Renal system
In the process of regulating the solute content of plasma (and thereby the solute content of the extracellular fluid), the kidneys play a critical role in the maintenance of normal excitability of nerve and muscle tissues. Recall that the resting membrane potential depends on the electrochemical gradients for sodium and potassium ions across the cell membranes. The kidneys are necessary to maintain plasma levels of these ions. If these levels change, muscle and nerve tissue may become either hyperexcitable—causing muscle spasms or cramps, tingling sensations, or even cardiac arrhythmias—or hypoexcitable—causing numbness and weakness. The kidneys also function in acid-base balance by regulating the rate of hydrogen ion excretion and the plasma concentration of bicarbonate. We learned that the respiratory system regulates acid-base balance by regulating plasma levels of carbon dioxide. The kidneys and respiratory system work together to maintain a normal plasma pH of 7.4 by managing the ratio of bicarbonate to carbon dioxide to keep it at a ratio of 20:1. For the kidneys to carry out all these functions (and more), they require a great deal of ATP to carry out active transport. The kidneys are under hormonal control and neural control by the autonomic nervous system.
Functions of the Urinary System
The kidneys perform the following primary functions:
1. Regulation of plasma ionic composition. By increasing or decreasing the excretion of specific ions in the urine, the kidneys regulate the concentration of the following ions in the plasma: sodium (Na+), potassium (K+), calcium (Ca2+), magnesium (Mg2+), chloride (Cl-), bicarbonate (HCO3-),hydrogen (H+), and phosphates (HPO42- and H2PO4-).
2. Regulation of plasma volume and blood pressure. By controlling the rate at which water is excreted in the urine, the kidneys regulate plasma volume, which has a direct effect on total blood volume and, therefore, on blood pressure.
3. Regulation of plasma osmolarity. Because the kidneys vary the rate at which they excrete water relative to solutes, they have the ability to regulate the osmolarity (solute concentration) of the plasma.
4. Regulation of plasma hydrogen ion concentration (pH). By regulating the concentration of bicarbonate and hydrogen ions in the plasma, the kidneys partner with the lungs to regulate blood pH.
5. Removal of metabolic waste products and foreign substances from the plasma. Because the kidneys excrete wastes and other undesirable substances in the urine, they clear the plasma of waste products and eliminate them from the body. These materials include metabolic by-products such as urea and uric acid that are generated during protein and nucleic acid catabolism, respectively, as well as foreign substances such as food additives, drugs, or pesticides that enter the body from the external environment.
Because water and small solutes are freely exchanged between the plasma and interstitial fluid throughout most of the body, as the kidneys regulate the volume and composition of the plasma they likewise regulate the volume and composition of interstitial fluid. In addition,changes in the interstitial fluid affect the intracellular fluid. Thus the kidneys ultimately control the volume and composition of all the body’s fluids. As we will see, the ability of the kidneys to form urine, and thus to perform their primary functions, hinges on their ability to filter and process large quantities of solutes and water.
The kidneys perform several secondary functions as well. They are considered endocrine organs because they secrete the hormone erythropoietin (which stimulates erythrocyte production by the bone marrow) and the enzyme renin (which is necessary for the production of angiotensin II, a hormone important in regulating salt and water balance for long-term control of blood pressure). The kidneys are also necessary for the activation of vitamin D3 (ultimately to 1,25@(OH)2 vitamin D3), an important factor in regulating blood calcium and phosphate levels. Furthermore, the kidneys can function during periods of fasting to maintain a steady supply of plasma glucose by carrying out gluconeogenesis, the process by which molecules such as glycerol and certain amino acids are used to synthesize glucose
