Equilibrium Chem (copy)
Equilibrium system
is a state in which the rates of the forward and reverse reactions are equal, leading to a stable concentration of reactants and products over time.
Chemical equilibrium- A condition where the concentrations of reactants and products remain constant, although they are still reacting, effectively reaching a dynamic balance.
Reversible process
involves a dynamic balance, where the system can shift in response to changes in conditions, such as temperature or pressure, to maintain equilibrium. shifts both directions

Equilibrium System
In an equilibrium system, the rates of the forward and reverse reactions are equal, leading to constant concentrations of reactants and products.
K eq > 1 = products favored, reactants are less concentrated at equilibrium, indicating a strong tendency for the reaction to proceed towards product formation.
K eq < 1 = reactants favored, indicating that at equilibrium, the concentration of reactants is higher than that of products.
Solids/ liquids are left out of equilibrium systems because their concentrations do not change during the reaction and do not affect the equilibrium expression.
Common Ion Effect- states that the presence of a common ion in a solution will shift the equilibrium position, reducing the solubility of a salt.
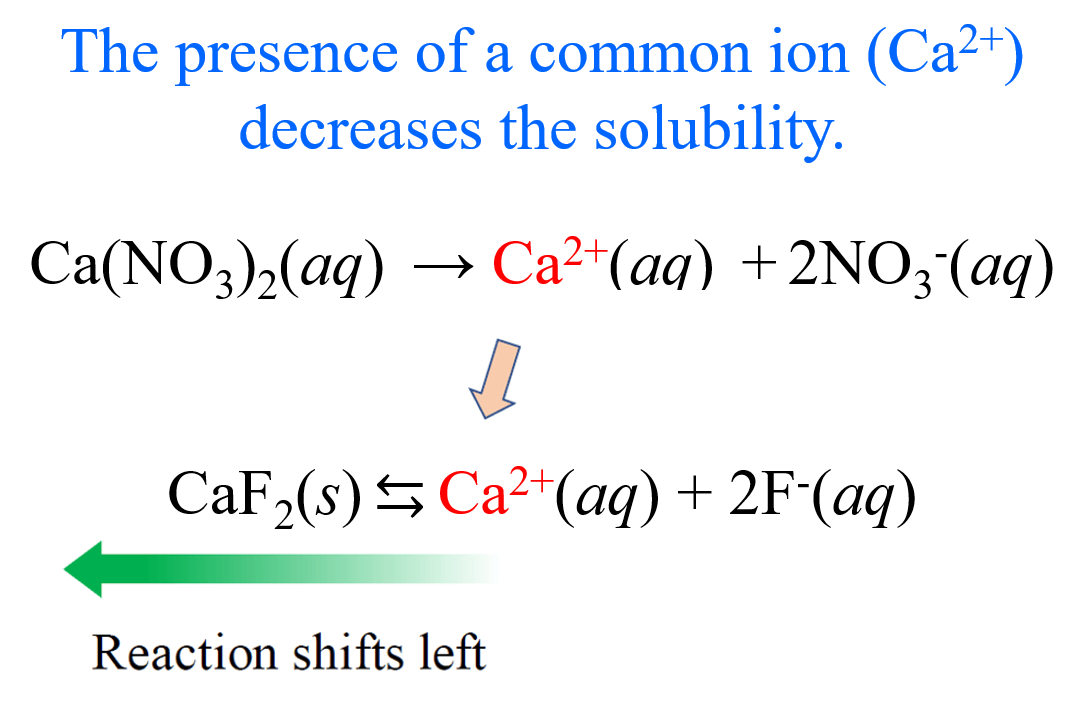
K sp | K eq |
specifically refers to the equilibrium constant for the dissolution of an ionic solid in a solution | general term for the equilibrium constant of any reversible reaction |
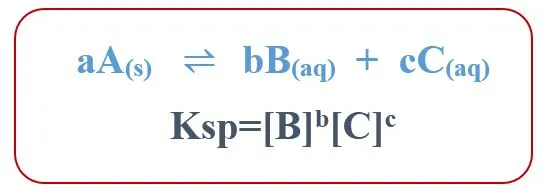
Rate of Chemical reaction- amount of Reactant changing per unit of time.
4 factors that affect rate the of chemical reaction are temperature, concentration of reactants, surface area and presence of catalyst.
Temperature: Generally, an increase in temperature accelerates reaction rates as it increases the kinetic energy of the molecules, leading to more frequent and effective collisions.
Concentration of Reactants: Higher concentrations of reactants lead to more collisions and thus can increase the reaction rate.
Surface Area: In reactions involving solids, increasing the surface area (e.g., by grinding) allows more particles to be exposed for reaction, increasing the rate.
