Yr 2 - Physical
Summary and key points from CH17 (Thermodynamics): :))))
The definitions:
Standard enthalpy of formation: Is the enthalpy change when one mole of compound is formed from its constituent elements under standard conditions, with all reactants and products in their standard states.
Standard enthalpy of combustion is the enthalpy change when one mole of compound is burnt completely in oxygen (undergoing complete combustion)
Standard enthalpy of atomisation: the enthalpy change when one mole of gaseous atoms are formed from element in its standard states under standard conditions.
Standard enthalpy of ionisation energy: The amount of energy required to change one mole of atoms in gaseous state to one mole of gaseous ions (each with a single + charge)
Standard enthalpy of electron affinity: The enthalpy change when one mole of gaseous atoms each become one mole of gaseous ions (each with a single negative charge)
First electron affinity is always negative
Second electron affinity is always positive (endothermic reaction, energy is put in)
Why is this the case? The electron is attracted by the positive ion so energy is put in to overcome this attraction.
Lattice enthalpy of formation (depends on charge of ion + the size of ion (stonger if both are smaller - for this don’t include covalent characteristic)): The standard enthalpy change when one mole of solid ionic compound is formed from its gaseous ions
Lattice enthalpy of dissociation: The standard enthalpy change when one mole of solid ionic compound dissociated into its gaseous ions
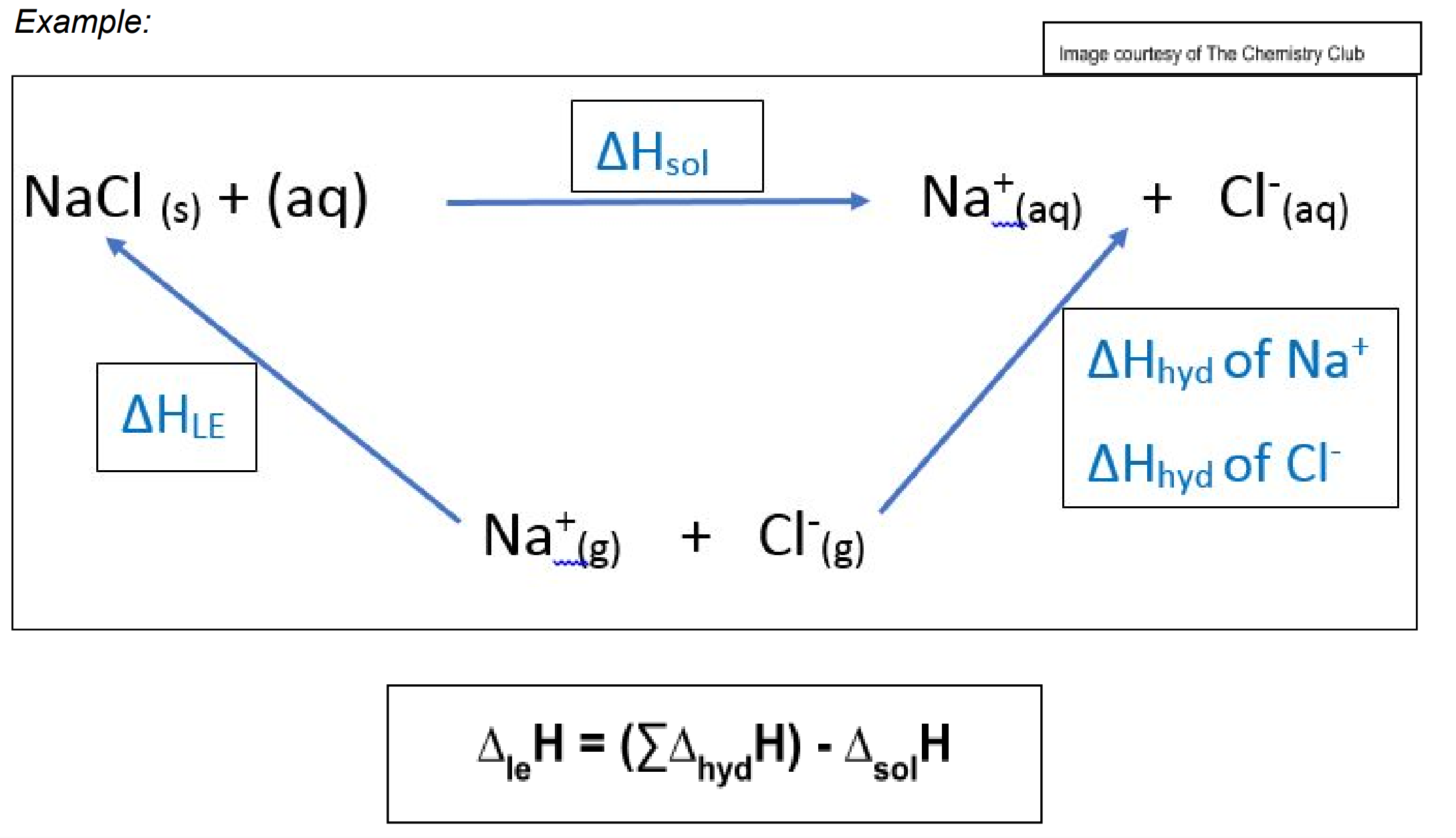
Enthalpy of hydration: The standard enthalpy change when water molecules surround one mole of gaseous ions form aqueous ions.
Enthalpy of solution: The enthalpy change when one mole of solute dissolves completely in sufficient solvent where ions are far apart not to interact with each other.
Mean bond enthalpy: When one mole of gaseous molecules each break a covalent bond to form two free radicals averaged over a range of compounds.