Structures of the Urinary System
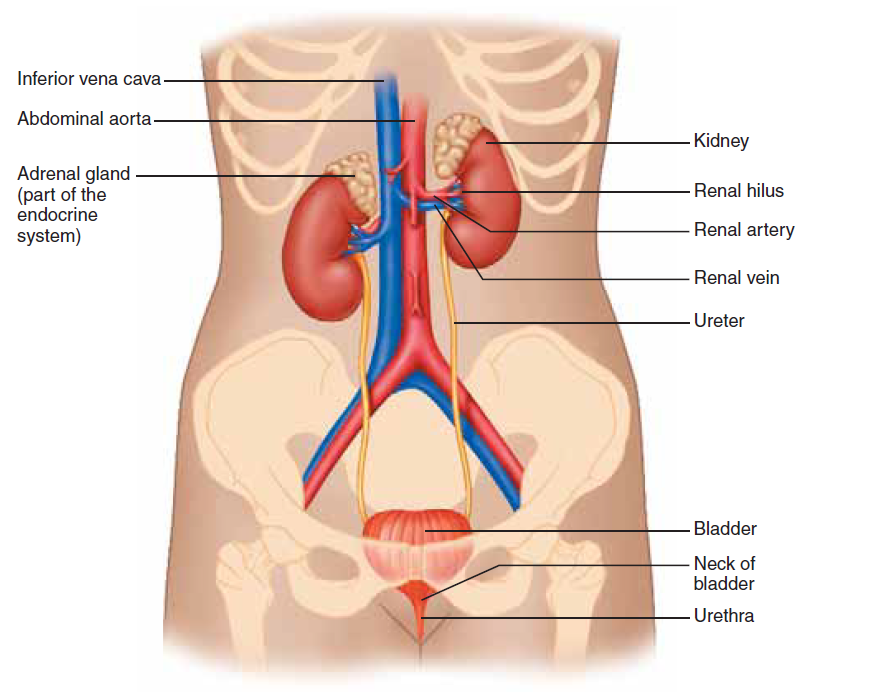
The urinary system consists of two kidneys, two ureters, the urinary bladder, and the urethra (Figure 18.1). Once formed by the kidneys, the urine is conducted to the bladder by the ureters. The bladderstores the urine until it is time to excrete it; at this time the urine moves through the urethra and out of the body. Sometimes, crystals in the kidneys form into stones, which also may pass down the ureter to the urethra. The kidneys are paired organs lying at the rear wall of the abdominal cavity just above the waistline, at about the level of the 12th rib. Each kidney is roughly bean shaped and is about the size of a fist. Although most abdominal organs are enclosed within the peritoneum, a clear membrane that lines the abdominal cavity, the kidneys are located between the peritoneum and the wall of the abdominal cavity. Thus they are described as retroperitoneal. The kidneys receive their blood supply from the renal arteries, which branch off the aorta and enter each kidney at a region called the renal hilus. Each kidney weighs only 115–170 grams (less than half a pound); their combined weight is less than 1% of the body weight of an average adult. Despite their small fraction of body weight, the kidneys receive about 20% of the cardiac output under normal resting conditions. This rich blood supply is crucial to the kidneys’ function not only because it provides them with oxygen and nutrients (kidneys account for 16% of total body ATP usage), but also because it enables the kidneys to remove (or clear) unneeded solutes and water from the blood at a rapid rate and eliminate them as urine. Meanwhile, the blood (minus these cleared materials) returns to the general circulation via the renal veins, which run parallel tothe renal arteries and drain into the inferior vena cava.
Macroscopic Anatomy of the Kidney
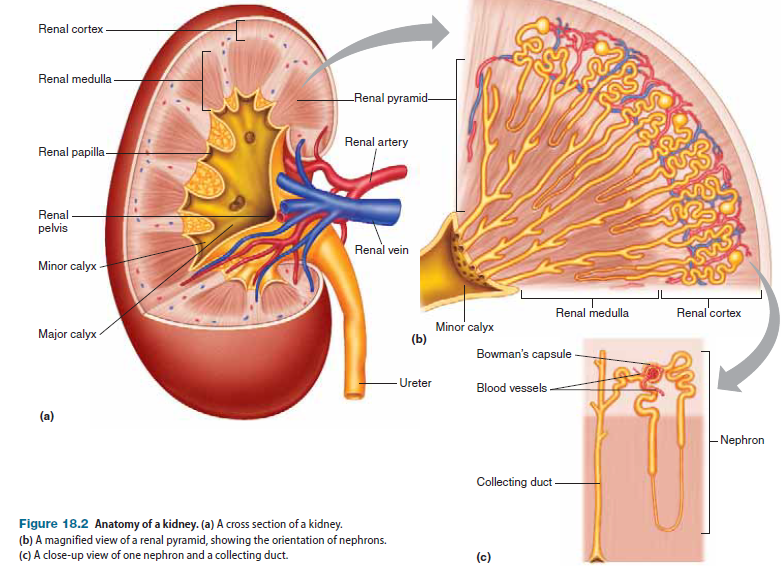
A cross section of a kidney reveals that it contains two major regions: a reddish-brown outer layer called inner region called the medulla, which is darker and has a striped appearance (Figure 18.2a). The medulla is subdivided into a number of conical sections called renal pyramids (Figure 18.2b). At the tips of the renal pyramids, in the areas known as papillae (singular: papilla), tubules called collecting ducts drain into common passageways called minor calyces (singular: calyx; see Figure 18.2a). The minor calyces converge to form two or three larger passageways called major calyces, which drain into a single funnel-shaped passage called the renal pelvis, the initial portion of the ureter. Within a kidney’s many renal pyramids are more than a million microscopic subunits called nephrons (Figure 18.2c), which are the functional units of the kidneys; they do the work of filtering the blood and forming the urine. The most obvious feature of the nephron is a long, coiled tube (called a renal tubule) that forms a hairpin loop about midway along its length. During the process of urine formation, fluid flows through the renal tubules, during which time the fluid’s composition is modified by exchange with the interstitial fluid.Fluid from individual tubules eventually drains into a set of common passageways called collecting ducts, where the composition of the fluid is further modified. The fluid that exits the collecting ducts and flows through the renal pelvis to the ureters is called urine.
Microscopic Anatomy of the Kidney
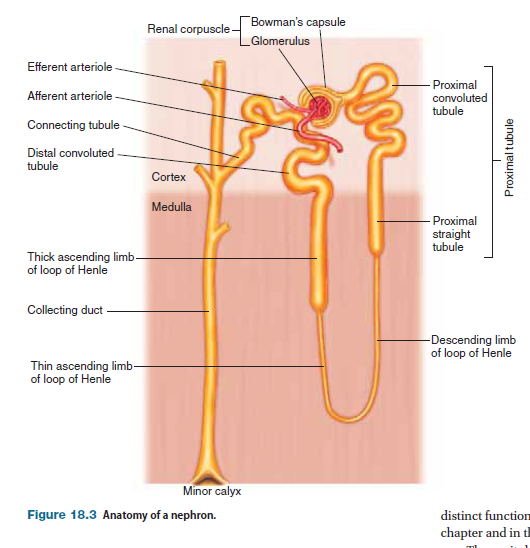
Each nephron is, in essence, a complete, self-contained “minikidney” that filters blood and forms urine. An individual nephron is composed of two parts: a renal corpuscle that filters the blood, and a renal tubule through which the filtrate travels and becomes modifiedto form urine.
