Respiratory System Part 3
Key Vocabulary:
Carbonic Acid: Weak acid that forms from carbon dioxide and water.
Medulla Oblongata: Lower portion of the brainstem.
Phrenic Nerve: Nerve that controls the diaphragm.
Pons: Middle portion of the brainstem.
Respiratory Acidosis: Acidic condition in the blood caused by increased carbon dioxide.
Respiratry Alkalosis: Alkaline condition in the blood caused by a decrease in carbon dioxide.
Surface Tension: Force created by weak bonds between water molecules.
Surfactant: Substance secreted by Type II Alveolar Cells that reduces surface tension.
Type I Alveolar Cells: Mucous secreting cells lining the inside of the alveolus.
Type II Alveolar Cells: Surfactant secreting cells lining the insise of the alveolus.
Introduction:
Breathing is automatically controlled by the nervous system which monitors the blood for oxygen, carbon dioxide and pH. Once oxygen reaches the alveolus, a delicate balance of cellular secretions helps to maintain the integrity of the alveolus so that oxygen and carbon dioxide can freely move between the alveolus and capillaries. Besides delivering oxygen to the cells and eliminating carbon dioxide, the respiratory system plays a vital role in maintaining blood pH which must be maintained within a very narrow range. Since there is such a close relationship between the respiratory system and pH, problems in the respiratory system can cause imbalances in pH called respiratory acidosis and alkalosis.
Respiration and the Nervous System
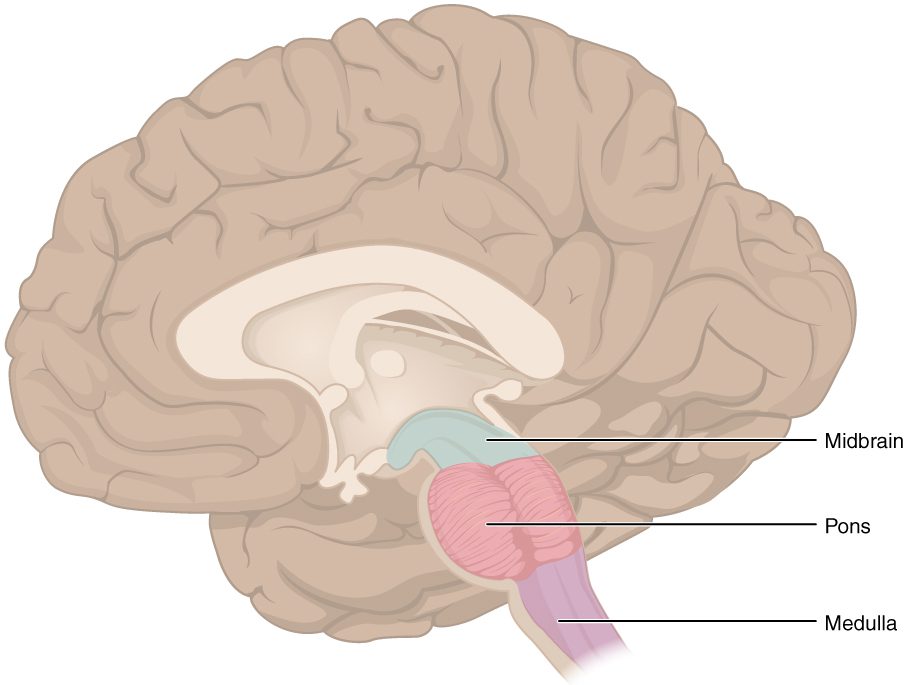
Neural control of respiration begins in the brainstem at the medulla oblongata and the pons. There are 2 paired groups of neurons in the medulla oblongata that work to control the rate and rhythm of breathing. Groups of neurons in the pons help to fine tune breathing and act as a backup system in case there is damage to the medulla oblongata.
The brainstem centers act unconsciously, but can be overriden by higher brain centers. For example, you can consciously increase or decrease your breathing.
The respiratory centers monitor the blood for oxygen, carbon dioxide and pH. There are also chemoreceptors located in other parts of the circulatory system such as in the carotid arteries and aorta.
The brainstem centers connect to the diaphragm by way of the right and left phrenic nerves.
Examine the parts of the brainstem involved in breathing:
Surface Tension and Surfactant
The alveoli contain 2 types of cells. The Type I cells secrete mucous on the inside of the alveolus. Since mucous contains mostly water, surface tension can develop which works to collapse the alveolus.
Surface tension is a force created by weak bonds between water molecules and creates an inward pulling force on the alveolus. The alveolar Type II cells secrete surfactant which works to break the weak bonds and reduce surface tension.
This concept is important in premature infants with underdeveloped lungs. In these cases, diminished secretion of surfactant can cause lung collapse or respiratory distress syndrome unless surfactant is administered.
Transport of Oxygen and Carbon Dioxide in the Blood
Most of the oxygen transported in blood is bound to hemoglobin to form oxyhemoglobin. A small amount of oxygen is dissolved in plasma. Each hemoglobin molecule can bind with four oxygen molecules. Hemoglobin can also release oxygen to form deoxyhemoglobin. There are almost 300 million hemoglobin molecules in one red blood cell. The functional characteristics of hemoglobin are also variable and respond to changes in PO2, pH, and temperature.