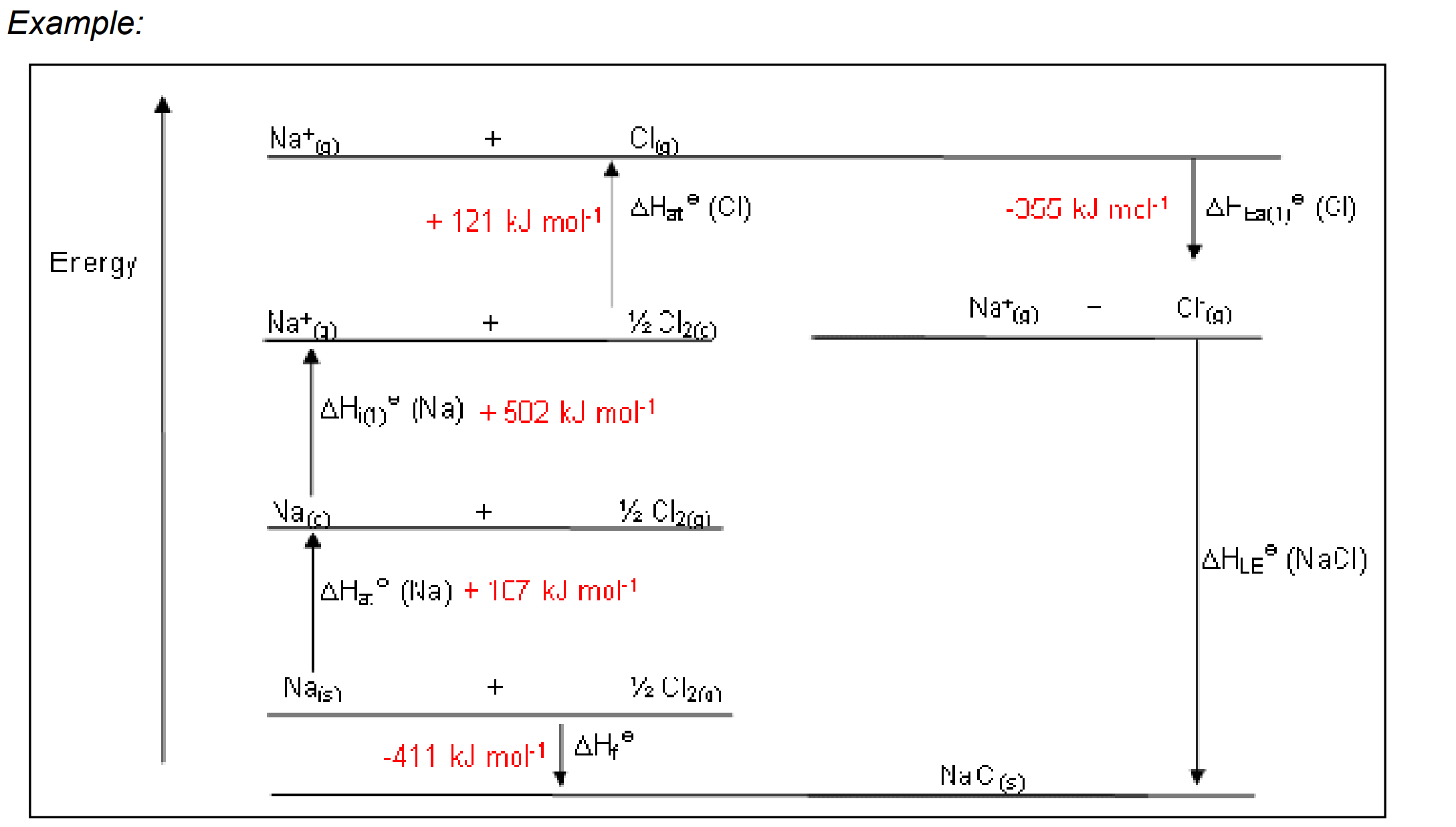
Born Haber cycle: A thermochemical cycle that includes all the enthalpy changes involved of the reaction.
Trends in lattice Enthalpies:
Larger ions = Smaller enthalpy, opposite charges do not approach each other when ions are larger.
Increased charge = larger enthalpy, more electrostatic forces of attraction
Factors that effect polarisation (increased covalent characteristics)
Positive ion - small + high charge (increases)
Negative ion - large + high charge (increases)
Example of Hess cycle and Born Haber:

Chemical reactions take place (feasible + spontaneous):
Entropy: A numerical measure of amount of disorder in a system
To calculate entropy (The product entropy - the reactant entropy)
Units: kJ/K mol
If entropy (+), likely to be spontaneous.
Gibbs free energy: combines the entropy and enthalpy (if negative reaction is feasible)

Key points:
When using delta S, make sure to divide by 1000.
The equation can be used as a straight line graph, where m = delta S, T = x, C = delta H and y = delta G
