Cancer Pathophysiology
Cancer
Learning Outcomes:
- Define cancer and cancer terminology
- Identify incidence and mortality rates
- Review cell cycle and examine this process as it relates to carcinogenesis
- Compare benign and malignant tumors including cancer cell characteristics
- Gain a better understanding of etiology and risk factors for cancer
- Describe nomenclature as it relates to neoplasms
- Describe classification and staging of malignant tumors
- Gain a better understanding of cancer treatment, including side effects and management
Definition
Cancer is the uncontrolled growth of abnormal cells in the body
- Cancerous cells are made of less well-differentiated cells that lost ability to control cell proliferation and differentiation into a mature cell
Statistics
- Nearly 1 in 2 Canadians will develop cancer at some point in their lives
- 1 in 4 Canadians will die from cancer at some point in their lives
- Cancers of the lung, breast, colon, and prostate account for half of all new cancer cases
- Breast cancer more common for women and prostate cancer more common for men
- Lung cancer is leading cause of cancer death
Cell cycle
- 5 phases of the cell cycle
- G zero
- G1
- S (synthesis)
- G2
- M (mitosis)
- Synthesis - DNA is synthesized and chromosomes are replicated
- Mitosis - cell divides and 2 daughter cells are formed
- G phases- cell is metabolically active or growing enzymes/proteins to prepare for DNA synthesis or mitotic division
- After mitosis- daughter cells either go into state of dormancy (G zero phase) where they are not actively proliferating OR if a stimulus for cell division exists, cell enter G1 to begin cell reproductive cycle again
- G1 determines overall length of cell cycle because cell spends hours or days in this phase
- Differentiation: the process by which proliferating cells become specialized
- Categories of differentiation and proliferation cells: cells that never/rarely divide, cells that continue to proliferate then die (PROGENITOR CELLS)
- Lastly is stem cells that can enter the cell cycle and produce progenitor cells when required
- Cancer cells can complete cell cycle faster by decreasing time spent in G1 phase
- Also less likely to enter or remain in G zero phase than normal cells
Cell Cycle Checkpoints
- G1-S: monitors whether DNA in chromosomes is damaged by radiation or chemicals
- G2-M: prevents entry into mitosis if DNA replication is not complete
Carcinogenesis Intro
- Process by which normal cells are transformed into cancer cells
- Caused by mutation of the genetic material of normal cells- upsets normal balance between proliferation and cell death
- Results in uncontrolled cell division and tumor development in body
Carcinogenesis Stages
- Initiation
- Exposure of cells to appropriate doses of carcinogenic agent that makes them susceptible to malignant transformation
- Promotion
- Unregulated and accelerated growth of the mutated cells and dysplasia
- Dysplasia usually indicates early neoplastic process
- Progression
- Where tumor cells acquire malignant changes and autonomous growth tendencies that promote invasiveness and metastatic capabilities
- Carcinoma in situ
- Transformation of a neoplastic lesion to one in which cells undergo no maturation and can be considered “cancer like”
- Remains localized and hasn’t invaded past the basement membrane into the tissues below surface
- Invasive cancer
- Cancer that has invaded beyond basement membrane and has potential to metastasize or spread to other body parts
Carcinogenesis- How it occurs?
