Kinetics
What speeds up a Chemical Reaction?
addition of a catalyst
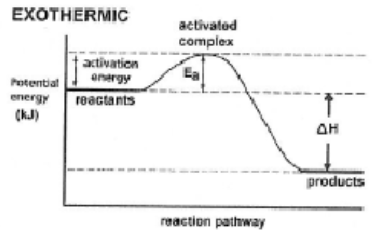
Catalyst provide a different pathway of lower activation energy (i.e. enzymes)
Increase the concentration of the reactants
Increase the temperature of the reactants
Increase the surface area
Grind it up into a powder
Reaction Orders
Zero order → concentration has no effect on rate
First Order → Doubling concentration doubles the rate
Second Order → Doubling concentration quadruples the rate
Third Order→ Doubling concentration changes the rate x8
1st Order reactions = half-life equation!
t1/2 = 0.693/k

Mechanisms
Transition states or intermediate
Formed and consumed durring a reaction; temporary chemical
ELementary Steps
Each step in a Mechanism
Rate laws = slow step
Order of Elementary Steps in a Mechanism
Unimolecular - 1st order
1 reactant with a coefficient of 1
Bimolecular - 2nd order
reactant coefficients add up to 2
Termolecular - 3rd order
reactant coefficients add up to 3
Collision Theory
Reactants must collide
With sufficient energy
Frequency
In the right orientation
Activation Energy - minimum energy needed to start a reaction
Reactions involving 2 or more molecules are likely to happen in multiple steps
Rate Law
A + B + C → D
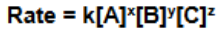
Exponent = order
K is a constant rate