Summary and key points for CH18 (kinetics Y2):
Rate of reaction: the change in concentration (products or reactants) over time
The rate constant (is affected by temperature and presence of catalyst) [affected by catalyst, because catalyst lowers the activation energy, lowering the activation energy increases the value of k]
rate = k[X][Y]2
Where X is 1st order
Where Y is 2nd order
The units for k depends
Rate has units mol/dm cubed s
Overall order is 1+2 =3
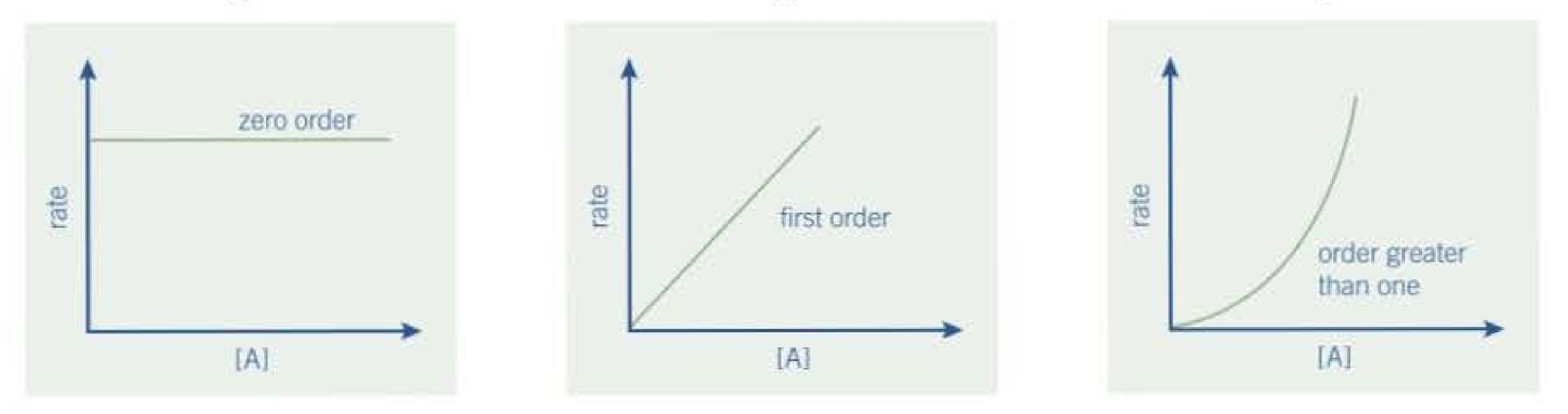
Rate-concentration graphs:
Concentration-time graph:
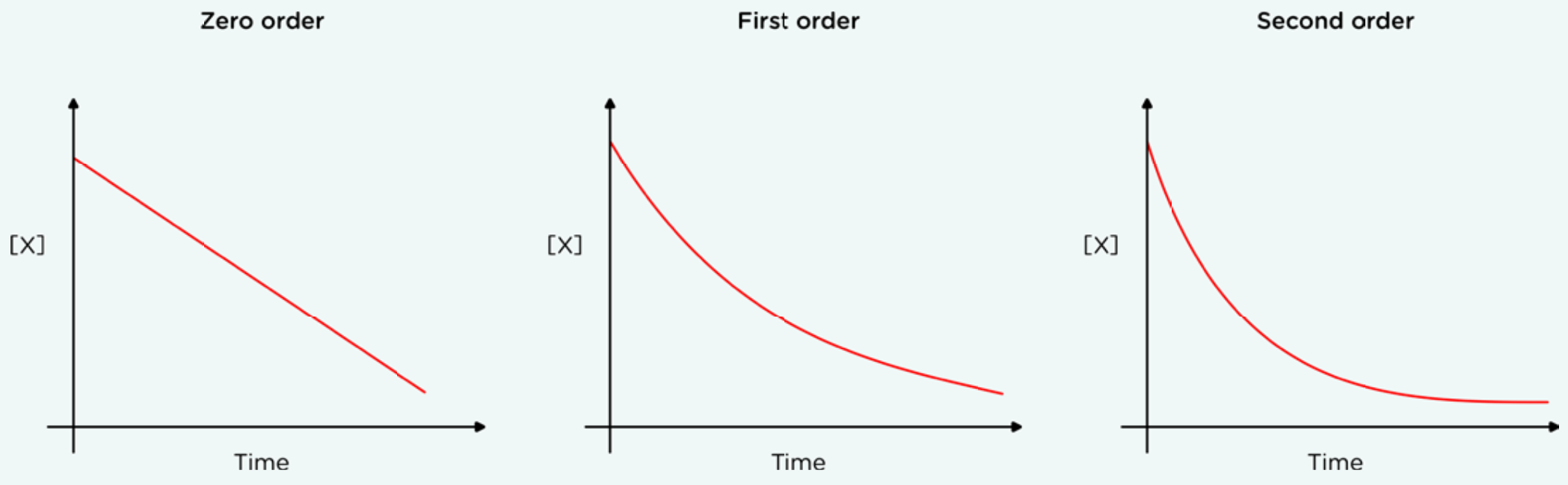
By finding the inital rate (when time =0), then the concentrations of all the reactants are known.
Arrhenius equation:
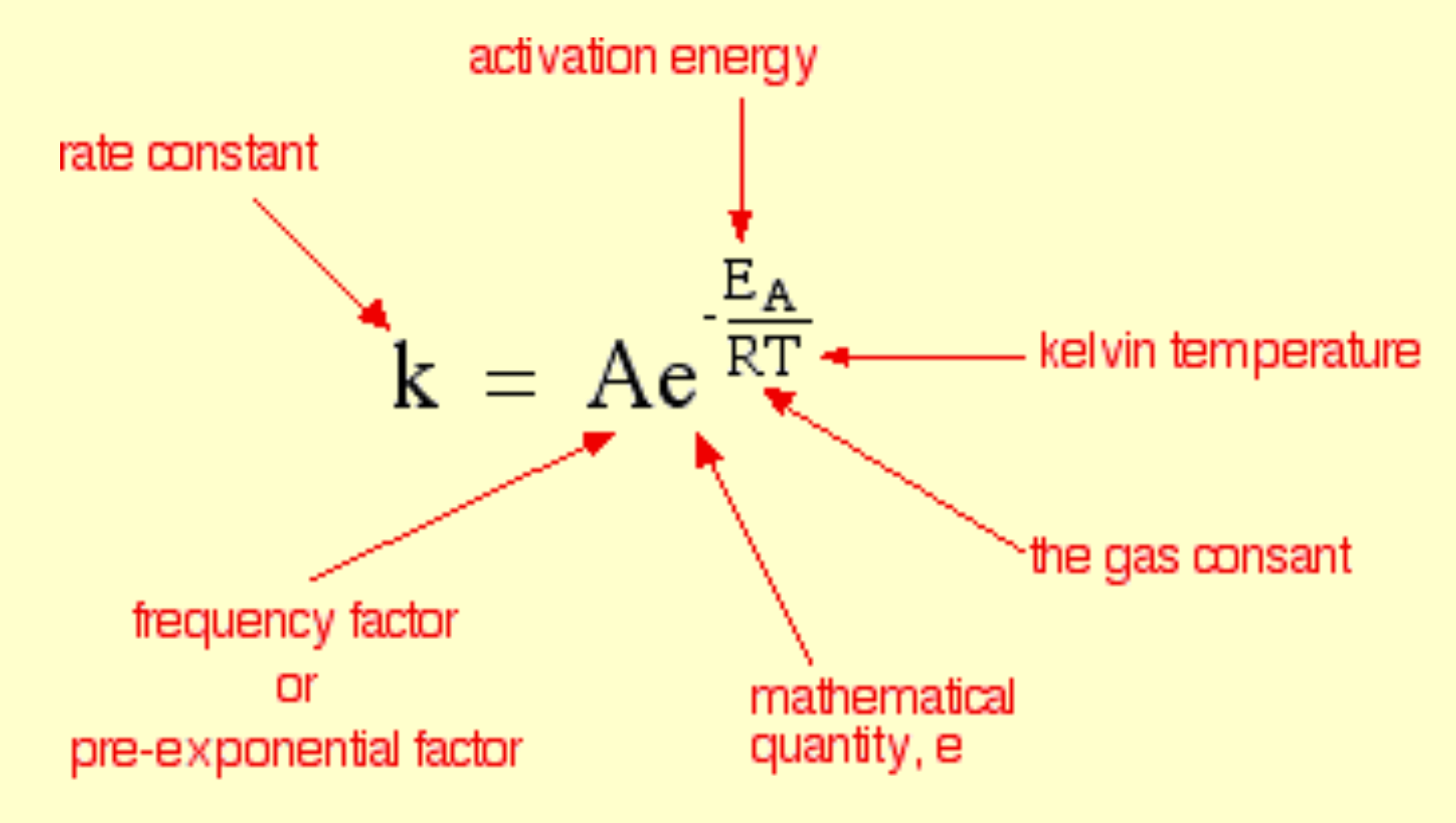
T = Temperature (kelvin)
R = gas constant
Ea= The activation energy (J/mol)
e = the exponential
e-(Ea/RT) = The fraction of molecules present in gas that have energies equal to or excess of activation energy at particular time.