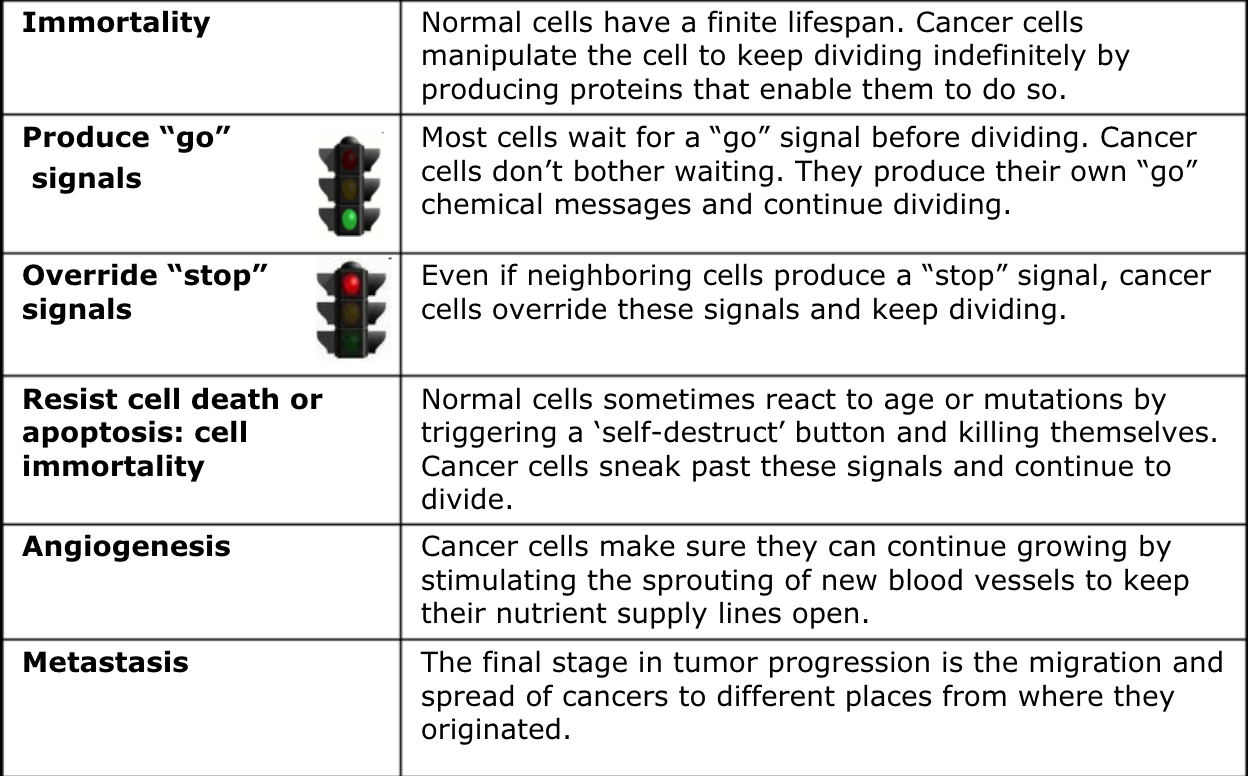
- Proto-oncogenes
- Encourage cell division
- When mutated they become oncogenes- stimulate excess division
- IMPORTANT* oncogenes result form activated or turning on of proto-oncogenes
- How it contributes to cancer:
- Develop cancer by instructing cells to make proteins or go signals that stimulate excessive cell growth and division
- Causing a cell’s growth-signaling pathway to become hyperactive
- Tumor suppressor genes (TSG)
- Inhibit cell division
- When mutated it inactivated these genes causing inhibition of cell division that normally prevents excessive growth
- IMPORTANT* cause cancer when they are inactivated/turned off
- How it contributes to cancer:
- When TSG does not function properly, cells with DNA damage continue to divide and accumulate more DNA damage that eventually lead a cell to grow and divide uncontrollably
- Like having a brake pedal that does not work
- ***people who inherit increased risk of developing cancer are often born with one defective copy of TSG
- Because genes come in pairs (one from each parent), inherited defect in one copy will not lead to cancer because other normal copy is still functional
- Defective TSG called APC gene causes familial adenomatous polyposis- condition where people develop thousands of colon polyps sometimes leading to colon cancer
DNA Sequencing
- 3 systems that help avoid runaway cell division
- DNA repair system
- Instruct a cell to repair damaged DNA
- Mutations in DNA repair system:
- A change in single base along the base sequence of a gene (like a typo error)
- One or more bases added or deleted
- Large segments of DNA molecule repeated, deleted or moved
- Mutations can lead to failure in repair
- When a mistake occur during DNA replication- repair proteins recruit enzyme EXO1 (exonuclease that chops off the mutant strand)
Apoptosis
- When old cells become damaged over time they’re eliminated by apoptosis
- Tumor suppressor p35 protein initiates cell suicide
- Tumor suppressor gene and p35 protein are most frequently mutated genes in human cancer
NK Cells
- Can target tumors and cancer cells and kill them
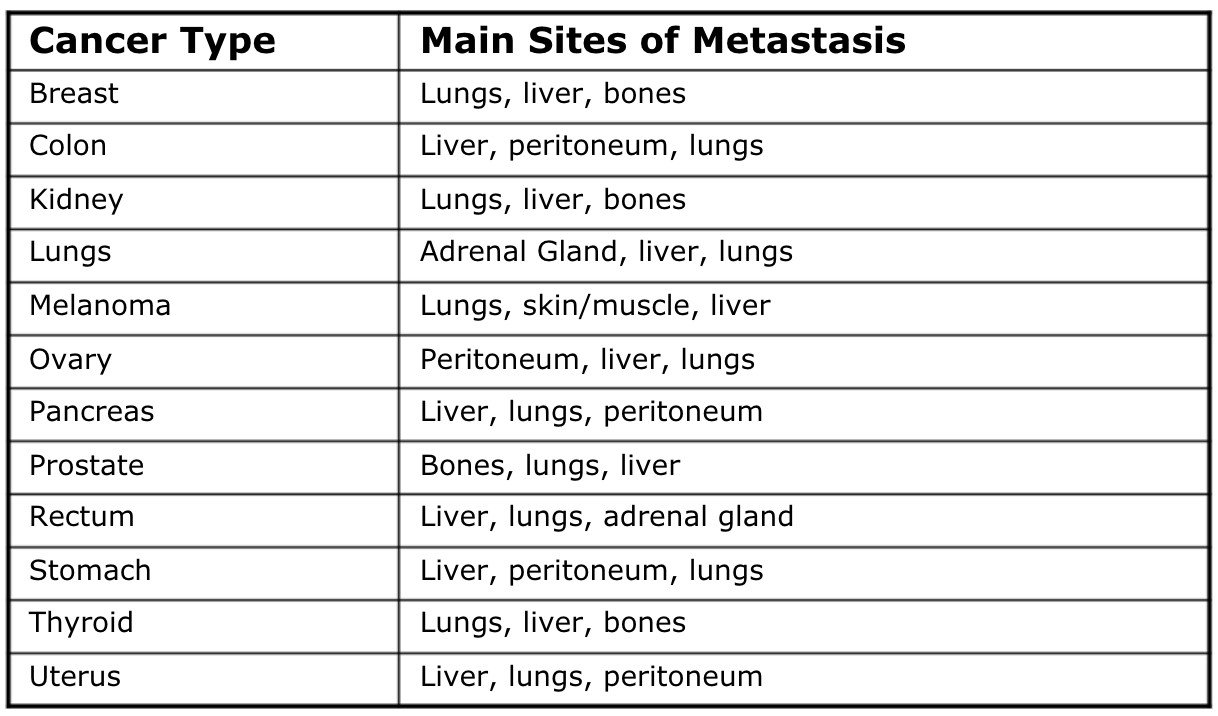
Metastasis
- Spread of cancer from original location to other parts of body
- Occurs in 2 ways:
- Malignant cells directly invade or extend into adjacent organs or sites
- Individual cancer cells move away from primary tumor and enter body or lymph circulation
- Most common sites are lungs, bones and liver
Angiogenesis
