoluble in water but soluble in some solvent like chloroform
Acts as insulators for animals against extreme cold - skin-fats-lean muscle. Fats can act as a 'barrier' protecting the body from stimuli such as temperature.
Forms of lipids
Fatty acids
Saturated fats - solid in room temperature such as margarine and animal fats.
Unsaturated fats liquid in room temperature such as vegetable oils and corn oils
Phosphoglycerides - also known as Phospholipid that makes up the cell membrane of cell. It gives protection, shape and regulation of materials from extracellular and intracellular.
Lecitin - egg yolk, yeast and live which breaks down fats into smaller molecules
Cephalin - brain tissues
Sphingolipid - found in brain, nerves and lungs. It serves as lubricant or surfactant of the lungs for fetus. In the eight month of pregnancy sphingomyelin runs out.
Wax - used for ointments, polishers and lubricant. Serves as protectant in leaves for water balancing and pest control. Waxes acts as hydrophobic layer in the leaf surface reducing the evaporation rate and making it hard for pest to destroy.
Terpenes - insoluble in water but soluble in organic solvent such as alcohol. The aroma in plants is due to terpenes such as: chlorophyll, carotene and mint oil.
Steroid - examples are cholesterol and sex hormones (estrogen and androgen).
High density lipoprotein are cholesterols that are said to be good because it helps clean up cholesterol in the bloodstream.
Low density lipoprotein are said to be bad because it lead to accumulation of cholesterol in the bloodstream.
Ways to remove cholesterol in the bloodstream is to exercises so that your body will use stored fats in the body as well as cholesterol in the bloodstream. Eat plants and fruits because they have cellulose that are polysaccharide therefore hard to breakdown and serves as the cleaner of bloodstreams.
Nucleic acid
Largest complex organic molecule composed of carbon, hydrogen, oxygen, nitrogen and phosphorus.
Contains heredity and genetic information.
Three basic parts are pentose carbon sugar, phosphate, and nitrogenous base.
The nitrogenous bases include:
A. Purines = adenine - guanine
B. Pyrimidine = cytosine - thymine
Deoxyribose nucleic acid - found only in the nucleus of the cell. Carries the instruction and control the activities of the cell.
Ribose nucleic acid - found inside and outside the nucleus(cytoplasm). Uses instructions from the DNA to make proteins.
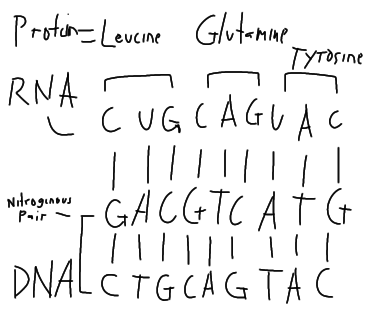
From nitrogenous bases of DNA (Adenine- thymine and cytosine-guanine) if forms pairs. The DNA send a signal and an enzyme called RNA Polymerase, reads the DNA and synthesize RNA from it. Nitrogenous base of RNA are Adenine-uracil and cytosine-guanine. This RNA carries genetic information from the DNA. RNA is then use as the base for protein synthesis in the ribosome. That's why protein contribute to our unique characteristics.
James Watson and Francis Crick describe the DNA as double helix. Consists of two hydrogen bond.
RNA - single strand found in nucleus and cytoplasm. (AUGC)
Importance: Nucleic acid DNA serves as genetic material for living organisms. DNA and RNA are responsible for protein synthesis