With higher temperatures there will be greater number of particles with molecules having greater than activation energy.
A = taking into account frequency of collisions and their orientation
We can convert the Arrhenius equation by taking logs to both sides:
ln K = -Ea/RT + ln A
ln K = (-Ea/R X 1/T) + ln A
Where:
lnK = y
-Ea/R = m (gradient)
To find activation energy multiply by 8.31
Divide the value by 1000 (and multiply by -1)
To find activation energy measured in kJ/mol
1/T = x
ln A = C
Y axis intercept
Rate determining step (rate limiting step) => the slow step
Summary and key points of CH19 (Equilibrium constant Kp):
Partial pressure = Total pressure (kPa) X mole fraction
Units are in Pascals, kPa or atm (depends on the question)
It is the partial pressure of products/ partial pressure of reactants
CURLY BRACKETS THIS DOES NOT INCLUDE CONCENTRATION
Factors effecting Kp

Summary and Key points CH20 (Electrode potentials and electrochemical cells)
Electrochemical cell: contains two half cells, connected with a wire, high resistance voltmeter and a salt bridge.
Salt bridge: Filter paper dipped in concentrated KNO3, allowing ions to flow through (completing the circuit), should be emerged in the solution not touching the electrodes.
Standard Hydrogen Electrode:
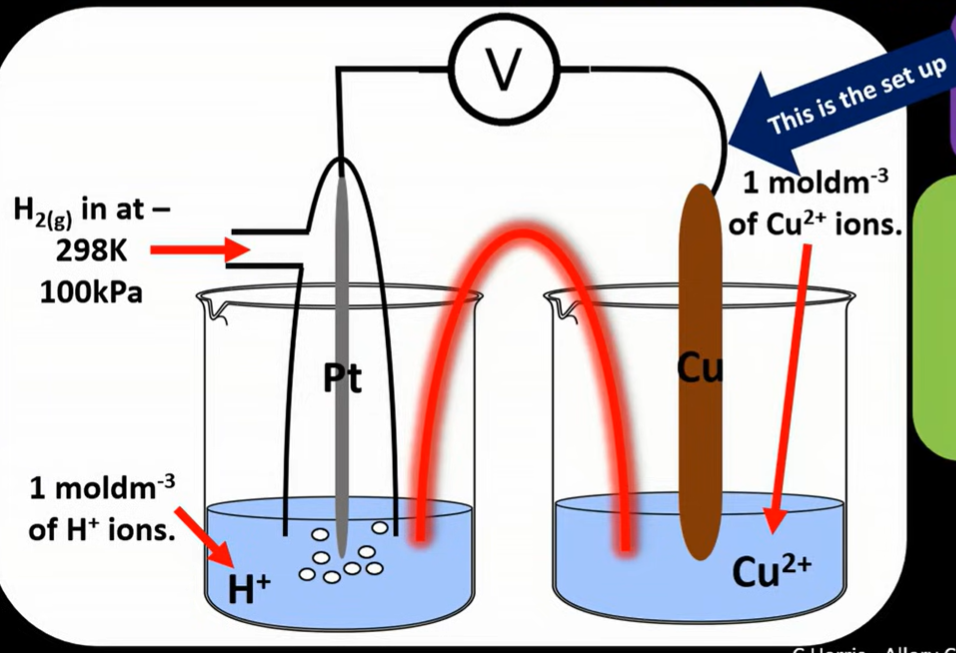
Standard conditions: 298K, 100kPa, 1 mol/dm cubed, allowing the EMF value to be calculated.
CAREFUL: when 1 mol/ dm cubed of H+ ions, this means 1 mol of HCl OR 0.5 H2SO4 (careful if dichrotic acid, dissociating 2 H+ for every sulfuric acid molecule)
Calculating EMF (Right (reduced) electrode - Left electrode)

Cell notation is a simpler way to represent the set up of a cell.

To find if the reaction between two half cells will be feasible the EMF should be positive.
Batteries:
Non-rechargeable:
Two half cells that provide a current when connected to an external circuit, when the chemicals are used up the cell is flat.