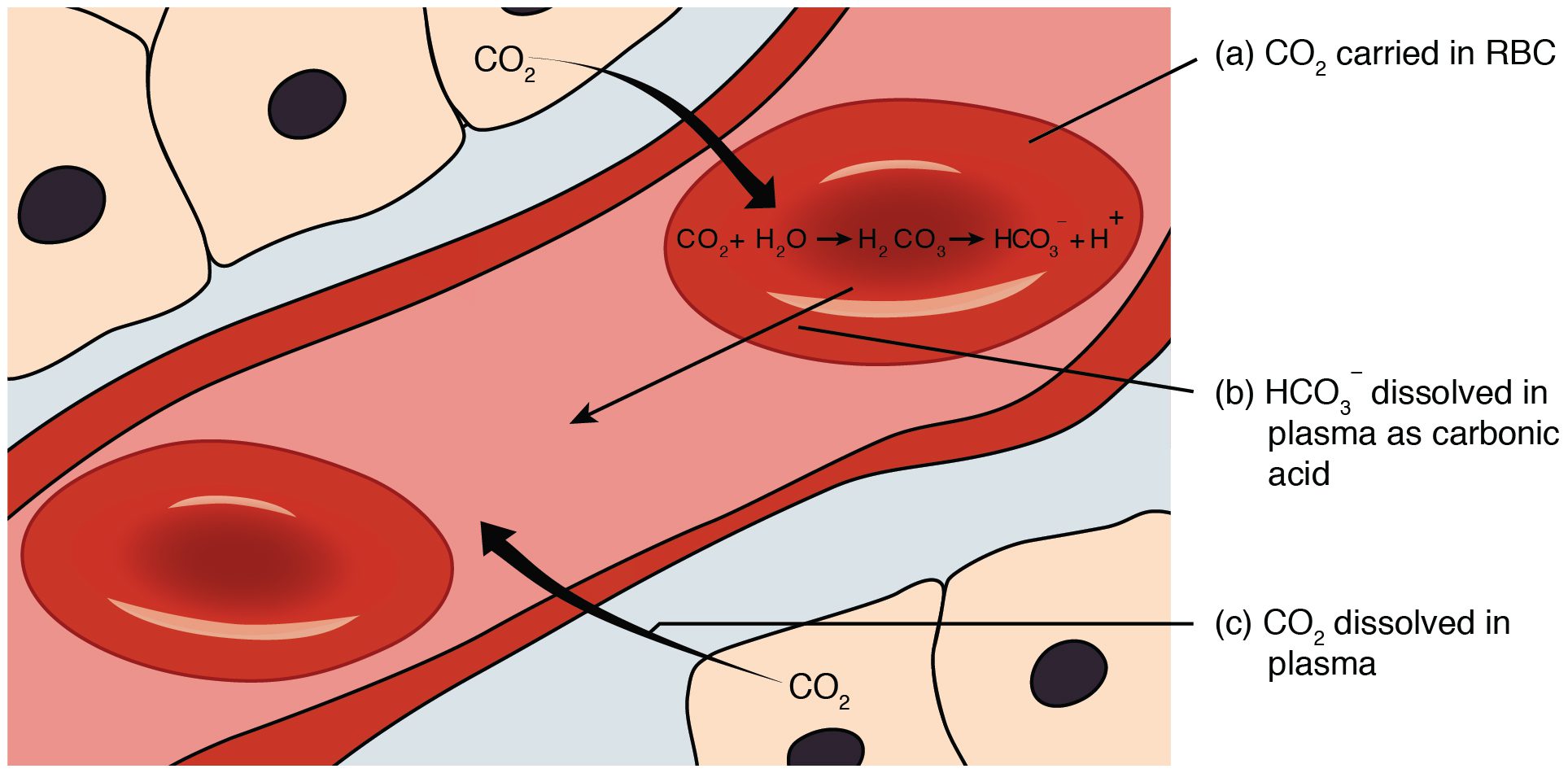
Carbon dioxide is transported in the blood by 3 mechanisms. These include carbon dioxide dissolved in plasma, carbon dioxide combining with hemoglobin and storage of carbon dioxide in the bicarbonate ion.
About 7% of the total carbon dioxide in blood is dissolved in plasma. Carbon dioxide also combines with hemoglobin to form a compound known as carbaminohemoglobin. About 23% of carbon dioxide is transported as carbaminohemoglobin. The majority of carbon dioxide (about 70%) is transported in the bicarbonate ion.
Carbon dioxide diffuses into red blood cells and encounters the enzyme carbonic anhydrase to form carbonic acid. Carbonic acid dissociates into bicarbonate and hydrogen ions. Bicarbonate ions diffuse out of the red blood cells into the plasma.
The reaction is reversible with either the storage or release of carbon dioxide depending on what is needed. For example in areas of low PCO2 such as in the alveoli the reaction will work in the direction to release CO2 for removal by the lungs. In areas of high PCO2 such as in the tissues the reaction will work in the direction to store CO2 in the bicarbonate ion.
Examine the following diagram illustrating how carbon dioxide is carried in the bicarbonate ion in the blood:
Respiratory Acidosis and Alkalosis
Because most of the carbon dioxide is transported by the bicarbonate ion with subsequent release of hydrogen ions, a buildup of carbon dioxide in the blood will produce a lower pH. Carbon dioxide and water combine to form carbonic acid in the blood. Carbonic acid dissociates into bicarbonate and hydrogen ions. If the respiratory system cannot release enough carbon dioxide, the subsequent production of hydrogen ions makes the blood acidic. This is known as respiratory acidosis and can result from obstructive diseases such as emphysema or chronic bronchitis. You can generate a mild case of respiratory acidosis by simply holding your breath. The cells continue to produce carbon dioxide but the lungs are not removing it through exhalation. Carbon dioxide builds up in the lungs producing the hydrogen ion byproduct and the blood begins to become acidic.
Likewise you can produce a mild state of respiratory alkalosis by hyperventilating. In this case too much carbon dioxide is removed by the lungs and the hydrogen ion concentration subsequently decreases.