Renal Corpuscle
A renal corpuscle consists of two parts: a spherical structure at the inflow end of the renal tubules called Bowman’s capsule, and a tuft of capillaries called the glomerulus. The renal corpuscle is the sitewhere blood is filtered and where tubular fluid, or filtrate, has its origin. Before the blood is filtered, it enters the glomerular capillaries via an afferent arteriole. As the blood flows through the glomerular capillaries, protein-free plasma passes through the walls of the capillaries into Bowman’s capsule by a process called glomerular filtration. The remaining blood leaves the glomerulus via an efferent arteriole. This arrangement of two arterioles in series with a capillary bed between them is unique to the renal corpuscle and allows greater regulation of glomerular filtration. The walls of the afferent and efferent arterioles contain smooth muscle that can contract or relax in response to input from paracrines and the sympathetic nervous system, thereby regulating their diameter and thus glomerular filtration.
Renal Tubule
As the glomerular filtrate is formed, it flows from Bowman’s capsule to the initial portion of the renal tubule, called the proximal convoluted tubule because of both its proximity to the capsule and its highly folded or convoluted structure; the fluid then moves to the proximal straight tubule. The two tubules together are called the proximal tubule.The proximal tubule empties into the loop of Henle, the portion of the tubule that makes up the hairpin loop of the nephron. The loop of Henle is divided into three sections: (1) the descending limb, (2) the thin ascending limb, and (3) the thick ascending limb. The descending limb is a thin tubule leading from the proximal tubule and extending into the renal medulla. At the tip of the loop, the tubule reverses direction, becoming the thin ascending limb, which extends toward the cortex. As the tubule approaches the cortex, it widens into the thick ascending limb. From the ascending limb of the loop of Henle, the fluid flows into the distal convoluted tubule, which resembles the proximal tubule in appearance but is considerably shorter. It next enters a short, straight terminal portion of the nephron, called the connecting tubule, which joins the nephron with the collecting duct. Several tubules empty their fluid into a single collecting duct. The collecting ducts then empty into the minor calyces, as previously described.
Cortical and Juxtamedullary Nephrons
Two classes of nephrons are distinguished based on their location: cortical nephrons and juxtamedullary nephrons.
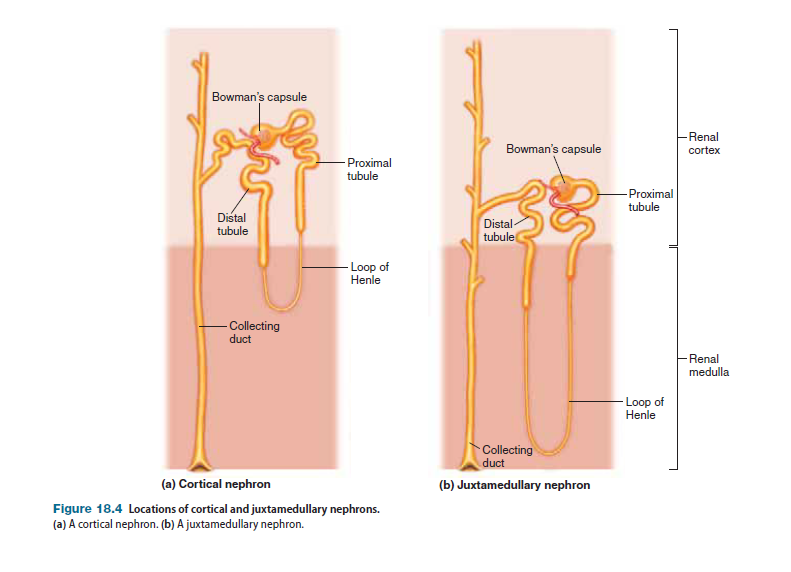
The vast majority of nephrons in the kidneys are cortical nephrons, which are located almost entirely within the renal cortex; only the tip of the loop of Henle dips into the renal medulla. In juxtamedullary nephrons, which constitute approximately 15–20% of all nephrons, the renal corpuscle is located near (juxta) the border between the cortex and medulla. The glomerulus, proximal convoluted tubule, and distal convoluted tubule are located in the cortex, whereas the loop of Henle dips deep into the renal medulla. Although the two types of nephrons are fundamentally similar, an important functional difference characterizes them: Whereas both cortical and juxtamedullary nephrons function directly in the processes involved in urine formation, juxtamedullary nephrons also function in maintainingan osmotic gradient in the renal medulla that is crucial to the kidneys’ ability to produce highly concentrated urine and thus conserve water under certain conditions.
The Juxtaglomerular Apparatus
At a site where the initial portion of the distal tubule comes into contact with a nephron’s afferent and efferent arterioles is a structure called the juxtaglomerular apparatus.