- Process of forming new blood vessels
- Begins when tumor becomes large enough where it needs to increase supply of nutrients & oxygen
- Low oxygen (hypoxia) triggers tumor and environment to release signals that result in growth of BV into the tumor
Tumor Angiogenesis
- The proliferation of a network of vessels that penetrates into cancerous growths, supplying nutrients & oxygen and removing waste products
- Steps:
- Cancerous tumor cells release molecules that send signals to surrounding normal host tissue
- Signaling activates certain genes in host tissue that make proteins to encourage growth of new blood vessels
Angiogenesis Inhibitors
- Endostatin
- Molecules that directly inhibit the growth of endothelial cells
- Thalidomide
- Prevents endothelial cells from forming new blood vessels
- Avastin (first to be FDA approved)
- Molecules that interfere with steps in angiogenesis signaling cascade
- Delays tumor growth
- Interferon-alpha
- Naturally occurring protein
- Inhibits the production of growth factors from starting the angiogenesis signaling cascade
Etiology
- Even though cancer is genetic, only 5-10% if inherited
- Chances of getting cancer increase with age
- Cancer screening
- High risk individuals for prostate cancer should start testing from age 45
- High risk: person w/ known gene mutation that increases risk for BC, first degree relative of someone with gene mutation, is assessed as having 25% or greater lifetime risk of breast cancer based on family hx, has had radiation therapy of the chest
- Carcinogens: substances directly responsible for damaging DNA, promoting or aiding cancer
- Include UV light, radiation, chemical, bacteria, viruses or medical treatments
- Lifestyle factors (alcohol, smoking, obesity, inactivity,
Nomenclature
- Neoplastic: abnormal growth of new tissue
- Benign: noncancerous tumor growth
- Composed of well differentiated cells, resembling cells of tissue of origin
- Characterized by slow progressive rate of growth that can stop or regress
- Lost ability to suppress genetic program for cell proliferation but retained program for cell differentiation
- Remain localized to site of origin, lack capacity to infiltrate, invade or metastasize to distant sites
- Develop surrounding rim of compressed connective tissue (fibrous capsule)- responsible for sharp line of demarcation between benign tumor and adjacent tissues (known as encapsulated, is a factor for surgical removal)
- Named by adding suffix “oma”
- Malignant: cancerous tumor growth