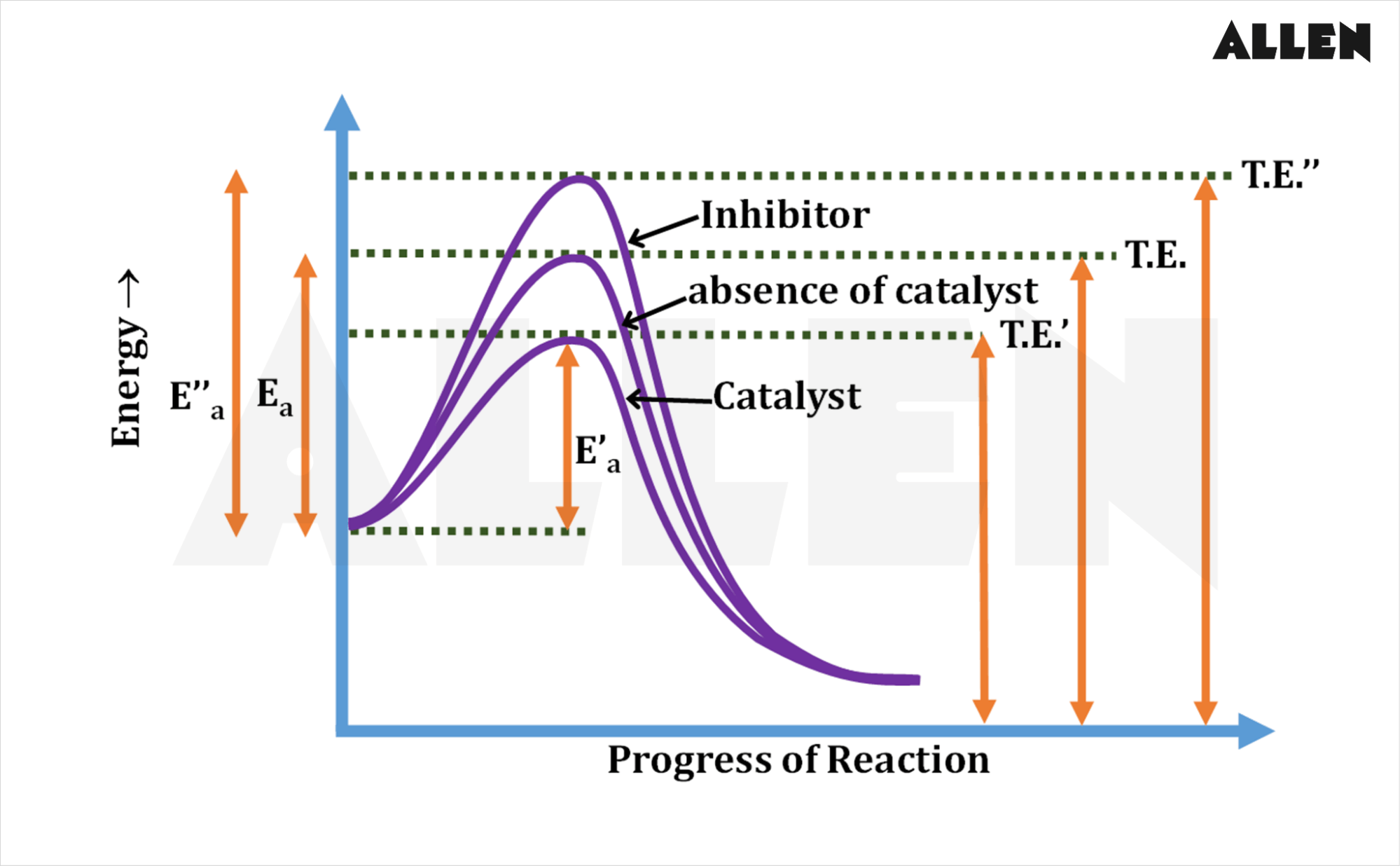
Presence of Catalysts: Catalysts increase reaction rates without being consumed in the reaction by lowering the activation energy barrier, facilitating easier collisions between reactants.
Le Chateliers- Principle states that if an external change is applied to a system at equilibrium, the system will adjust itself in such a way as to counteract the change and restore a new equilibrium.
Changes in Concentration:
When the concentration of a reactant or product is increased, the system will shift in the direction that consumes that substance to restore equilibrium. For example, if the concentration of products increases in the reaction A + B ⇌ C + D, the equilibrium will shift to the left to reduce the concentration of C and D.
Conversely, if the concentration of a reactant is decreased, the system will shift to the right to produce more of that reactant.