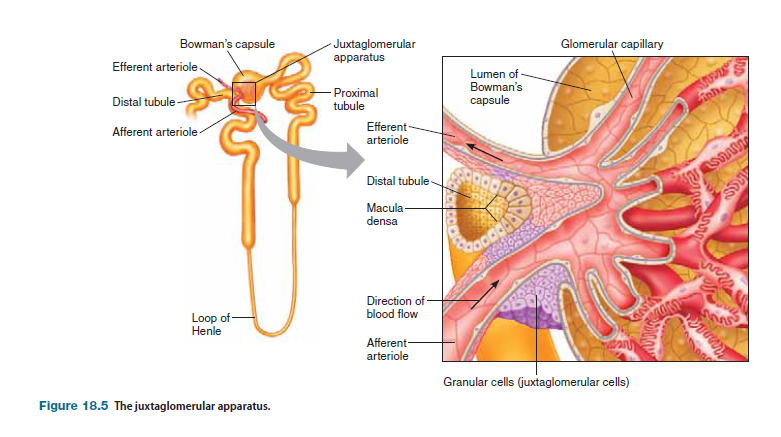
The juxtaglomerular apparatus has two components: (1) a specialized cluster of the tubule’s epithelial cells, called the macula densa, and (2) granular cells (or juxtaglomerular cells) in the wall of the afferent (and to a lesser extent the efferent) arterioles. Granular cells are named based on the presence of numerous cytoplasmic secretory granules that contain a product called renin. The juxtaglomerular apparatus plays an important role in regulating blood volume and blood pressure
Blood Supply to the Kidney
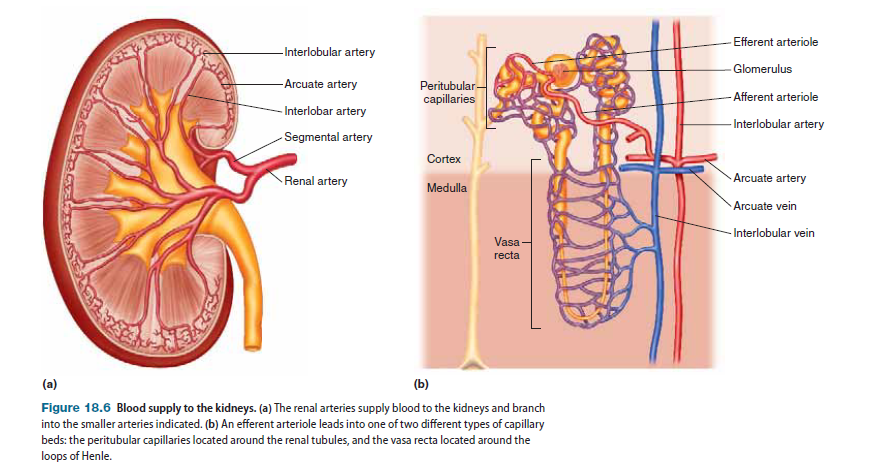
Within the kidney, the renal artery branches into segmental arteries, which in turn branch into a number of smaller interlobar arteries, which then feed into another set of arteries called arcuate arteries. The arcuate arteries branch into interlobular arteries, from which blood is carried to individual nephrons by the afferent arterioles, which next lead into the glomerular capillary beds. Coming off each of the glomerular capillary beds is the efferent arteriole, which then gives rise to one of two types of capillary beds (Figure 18.6b): (1) peritubular capillaries, which branch from the efferent arterioles of cortical nephrons and are located close to the renal tubules, and (2) vasa recta, which branch from the efferent arterioles of juxtamedullary nephrons and are networks of blood vessels forming hairpin loops that run along the loops of Henle and collecting ducts, dipping deep into the renal medulla. Each of these capillary beds has a distinct function in the formation of urine. The peritubular capillaries and vasa recta drain into the interlobular veins. From here, blood is carried away from nephrons by the arcuate veins, and then by the interlobar veins, which run parallel to their respective arterial counterparts, eventually draining into the renal vein.
Basic Renal Exchange Processes
In the kidneys, water and solutes are exchanged between plasma and fluid in the renal tubules to regulate the composition of plasma. Substances ultimately removed from the plasma are excreted in urine. Three exchange processes occur within the renal nephrons:
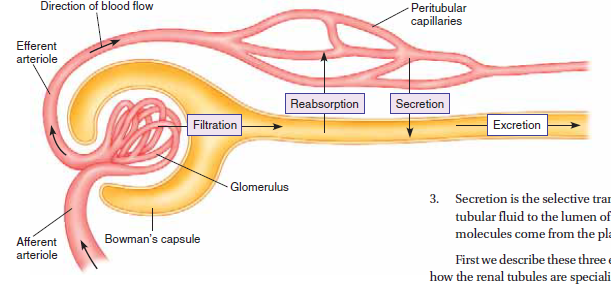
1. Glomerular filtration is the bulk flow of protein-free plasma from the glomerular capillaries into Bowman’s capsule.
2. Reabsorption is the selective transport of molecules from the lumen of the renal tubules to the interstitial fluid outside the tubules. Reabsorbed molecules eventually enter the peritubular capillaries by diffusion, and are then returned to the general circulation.
3. Secretion is the selective transport of molecules from the peritubular fluid to the lumen of the renal tubules. These secreted molecules come from the plasma of the peritubular capillaries. First we describe these three exchange processes; then we look at how the renal tubules are specialized for reabsorption and secretion.