- Less well differentiated, lost ability to control cell proliferation/differentiation into mature cell
- Anaplasia: loss of cell differentiation in cancerous tissue
- Poorly differentiated: poorly resembles cell it arose from
- Undifferentiated: malignant cells are immature, embryonic and no resemblance to cell it arose from
- Grow rapidly in disorganized/uncontrolled manner to invade surrounding tissues and blood vessels
- Rob normal tissue of essential nutrients and release enzymes, toxins and cytokines that destroy normal tissue
- Have cells that break loose and form metastases
- Have suffix “carcinoma” or “sarcoma”
Staging Malignant Tumors
- Stage I: small, localized, curable
- Stage II: locally advanced
- Stage III: locally advanced, lymph node involvement
- Stage IV: inoperable, metastatic
- IA: no symptoms
- IIB: symptoms like fever, night sweats and weight loss
- TNM classification: tumor, nodes, metastases
- Only lymph nodes draining area of the primary tumor are considered in classification
Molecular Tests
- Tumor markers - PSA, CEA, AFP CA125 and Estrogen receptors- occur in blood or tissue useful in patient diagnosis or management
- PSA- prostate specific antigen measures levels of PSA in blood, high levels can be marker for prostate cancer
- Can also be high in men with infection/inflammation of prostate or benign prostate hyperplasia
- CEA- carcinoembryonic antigen
- Type of protein that can be found in many different cells of body but usually associated with some tumors
- Benign and malignant conditions can increase CEA level
- Colon and rectum cancer most commonly increase CEA
- ***best use of CEA is as a tumor marker- especially for cancers of GI tract
- Rising CEA indicates progression or recurrence of cancer
- AFP- normal fetal serum protein synthesized by liver, yolk sac and GI
- Major component of fetal plasma normally in pregnant women
- Rise is usually only seen in diseases like benign liver diseases and hepatocellular carcinoma
- CA 125- antigen present on 80% of ovarian carcinomas
- Circulates in serum of patients w/ ovarian carcinomas, used as marker to monitor disease
- Decrease =good therapy, increase =recurrence
- ER+- have receptors for estrogen on surface, growth requires presence of estrogen
- ER+ tumors more affected by hormonal treatment and less aggressive
Radiation Therapy
- Immediately kills cells, delays or stops cell cycle progression or causes damage to cell’s DNA causing cell death after replication
Chemotherapy