Uses: small devices (doorbells, remote controls and torches)
adv: cheap // disadv: Casing eventually dissolves and leaks acidic ammonium chloride paste
Rechargeable:
During recharging apply an external voltage greater than the cell voltage to reverse cell reactions
Uses: Laptops, smart phones and other mobile devices
Adv: can recharge without having to run down // disadvantage: costly, sensitive to high temperatures, may explode on heating
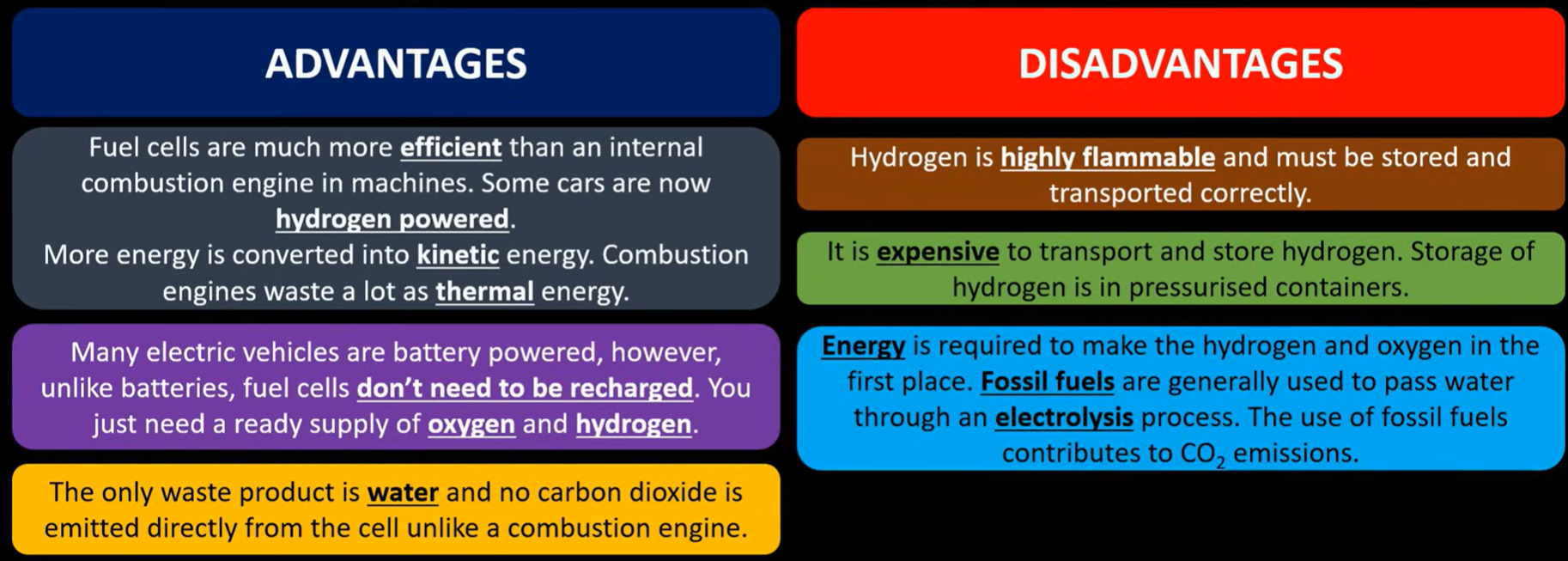
Fuel:

Summary and Key points CH21 (Acids, bases and buffers)
Bronsted Lowry definition
Acid: A substance that can donate proton
Base: A substance that accepts a proton
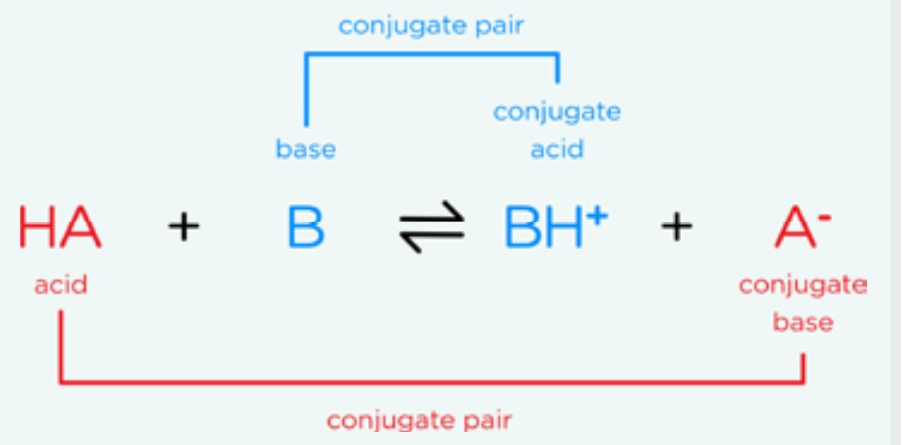
Key point: when H+ dissociates in water, it is almost never found alone, it is mostly bonded with water H3O+ (oxonium ion)
Ionic product of water:
Kw = [OH-] [H+] = 10-14 (at 298K)
In pure water [OH-] = [H+]
The pH scale (calculating pH of solution)
pH = -log10[H+]
Acid dissociation constant Ka:
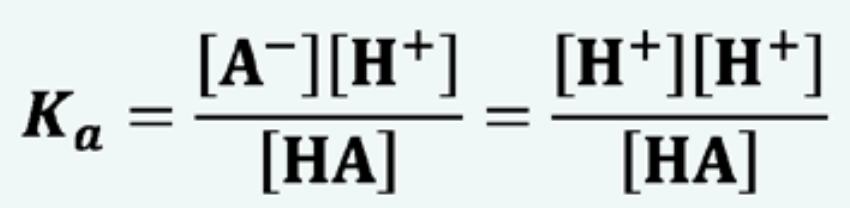
This is because, we assume the concentration of [HA] remains roughly the same as only partial dissociation of the acid. Also assume the concentration of A = H, because they HA dissociates in a ratio of one, with each H there is a A molecule.
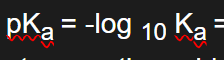
pKa = -log 10 Ka => can be used to measure how strong the acid is, the smaller the pKa the stronger the acid
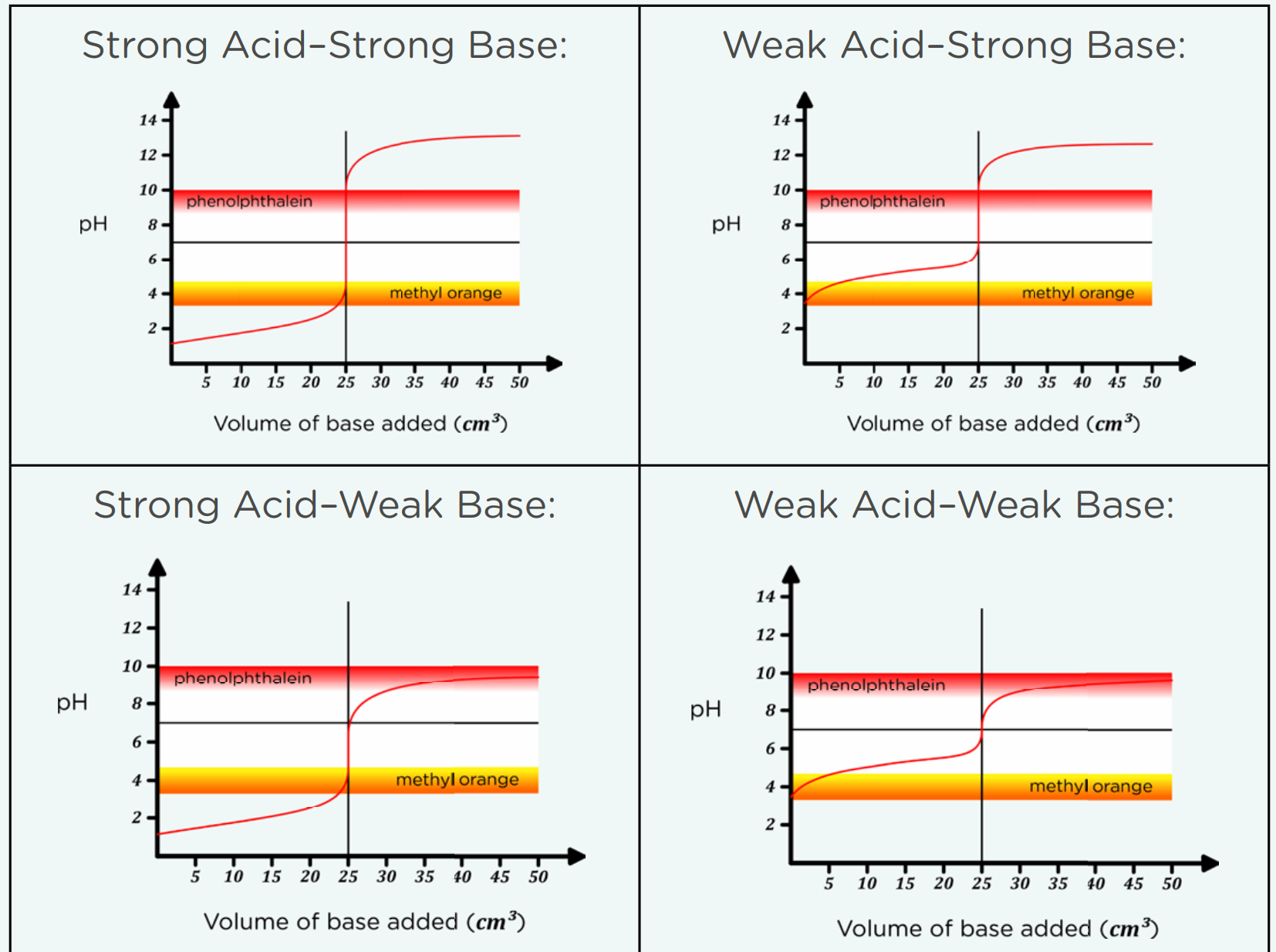
Colour change occurs at equivalent point:
Phenolthalein: colourless (acid) —> pink (alkaline)
Methyl Orange: Red (acid) —> Yellow (alkaline)
Key definitions:
Equvalence point: When equal amount of [OH-] = [H+]
End-point: When the amount of acid or alkaine added, causes the indicator to change colour
Acidic buffers: Weak acid + Soluble salt of acid conjugate
Alkaline buffers: Weak acid (partially dissociated) + strong alkaline
Buffers: Resist changes in pH, when small amounts of acid or alkaline are added