Glomerular Filtration
Filtration at the renal corpuscle is driven by Starling forces (hydrostatic and osmotic pressure gradients) existing across the walls of glomerular capillaries. These Starling forces are the same forces that drive the filtration of fluid from capillaries throughout the body. The filtrate resembles plasma in composition, exceptthat it lacks most of the proteins found in plasma.
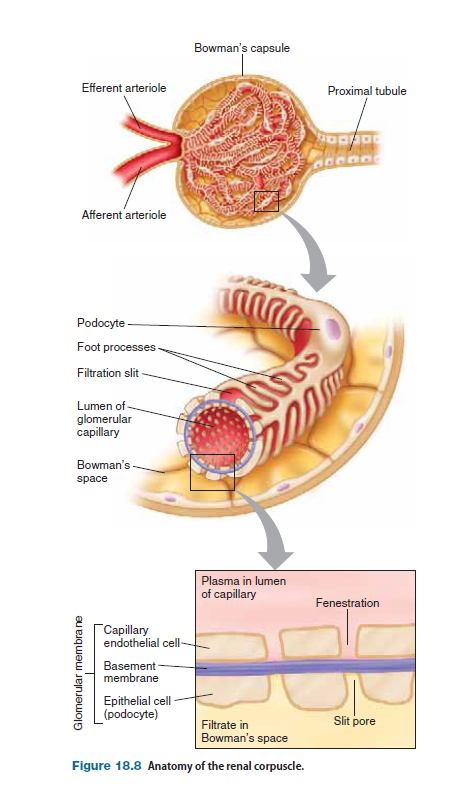
The wall of Bowman’s capsule and the wall of the renal tubule are composed of a continuous layer of epithelial cells. In Bowman’s capsule, this epithelium folds upon itself to envelop the glomerular capillaries. Below the epithelium is a basement membrane that acts as the primary filtration barrier for proteins. The glomerular filtrate must cross three barriers to enter Bowman’s capsule: (1) the capillary endothelial cell layer, (2) the surrounding epithelial cell layer, and (3) the basement membrane that is sandwiched between them. The combination of these three layers makes up what is called the glomerular membrane or filtration barrier. The epithelial cells that cover the glomerular capillaries have special extensions or foot processes, giving them their name podocytes. As fluid moves out of the glomerular capillaries, it passes through gaps between the podocytes, called slit pores. The sizes of slit pores are regulated by slit diaphragms. The presence of fenestrations (pores) in the capillary endothelium, the large number of slit pores in the surrounding capsule epithelium, and the large surface area of the filtration barrier combine to make the renal corpuscle favorable for the bulk flow of protein-free fluid between blood and the lumen of Bowman’s capsule (called Bowman’s space). The sum of the Starling forces in the renal corpuscle is called the glomerular filtration pressure, which is analogous to the netfiltration pressure. Four Starling forces play key roles in glomerular filtration.
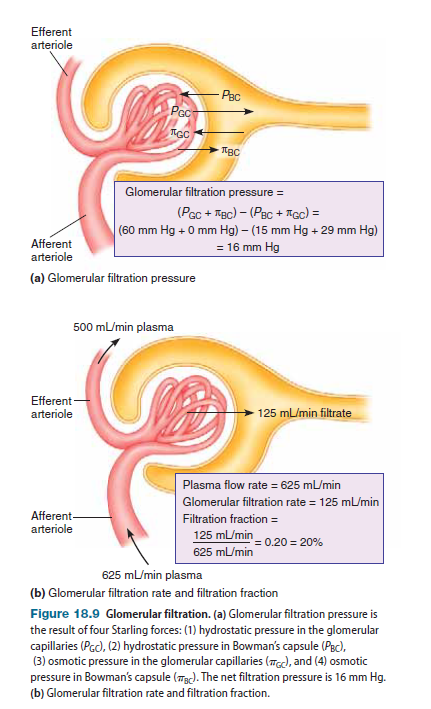
Glomerular capillary hydrostatic pressure. The glomerular capillary hydrostatic pressure (PGC) favors filtration and is equal to the blood pressure in the glomerular capillaries— approximately 60 mm Hg. This pressure is substantially higher than the hydrostatic pressure in most other capillaries because of the high resistance of the efferent arteriole, which is located downstream from the glomerular capillaries. As a general rule, the presence of high resistance in any network of vessels tends to raise the pressure in vessels located upstream while lowering the pressure downstream, just as tightening a clamp on a water hose causes the pressure downstream to fall (and reduces the flow of water) but increases the pressure upstream because of the “backup” of water.
2. Bowman’s capsule osmotic pressure. The osmotic pressure in Bowman’s capsule (pBC) favors filtration. Recall that osmotic pressure of a fluid is created by the presence of nonpermeant solutes. Because proteins are generally the only solute that cannot move between plasma and Bowman’s capsule, these molecules generate the osmotic force. The presence of proteins in the interstitial fluid surrounding the glomerulus tends to pull fluid out of the capillaries and into the capsule. Because very little protein leaves the capillaries with the filtrate, the protein concentration in Bowman’s capsule is very small, and thus the osmotic pressure is negligible under normal conditions. (In certain diseases that cause damage to the glomerulus, however, significant quantities of protein can leak out of glomerular capillaries, creating a significant osmotic force for filtration.) The net pressure favoring filtration at the renal corpuscle under normal conditions is PGC + pBC = 60 mm Hg + 0 mm Hg = 60 mm Hg
3. Bowman’s capsule hydrostatic pressure. Bowman’s capsule hydrostatic pressure (PBC) opposes filtration and is typically about 15 mm Hg. This pressure is considerably higher than the hydrostatic pressure in the interstitial fluid surrounding most capillary beds because the relatively large volume of fluid that filters out of the glomerular capillaries is “funneled” into the restricted space of Bowman’s capsule.
