Rates of reaction
Variables that affect reaction rate
Temperature
Concentration
presence of a catalyst
surface area
pressure
What has to happen for stimulated particles to react
The reacting particles have to collide
They have to have activation energy(Ea) to have a successful collision
Collision theory
High Temperature
More collisions per second
The particles move faster therefore there are more collisions
High temp—> particles have more energy—> particles move faster—> more frequent collisions
More energy per particle
The average energy increases
High temp—> more energy—> more likely to have equal or greater energy than the Ea —> more frequent successful collisions
High surface area
More collisions per second
More particles are able to be collided with
High concentration
More collisions per second
There are more particles to collide with the other particles
High concentration—> more particles per unit of volume—> particles will travel shorter distances before colliding—>more frequent collisions
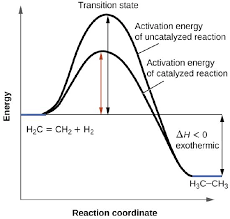
Catalyst
A substance used to increase the rate of reaction but is not used up in the reaction. it can often be recovered and reused
It does this by providing an alternate reaction pathway which requires less Ea.
As particles require less Ea to react the proportion of collisions that result in a successful reaction is higher.
More frequent successful collisions means a faster rate of reaction.