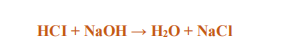titrations
A volumetric analytical method used to quantitatively determine the concentration of a particular
compound in a solution. This technique involves reacting a substance with an unknown concentration with a
standard solution whose concentration is precisely known.
a typical titration is based on the reaction between the titrant and the analyte
A titrant: a solution of precisely known concentration that is carefully added
An analyte: the "unknown" solution for which you would like to know
Equipment:
a standard solution of sodium hydroxide (NaOH) with a concentration
of 0.1 M
Phenolphthalein: an indicator to show the endpoint of the titration by allowing for a color change.
Equation: