Organic Chemistry
Fats and Oils
Triglycerides are an important group of esters known as fats and oils
Triglycerides are 3 fatty acids attached to a glycerol backbone
Fatty acids are a carboxylic acid with a long aliphatic chain

Fats and oils are formed from the esterification reaction between glycerol and fatty acids.

Types of fats and oils
Solid fats
Straight chain fatty acids
Saturated or trans fatty acids
Fat crystals suspended in oil
Liquid fats (oil)
Contain kinked chain fatty acids
Mono-or polyunsaturated fatty acids
No fat crystals
Melting temperature
Melting temperature is directly proportional to degree of saturation
Higher saturation (less double bonds) will have a higher melting point
Lower saturation (more double bonds) will have a lower melting point
Animal Fats
Saturated fats (no double bond) are usually animal fats.
These have higher melting points and are more likely to be solid at room temperature.
Plant Fats
Unsaturated fats (double bond) are usually plant fats.
Lower dispersion forces
Lower melting points and are more likely to be liquid at room temperature
Hydrogenation reaction
Converts unsaturated liquid vegetable oils into more versatile solid products
Addition reaction where hydrogen atoms add to some of the double bonds
This forms an undesirable side reaction
Converts some of the cis double bonds into the trans form
The trans isomers have higher dispersion forces and allow their molecules to pack more efficiently than the cis form
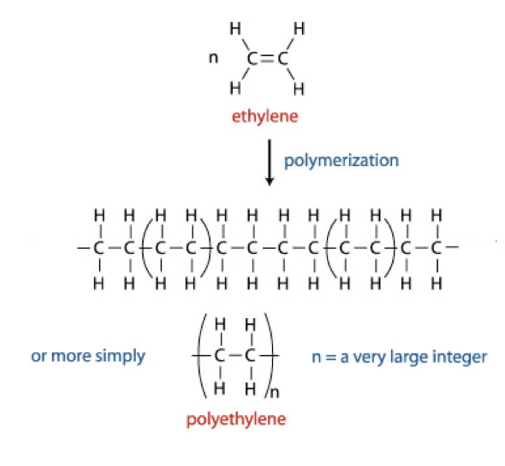
Polymers
Polymers are very large organic molecules made up of small repeating units.
They are formed from reactions between small organic molecules called monomers.
The process of forming a polymer is called polymerisation.
Addition polymers are formed from certain alkenes (containing a C=C bond).
In these reactions, each molecules undergoes an addition reaction and forms a single bond with the next molecule.
Not a spontaneous reaction requires high temperatures and high pressures. This makes short chains of polyethene form and branch off the main polymer chain. Forming low density polymers which are loosely packed. This is more flexible.
If a catalyst is used and lower pressure than it produces a polymer with less branches. These are tightly packed and called high-density polymer.
Condensation polymers are formed from monomers containing two reactive functional groups, typically:
a carboxylic acid and alcohol
a carboxylic acid and amine
In these reactions, two monomer molecules combine, producing water.
The monomers must possess two reactive functional groups (e.g. one monomer with two alcohol groups and another monomer with two carboxylic acid functional group)
Polypeptides
Amino acids have the general formula:
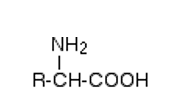
For drawing polypeptides, we rewrite the formula as:
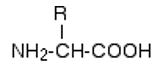
Amino acids join together in a condensation reaction
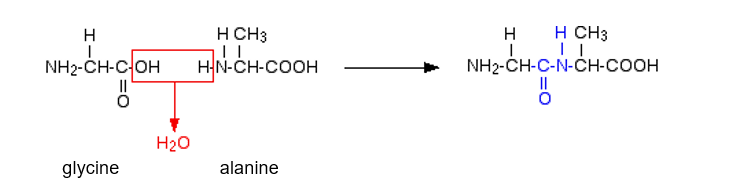
The bond formed between the two amino acids is called a peptide bond. When a large number of amino acids are joined by peptide bonds the resultant structure is known as a polypeptide.
Nylon
A type of condensation polymer
Polymer molecules that are composed of polyamides
Properties of nylon
It is strong and elastic
It is easy to launder
It dries quickly
It retains its shape
It is resilient and responsive to heat setting
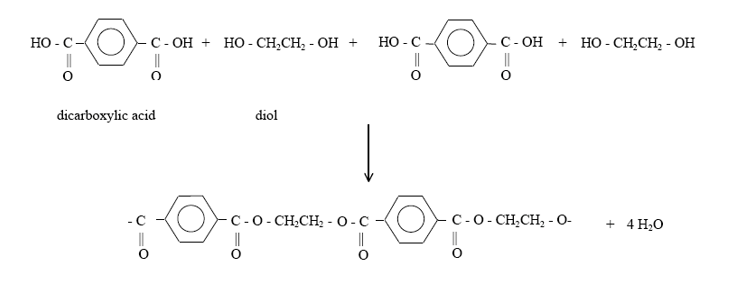
Polyester
Polyester are long-chained polymers composed of ester groups in the main chain
Formed from a dicarboxylic acid and a diol
Most common is called polyethylene terephthalate (PET).
This is made from benzene-1,4-dicarboxylic acid and an alcohol, ethane-1,2-diol
In PET fibres, the molecules are mainly arranged in one direction, in film, they are in two directions and for packaging, they are in three directions
Uses
capacitators, graphics, film base and recording tapes
fibres for a very wide range of textile fibres
bottles
food packaging
electrical components
magnetic tape
backing for adhesive tape
sail cloth
Properties
Can be produced with varying degrees to crystallisation providing a range of rigidity absorbs very little water
Good gas barrier
Excellent moisture barrier
Chemically resistant to acids, oils, alcohol
Highly transparent and colourless
High mechanical strength
Low density
Impact resistant
Alpha amino acids
Amino acids contain a carboxyl (-COOH) group and an amine (NH2) group.
In alpha amino acids, these groups are attached to the same carbon atom.
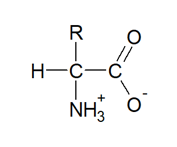
Zwitterions
Protons move from the OH group in the COOH group and join the NH2 group to form a Zwitterion which looks like this.
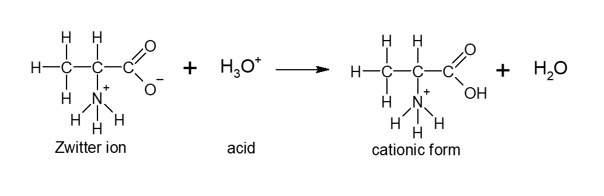
These zwitterions react with acids to produce a cationic form and water.
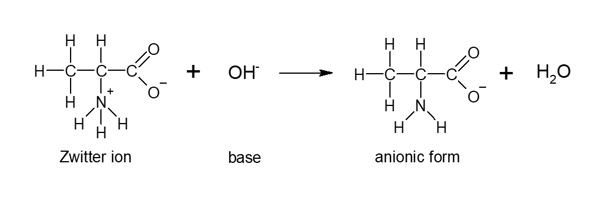
These zwitterions react with bases to produce an anionic form and water.
Peptide formation
The amino acids form peptides by loosing the OH in the COOH group and attaching the amino group from the next monomer by removing a hydrogen ion.
Protein
Proteins are a diverse group of large and complex polymer molecules, made up of long chains of amino acids
Proteins act as:
Structural components of tissues (such as muscles)
Hormones (such as insulin)
Antibodies (part of the body’s immune system)
Biological catalysts (enzymes)
What is the difference between peptides and proteins?
Proteins do not have a “n” because they are not repeating polymers.
For proteins the sequence is important e.g. Ala-Cys-Asn is not the same as Ala-Asn-Cys.
Structure and function of proteins
Three levels of structure can be identified as contributing to a protein’s function:
Primary structure: the sequence of amino acids
Secondary structure: the regular arrangement of various sections of the protein chain
Tertiary structure: the protein’s overall 3-D structure
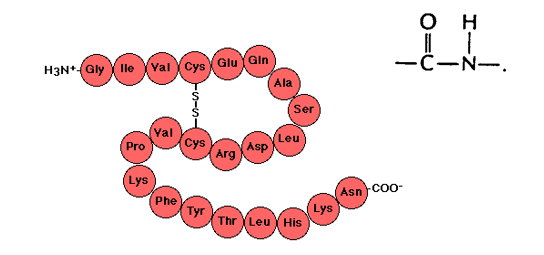
Primary structure of proteins
The primary structure of proteins is the linear sequence of individual alpha amino residues
Amino acids join by forming amid (or peptide) bonds
This the part of the protein sequence containing the amino aids Alanine, Glycine and Serine
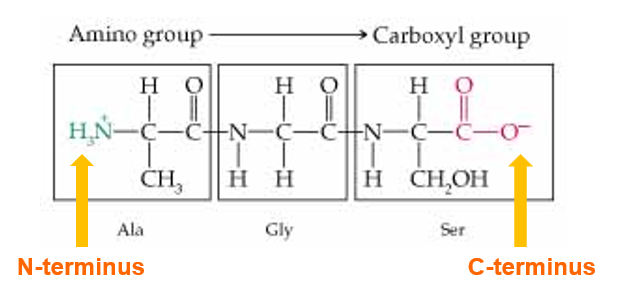
The amino acid sequence is always listed from the N-Terminus of the protein and the 3 letter abbreviation is used for each amino acid. In this case Ala-Gly-Ser.
Secondary structure of proteins
The secondary structure of a protein relates to the way individual amino acids in a protein chain bond to other amino acids in the same or adjacent chains.
Common secondary structures are:
α-Helix
β-pleated sheets
Both of these structures are held together by H-bonding.
α-Helix
The H-bonding will occur between the lone pair of electrons on the oxygen of the carbonyl (C=O) group and the polar hydrogen atom on an amide group (NH).
The amide group (NH) must be 4 residues (4 peptide links) further along the spiral from the carbonyl group (C=O) in order to form an α-Helix.
β-pleated sheets
In a beta-pleated sheet, the chains are folded so that they lie alongside each other
This is structure is based on Hydrogen bonding between the lone pairs of electrons from the oxygen of the carbonyl group and the polar hydrogen on the amide group
β-pleating will occur when the amino acids in the polypeptide chains to fit closely together
The secondary structure plays an important role in determining the functioning of the protein. Factors such as heat, pH and the presence of salts can affect the H-bonding and therefore the stability of the secondary structure.
Tertiary structure
The tertiary structure of a protein is held together by interactions between the side chains - the “R” groups.
There are several ways this can happen including:
Dispersion forces between non-polar side chains
Hydrogen bonding between polar side chains
Ionic attraction between ionisable side chains such as COO- or NH3+
Dipole-Dipole attraction between polar side chains
Disulfide bridges (a covalent bond between Sulfur atoms on the amino acid cysteine)
Biodiesel
Biofuels like bioethanol and biodiesel are produced from biomass and are considered fossil fuel alternative
Biofuels are renewable resources while the petroleum they replace is not
Biofuels are essentially natural products and exposure to these is of limited or no consequence to human health or the natural environment
Petroleum based fuels are toxic and harmful when accidentally released into the environment
Carbon dioxide emissions from biofuels can be balance with the Earth’s carbon cycle as the carbon in these fuels simply replaces atmospheric carbon dioxide absorbed by plants as they produce the biomass originally used to make the fuel.
Biofuels such as bioethanol and biodiesel are considered fossil fuel alternatives
Ethanol production
There are two methods to produce ethanol
Ethanol production from ethene (acid catalysed)
Ethanol production from fermentation (enzyme catalysed) → bioethanol
Only the second method is consistent with green chemistry
Ethanol production from ethene
This is the acid-catalysed addition of water to ethene
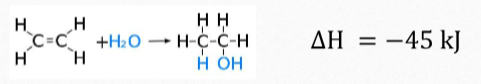
Steam and ethene are passed over a bed of silica particles coated with pure phosphoric acid (the catalyst) at a temperature of 300 degrees and a pressure of 60-70 atm.
Use:
Production of other industrial chemicals, not used as fuel or beverage
Advantage:
This is a much quicker process than the production of ethanol from fermentation. Can be set up as a continuous process.
Disadvantage:
Produced from non-renewable petroleum
Ethanol production from fermentation (bioethanol)
Bioethanol is an alcohol produced by the natural fermentation of the carbohydrates (such as starch) in sugar beet/cane or wheat crops.
Bioethanol can be produced form the fermentation of plant sugars (produced in photosynthesis)
Glucose from photosynthesis

As this step is taking in carbon dioxide from the atmosphere, the combustion of bioethanol, which produces carbon dioxide could be considered carbon-neutral.