- Cells mainly affect by chemo
- Blood cell forming bone marrow, hair follicles, lining of the mouth and digestive system
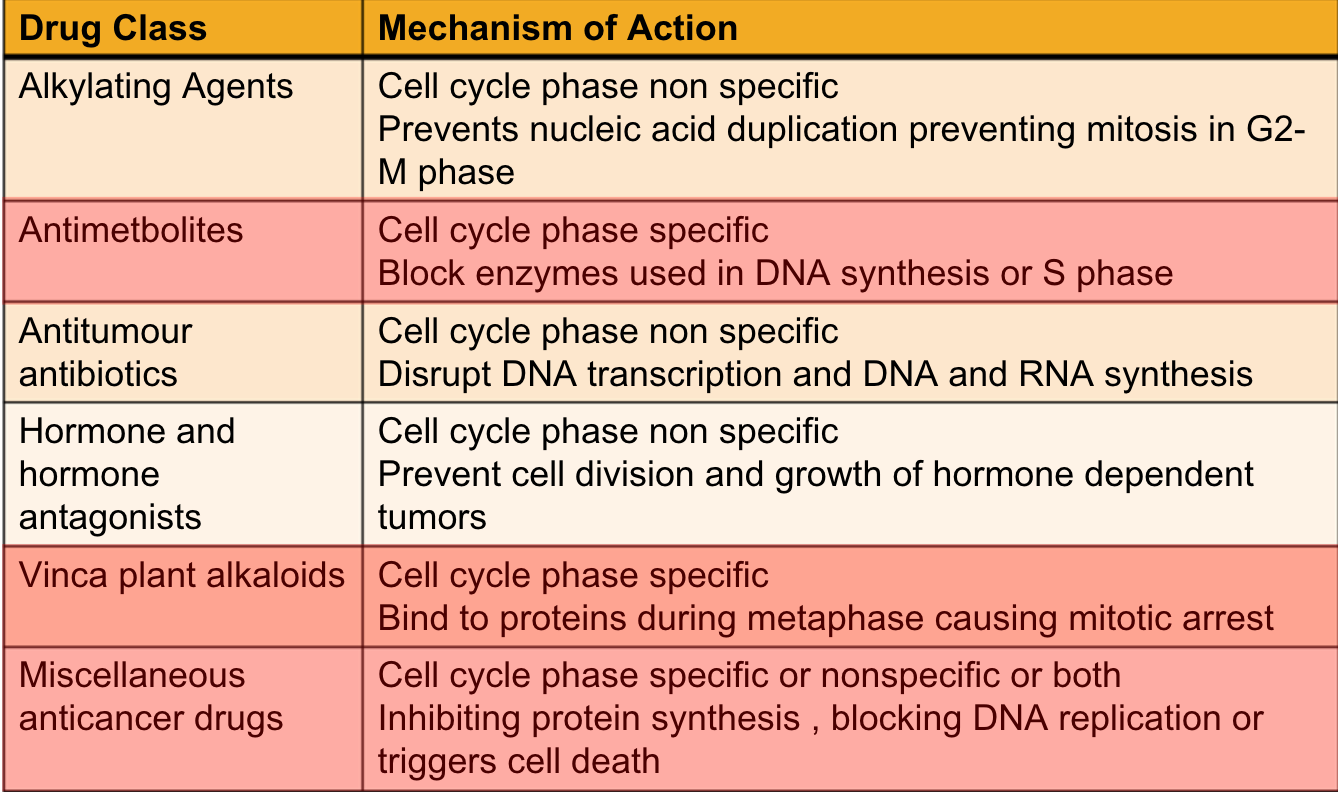
- Chemotherapeutic drugs most effective against frequently dividing cells or all phases of cell cycle except G zero
Classifications
- Cell cycle phase nonspecific drugs are active on cells in dividing or resting state
- Effective on large tumors that have few active cells dividing at time of admin
- Usually given as single bolus injections
- Cell cycle phase specific drugs are given in minimal concentrations through continuous dosing methods
Hormonal Therapy
- Admin of drugs designed to disrupt hormonal environment of cells
- Used for cancers that are responsive to or dependent on hormones for growth
- Can treat hormone receptors positive breast cancers
- By lowering amount of estrogen in body
- Or by blocking action of estrogen on breast cancer cells
- Estrogen makes hormone receptor positive breast cancers grow
- Hormonal therapies are not effective against hormone-receptor-negative breast cancers
Biotherapy
- Biologic response modifiers can trigger immune system to indirectly affect tumors
Targeted Therapy
- Drugs that selectively attack malignant cells while leaving normal cells unharmed
- “Molecularly targeted drugs/therapies”
- Interfere with cancer cell division, processes of apoptosis or angiogenesis
- Thalidomide
BMT and PBSCT
- Restore stem cells that have been destroyed by high doses of chemo or radiation
- 3 types of transplants
- Autologous- patients receive their own stem cells
- Syngeneic- patients receive stem cells from identical twin
- Allogeneic- patients receive stem cells from brother, sister or parent
Side Effects of Treatment
Intergumentary
- Alopecia
- Hair loss occurs 10-21 days after drug treatment, is temporary will regrow when drug discontinued
- Hair thinning
- Local or systemic hypersensitivity reactions
- Review pt’s allergy hx, monitor for hypersensitivity of anaphylaxis, test doses as ordered, maintain good hygiene, avoid perfume lotions
- Extravasation
- Inadvertent leakage of chemo drug from a vessel into surrounding tissue
- Assess for immediate/delayed pain, tightness, blister or sloughing of tissues
- Prompt admin of antidotes to minimize tissue damage
MSK
- Aches/pain
- Pain meds
- Fatigue
- Conserve energy & plan rest periods
Nervous System
- Neurotoxicity
- Monitor for signs of weakness, numbness, tingling extremities and foot drop
- Ototoxicity