4. Glomerular osmotic pressure. The glomerular osmotic pressure (pGC) opposes filtration. The glomerular osmotic pressure opposes filtration because the presence of proteins in the plasma tends to draw filtrate back into the glomerulus. The osmotic pressure in the glomerulus is approximately 29mm Hg, which is higher than the typical 25 mm Hg osmotic pressure found in most systemic capillaries, because the blood that flows through these capillaries loses a substantial fraction of its water as a result of glomerular filtration, and this loss of water causes the concentration of plasma proteins to increase. The net pressure opposing filtration at the renal corpuscle under normal conditions is PBC + pGC = 15 mm Hg + 29 mm Hg = 44 mm Hg
Glomerular Filtration Rate
The average net glomerular filtration pressure (GFP) is GFP = (PGC + pBC) - (PBC + pGC). Substituting the values for the Starling forces into this equation gives us the glomerular filtration pressure:
(60 mm Hg + 0 mm Hg) - (15 mm Hg + 29 mm Hg) = 16 mm Hg
Under normal conditions, approximately 625 mL of plasma flows through the kidneys each minute. The volume of the plasma filtered per unit time is called the glomerular filtration rate (GFR), and it is approximately 125 mL/min. Thus, over the course of a day, the kidneys filter 180 liters of plasma! This is an enormous volume of fluid, considering that the body’s total plasma volume is only approximately 2.75 liters for an average adult. In fact, the GFR is so high that a volume of fluid equivalent to total plasma volume filters through the glomeruli every 22 minutes.
Filtration Fraction
The fraction of the renal plasma volume that is filtered is called the filtration fraction. It is equal to the glomerular filtration rate divided by the renal plasma flow rate:
Filtration fraction =GFR/renal plasma flow=125 mL/min/625 mL>min = 0.20 = 20%
Thus 20% of the plasma that flows through the kidneys is filtered into Bowman’s capsule.
Filtered Load
The quantity of a particular solute that is filtered per unit time is known as the filtered load. When molecules of a solute are small enough to move across the glomerular membrane without significant restriction, as most solutes are, that solute is said to be freely filterable. When such a substance is filtered, its concentration in the glomerular filtrate is virtually identical to its concentration in the plasma. Consequently, the filtered load, usually expressed in mole/min or the equivalent, equals the product of the GFR and the solute’s plasma concentration (which is designated by the symbol P):
Filtered load = GFR * plasma concentration of X
= GFR * PX
As an example, let’s determine the filtered load of glucose. We already know that the normal GFR is 125 mL/min. Given that the normal plasma glucose concentration is 100 mg/dL = 1 mg/mL, then the filtered load of glucose under normal conditions is
Filtered load = 125 mL/min * 1 mg/mL = 125 mg/min
Regulation of Glomerular Filtration Rate
Although 180 liters of fluid filters into the renal tubules every day, only about 1.5 liters of urine is normally excreted during the same period of time. The reason for this low excretion rate is that more than 99% of the fluid filtered out of the plasma is normally reabsorbed. Because the GFR is so large, however, even a small percentage change in its value will have an enormous effect on the volume of fluid filtered and, therefore, on the quantity of material that must be reabsorbed to maintain the same urinary output.A 10% increase in GFR, for example, would translate into an extra 18 liters of fluid entering the kidney tubules per day. Under most circumstances, however, GFR remains relatively constant because of the intrinsic and extrinsic mechanisms.
Intrinsic Control of Glomerular Filtration Changes in mean arterial pressure (MAP) can potentially alter the GFR because the arterial pressure affects the glomerular capillary pressure, which in turn influences the glomerular filtration pressure. Although mean arterial pressure is regulated to stay constant by baroreceptor reflexes and other mechanisms, it does change in certain situations, such as during exercise. When arterial pressure increases, glomerular capillary pressure also tends to rise, which increases the glomerular filtration pressure and hence GFR. Conversely, when mean arterial pressure falls, the glomerular capillary pressure also tends to fall, which decreases the glomerular filtration pressure and GFR. Such changes in GFR are undesirable because they tend to make urine flow increase or decrease, respectively, which interferes with the kidneys’ ability to regulate the volume and composition of the plasma.
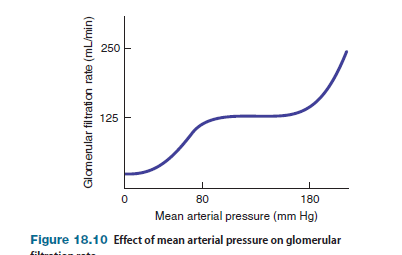
Although variations in mean arterial pressure pose a potential problem, the kidneys can tolerate a change in mean arterial pressure over a fairly wide range (approximately 80–180 mm Hg) with very little change in GFR, because three intrinsic mechanisms regulate the GFR in the face of changes in arterial pressure.