HA H + A
When acid is added, more H ions so shift to the left to reduce the H ions added, so pH remains fairly constant
When Alkaline added, H ions are used up, so shift to the right, so pH remains fairly constant
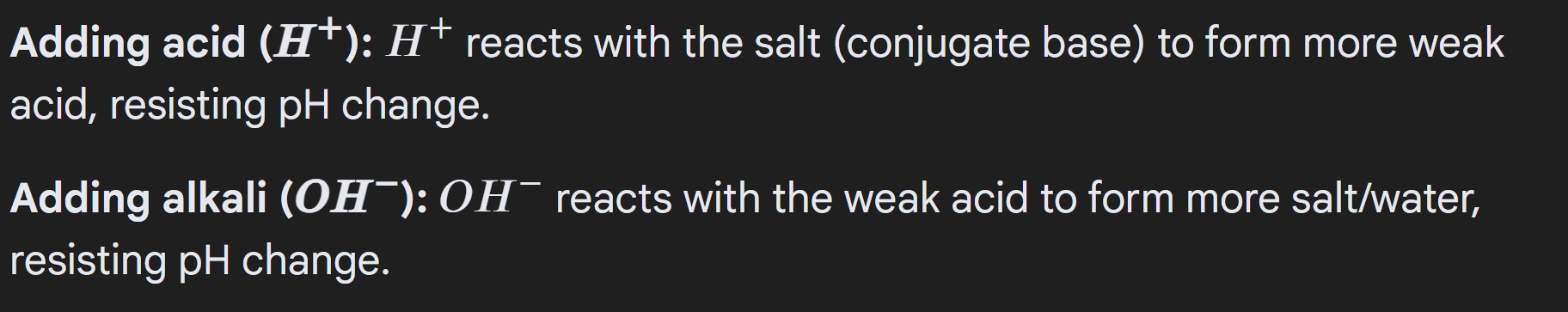
Adding acid, reacts with the salt => so decreases the moles of salt (everything else same) (check tmr if the moles of acid increases?)
Adding alkaline, reacts with the acid => so decreases the moles of acid (everything else is the same)
Everything about Electrochemistry
Electrical potential can not be measured directly, potential difference is measured instead
The electrical circuit: 2 electrodes (in their solutions), high resistance voltmeter, salt bridge (filter paper dipped in saturated KNO3) => piece of paper is used instead of wire to avoid further metal/ion into the solution
What is a salt bridge (common question)? Allows ions to move through, to complete the circuit
The negative electrode the (Most positive EO) better reducing agent so oxidises and looses electrons more easily, and by convention electrons flow from negative electrode to positive electrode. The negative electrode is to the left.
The acronym: NO PRoblem (negative oxidised and posititive reduced)
The right is more positive electrode, the species being reduced and gains electrons
Standard hydrogen electrode:
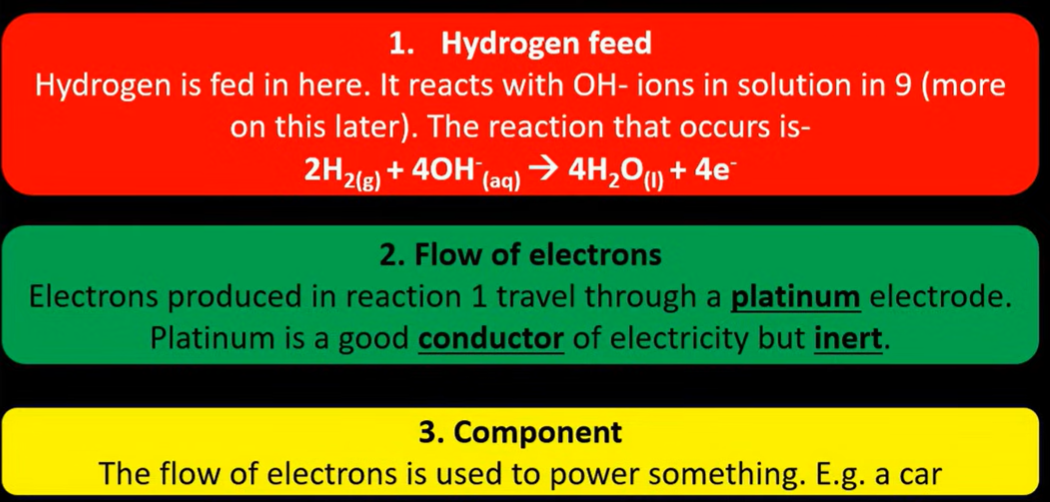
Where Hydrogen gas is bubbled into a solution containing H+ ions, since hydrogen doesn’t conduct, electrical contact is made from platinium rod (finely coated to increase surface area)
What are 2 main properties of platinum? Very unreactive, and good conductor
Standard conditions to measure electromotive force EO => 100kPa, 298K, 1 mol dm cubed of all solutions
The standard hydrogen electrode is defined as 0.00V by convention, so when another electrode connected under standard conditions, you can measure EMF the E of the species.