Changes in Pressure:
For gaseous reactions, changes in pressure can affect the equilibrium position. Increasing pressure will shift the equilibrium towards the side with fewer moles of gas, as this will reduce the pressure. For instance, in the reaction 2A(g) + B(g) ⇌ C(g), if the system is compressed, the reaction will favor the formation of products (C) if it has fewer moles of gas.
Reducing pressure will favor the side with more moles of gas.
Changes in Temperature:
Le Chatelier's Principle also applies to temperature changes. Increasing the temperature will favor the endothermic direction of a reaction (the direction that absorbs heat) while decreasing the temperature will favor the exothermic direction (the direction that releases heat). For instance, in an exothermic reaction, raising the temperature will shift the equilibrium to the left, favoring reactants, and lowering it will favor the products.
Changes in Volume: Reducing the volume of a reaction mixture will favor the side of the equilibrium with fewer moles of gas, in line with Le Chatelier's Principle, while increasing the volume will favor the side with more moles of gas.
*When Volume lessens, side with less mols preferred, when volume mores, side with more mols is favored
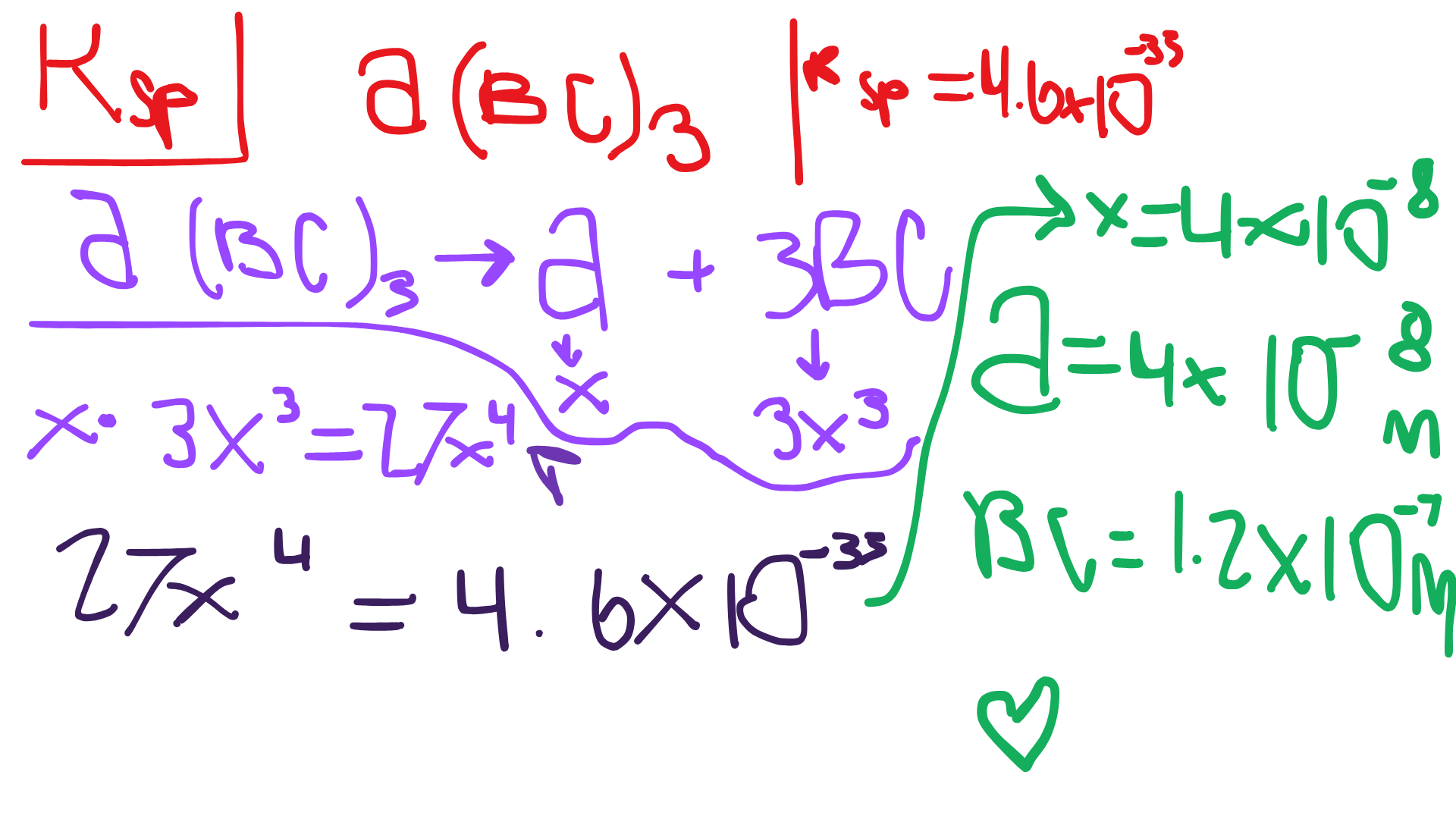
Enzymes = catalyst for biological processes
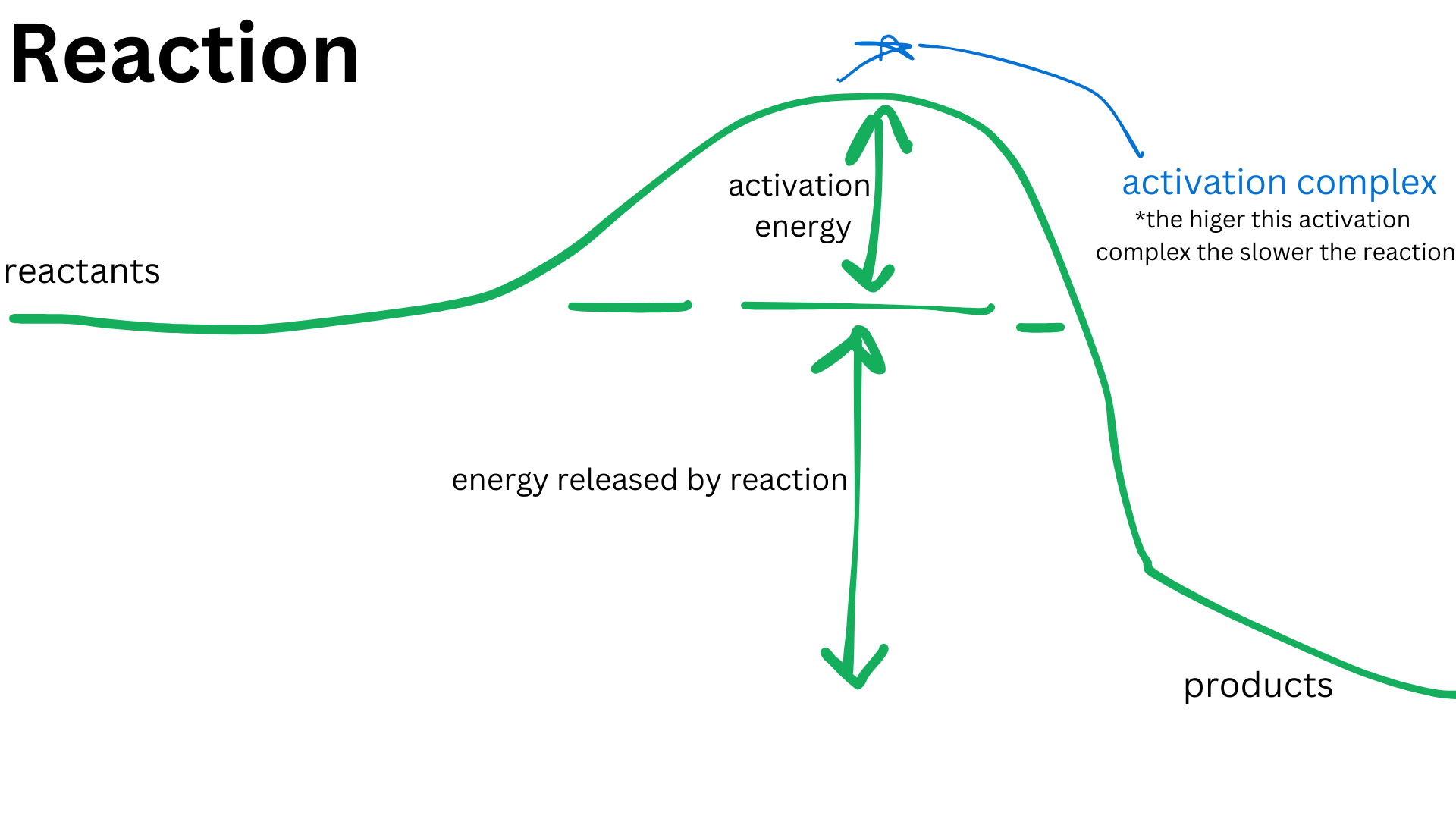
Activation Energy : The minimum energy required for a reaction to occur. This energy barrier must be overcome for reactants to transform into products, and it is crucial in determining the rate of a chemical reaction.
Activation complex : A temporary arrangement of atoms that forms during the transition state of a chemical reaction, which is characterized by a high energy level and is essential for the formation of products.
High activation energy = slow, low activation energy = fastreaction rates, impacting how quickly reactants are converted into products.