Two of these intrinsic mechanisms—myogenic regulation of afferent arteriolar smooth muscle and tubuloglomerular feedback—operate by changing the resistance of the afferent arteriole; the third factor, mesangial cell contraction, acts by changing the permeability of the filtration barrier.
Myogenic regulation of the GFR (Figure 18.11a) is similar to the myogenic regulation of blood flow that occurs in other parts of the body. The smooth muscle of the afferent arteriole is sensitive to stretch and responds to this condition by contracting. When mean arterial pressure rises, pressure in the afferent arteriole also rises, which causes its wall to stretch. Pressure also rises in the glomerular capillaries, which raises the glomerular filtration pressure and the GFR. However, in response to stretching, the afferent arteriole constricts, which increases its resistance to blood flow. As a result, the pressure in blood vessels downstream, including the glomerular capillaries, decreases. This decrease in pressure counteracts (but does not change) the initial rise in pressure that triggered constriction of the afferent arteriole. Thus, through this negative feedback mechanism, the glomerular capillary pressure, and hence the GFR, tends to stay nearly constant. As expected, a fall in meanarterial pressure has the opposite effects, triggering relaxation of the afferent arteriole and a subsequent rise in glomerular capillary pressure that counteracts the initial fall. The smooth muscle of the afferent arteriole is sensitive not only to stretch but also to chemical agents that are secreted by cells of the macula densa, which are located in the nearby distal tubule. In tubuloglomerular feedback, a change in GFR causes a change in the flow of tubular fluid past the macula densa, which alters the secretion of certain (as yet unidentified) paracrines from the macula densa. These paracrines then trigger contraction or relaxation of the afferent arteriole, which causes a change in glomerular capillary pressure and GFR in the direction opposite to that of the original change. As a consequence of this negative feedback control of GFR, the flow of fluid past the macula densa changes such that it opposes the change in flow that triggered the response initially. For example, if an increase in GFR causes the flow of tubular fluid to increase, then the afferent arteriole constricts and the GFR decreases, thereby decreasing the flow. Thus the resistance of the afferent arteriole varies in such a way that the flow of fluid past the macula densa tends to be held constant. Figure 18.11b shows what happens when an increase in blood pressure causes an increase in GFR. The third mechanism of GFR autoregulation is similar to myogenic regulation, except that it targets mesangial cells instead of blood vessels. Mesangial cells are modified smooth muscle cells located around glomerular capillaries that function much like precapillary sphincters. A contraction of the mesangial cells will decrease blood flow through some glomerular capillaries, thereby decreasing the surface area over which filtration can occur. The contractile state of mesangial cells depends on the pressure within the capillaries. An increase in blood pressure, which increases GFR, stretches mesangial cells. In response to the stretch, the mesangial cells contract, decreasing the surface area of capillaries available for filtration, which in turn decreases the GFR until it reaches the normal rate.
Extrinsic Control of Glomerular Filtration and Renal Blood Flow We just learned that glomerular filtration is regulated by intrinsic control mechanisms when mean arterial pressure varies between 80 and 180 mm Hg. However, when mean arterial pressure goes above or below this range, the GFR rises or falls, respectively, because intrinsic mechanisms are no longer able to prevent the glomerular capillary pressure from changing.
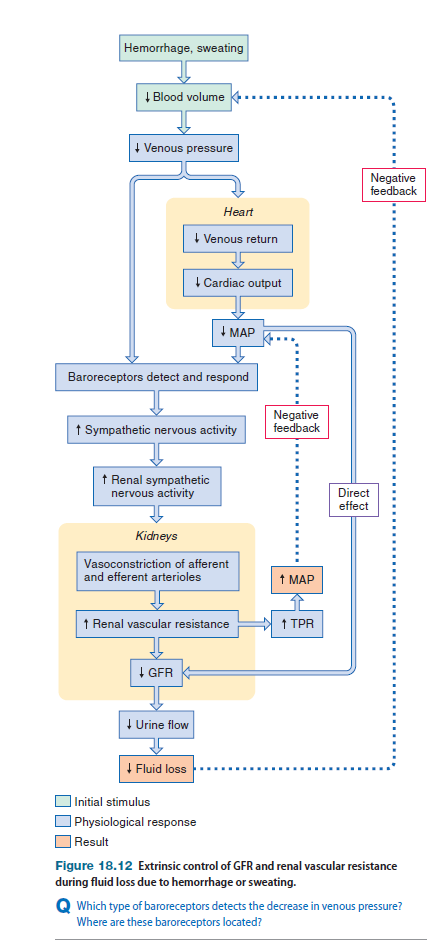
shows what happens when the mean arterial pressure falls due to hemorrhage or excessive sweating. When MAP falls to less than 80 mm Hg, GFR decreases directly due to the lowered filtration pressure. In addition, the fall in MAP triggers an increase in sympathetic nervous activity via baroreceptor reflexes. In response to increased input from sympathetic nerves, smooth muscle in the afferent and efferent arterioles contracts, and both afferent and efferent arterioles constrict, which increases the overall resistance of the renal vasculature and decreases the GFR. The increase in renal vascular resistance acts to decrease renal blood flow and also raises the total peripheral resistance, which in turn increases MAP. The decrease in GFR also decreases urine output, which helps the body conserve water. This process minimizes reductions in blood volume, which in turn counteracts further decreases in arterial pressure.