(-) => better reducing agents
(+) => better oxidising agents
EMF = E (on right) - E (on left)
For reaction to be feasible the EMF IS POSITIVE
OR (most positive - most negative)
Conventional representation of cells:
Vertical line shows the phase boundary between states
Same state is separated by comma
Double line represents the salt bridge
The species with highest oxidation states are next to the salt bridge
Reduced on right of salt bridge, oxidised on left
If the species is a gas or has two ions in solutions add platinum electrode
Key questions on application of batteries:
Porous separator to allow ions to move through
Paste (electrolyte to allow ions to move through)
Why carbon electrode used? Conducts electricity well
Rechargeable batteries:
Apply a stronger voltage allowing electrons to flow in opposite direction allowing reactants to reform
LITHIUM ION => SPEC
Negative electrode - powdered graphite, where the lithium releases electrons, and the ion moves to the positive electrode
Positive electrode contains cobalt IV oxide
Both act as a support medium for lithium
Lithium is lightest metal and strongest reducing agent
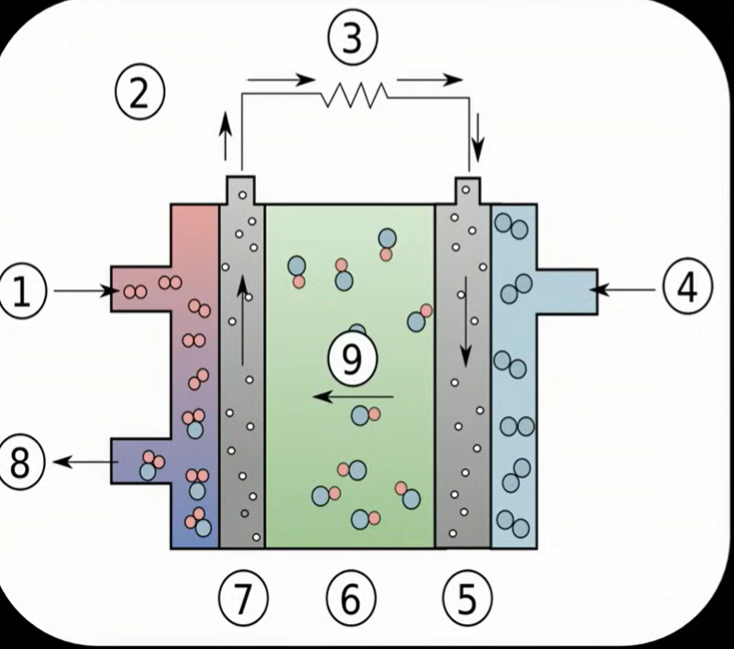
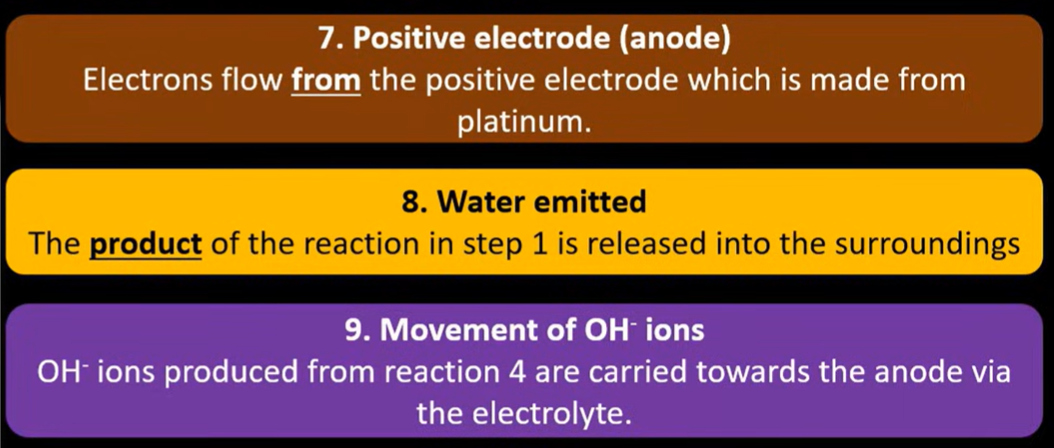
Fuel cell => spec
Requires continuous supply of reactants so concentrations are constant
Hydrogen gas is oxidised into hydrogen ions at negative electrode (platinum)
Oxygen is reduced to water at positive electrode (platinum)
Hydroxide ions travel to the negative electrode where ions combine to form water molecules
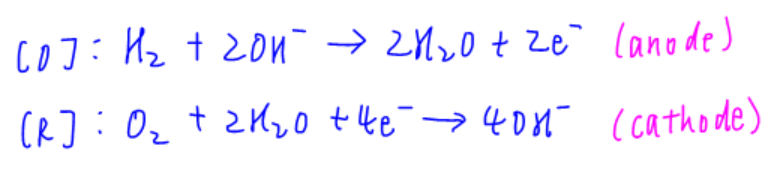
Overall:
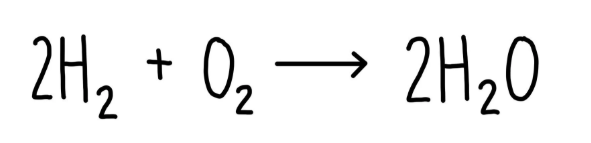

The weak acids you need to know for a-level acids, bases and buffers topic:

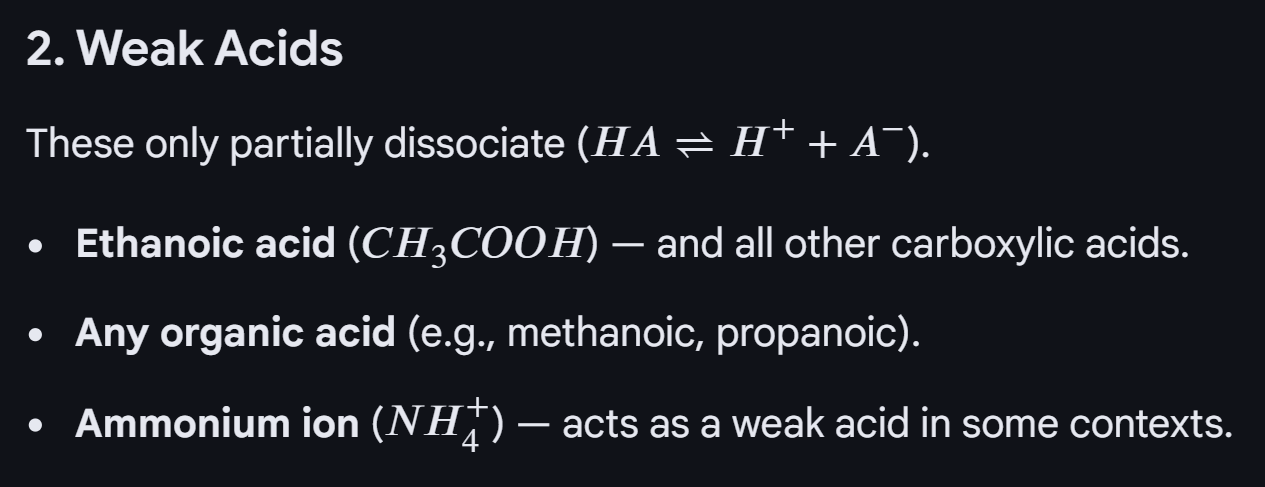

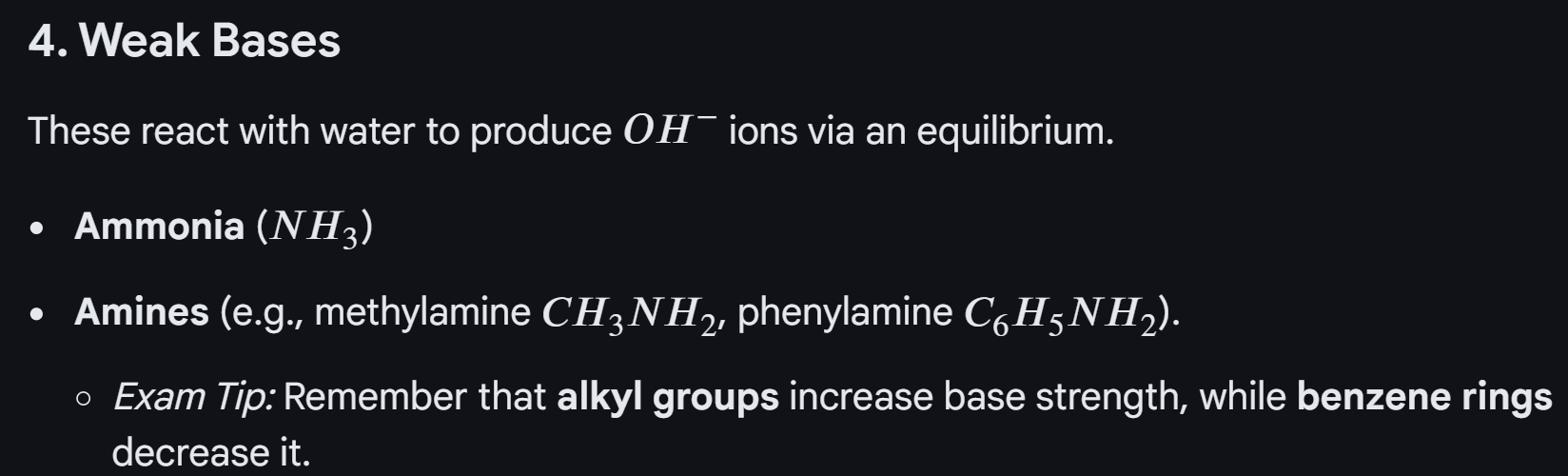
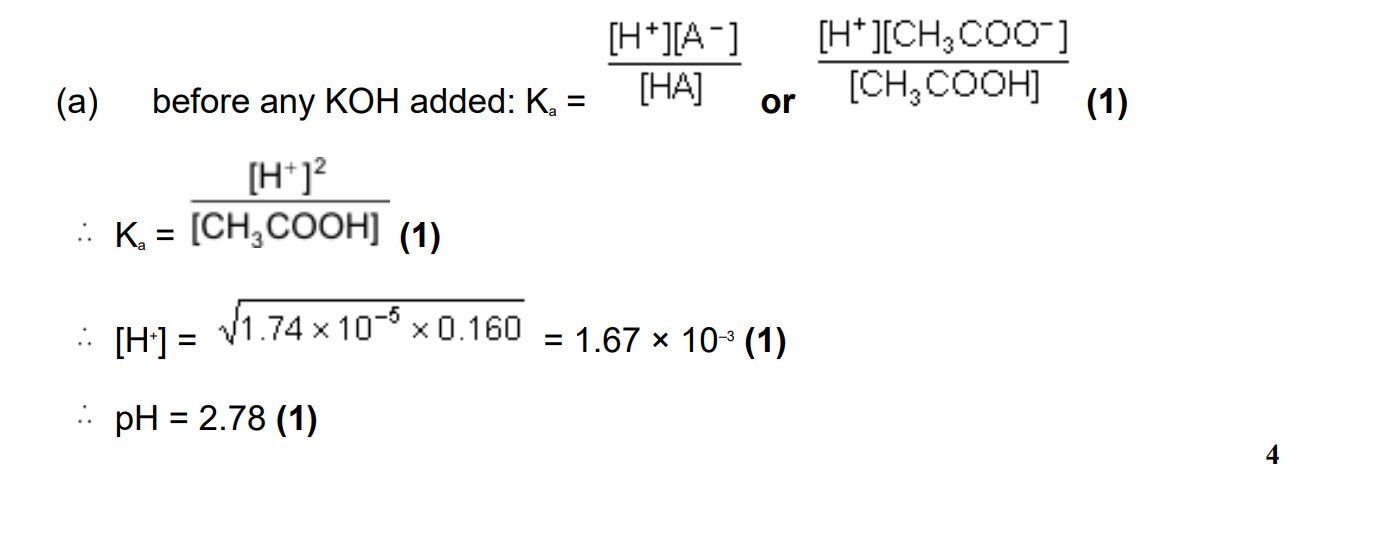


In conclusion: when we have to make a buffer we only do the subtraction to get the difference finally and substitute it into what HA, but when you have a buffer already and add acid (HA increases, A decreases) and when you add alkaline (HA decreases, A increases OR use the equation of Kw)