- Some chemo drugs can cause hearing changes, monitor for tinnitus, hearing loss and vertigo
- Sleep pattern disturbances
- Vitamins, corticosteroids and neuroleptics for N/V can negatively impact sleep
- Anxiety and depression
- Set small achievable daily goals, participate in enjoyable and divisional activities and share feelings
- Memory changes
- “Chemo fog”
- Use calendars and lists, provide pill boxes or dosettes
Endocrine System
- Hypercalcemia
- Monitor serum calcium levels, polyuria and mental status changes
- Hyperglycemia
- Patients on steroids for cancer can develop high BS
- Hyperkalemia
- Rapid amount of cellular destruction causes contents of cell to move into blood stream (tumor lysis syndrome)
- Hypernatremia
- Caused by dehydration, loss of fluids. Monitor serum sodium levels, symptoms of thirst, dry mucous membranes, poor skin turgor, restlessness and lethargy
- Hyperuricemia
- Monitor serum and urine uric acid levels, daily I/O, rigorous hydration if indicated
Cardiovascular System
- Cardiac toxicity
- Drugs- cyclophosphamide and doxorubicin
- Baseline ECG, echo, cardiac enzymes before chemo, monitor for changes
Digestive System
- Hepatotoxicity
- Monitor liver function tests, assess for jaundice, tenderness over liver, urine and stool color changes
- Anorexia
- Eat small frequent meals high in protein, monitor weight
- N/V and constipation
- Diarrhea
- Mucositis/Stomatitis
- Symptoms appear 3-5 days after local radiation or systemic chemo
- Can be painful enough to require analgesic IV drip
Urinary System
- Renal toxicity
- Assess baseline, encourage oral intake, monitor I/O and weight changes
- Cystitis
- Some chemo can cause inflammation and bleeding of bladder lining
- Increase fluid intake, empty bladder frequently, administer antidote
Pulmonary System
- Pulmonary toxicity
- Individuals over 70 at greater risk
- Assess baseline resp function, monitor resp status
Reproductive System
- Reduced fertility
- Fetal death
Lymphatic and Hematological
- Neutropenia
- Abnormally low count of neutrophils in blood stream (> 2000 cells/cubic mm)
- Monitor CBC, infection, sepsis, frequent temperatures, health teaching
- Thrombocytopenia
- Reduction in number of circulating platelets below 30,000 per cubic mm
- Monitor CBC, assess for bruising, purpura, petechiae, nose bleeds, bleeding gums or tarry stools
- Platelet transfusions can be required
- Anemia
- Abnormal or low hematocrit and hemoglobin below 80g/L
- Monitor CBC, assess paleness, chest pain, SOB, heart palpitations, dizziness, lethargy