Reabsorption
Reabsorption refers to movement of filtered solutes and water from the lumen of the tubules back into the plasma. If solutes and water were not reabsorbed, all filtered materials would be excreted, and it would take just eight minutes for a person to lose a liter of fluid in the urine (assuming that the GFR stays constant at its normal rate). Given that 1 liter of fluid is equivalent to 20% of total blood volume, the ability of the kidneys to reabsorb filtered solutes and water is an absolute necessity of life. In fact, 100% of many substances filtered at the glomerulus are reabsorbed by the renal tubules. The reabsorption of other substances is regulated to vary their excretion rate, which in turn regulates the concentration of these substances in the plasma. In this section, we first look at general properties of solute reabsorption; we then describe how water reabsorption across the tubular epithelium is coupled to solute reabsorption.
Solute and Water Reabsorption
Table 18.1 lists the quantities of several freely filtered substances that are filtered and reabsorbed in a single day under normal circumstances. The sheer mass of material that must be transported from tubular fluid back into the plasma by the renal tubule epithelium every day reveals how extensive the reabsorption process is. Many solutes are reabsorbed actively; that is, they are transported against their electrochemical gradients as they move from the tubular lumen to the plasma. Reabsorption of most solutes occurs in the proximal and distal convoluted tubules. Recall that the efferent arteriole branches into peritubular capillary beds adjacent to the renal tubules in the renal cortex. The interstitial fluid between the epithelium of the renal tubules and the peritubular capillaries (peritubular fluid) fills the peritubular space. When a substance is reabsorbed, it must move across twobarriers: the tubule epithelium and the capillary endothelium.
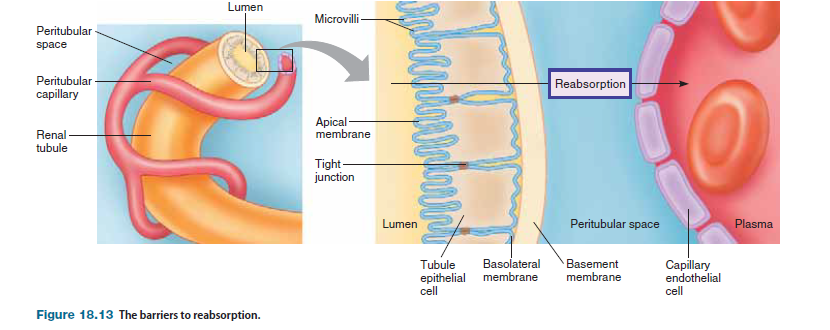
Capillaries are a barrier only to movement of macromolecules, such as proteins, and cells. Thus the epithelial cells lining the renal tubules form the primary barrier to reabsorption of the various solutes. Because tight junctions connect the epithelial cells lining the renal tubules, movement of molecules between cells is restricted. The plasma membrane of the epithelial cells facing the tubule lumen is called the apical membrane and has microvilli; the plasma membrane facing the interstitial fluid is called the basolateral membrane. Microvilli on the apical membrane are abundant in the more proximal portions of the tubule but are sparse in the distal portions. Beneath the basolateral membrane is a basement membrane that does not contribute significantly as a barrier. Some molecules are passively reabsorbed, whereas others require energy.
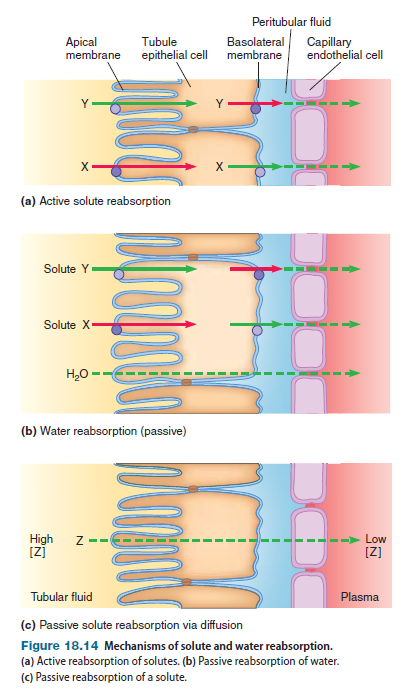
depicts the reabsorption of three uncharged solutes (X, Y, and Z) and water. (Note that the movement of charged substances is affected by electrical forces in addition to the forces described next.) Although each substance moves in the same direction, the actual mechanism of reabsorption differs.
Active Reabsorption of Solute In the examples shown in Figure 18.14a, substances X and Y are both actively transported, but through different mechanisms. Both substances are transported by mechanisms involving active transport across one membrane of the tubule in conjunction with passive movement across the other membrane. Recall that active transport requires energy to move a molecule against its electrochemical gradient. This energy can come either directly from ATP (primary active transport) or from an ion electrochemical gradient created at the expense of ATP (secondary active transport). Active transporters for substance X located on the apical membrane of the tubule epithelial cell transport X into the cell, creating a high intracellular concentration of X (Figure 18.14a). Carrier proteins for X are located on the basolateral membrane. As the concentration of X inside the cell rises due to active transport, this solute moves out of the cell into the peritubular fluid by facilitated diffusion; X then diffuses into the plasma. Active transporters for substance Y located on the basolateral membrane of the tubule epithelial cell transport Y out of the cell and into the peritubular fluid; Y then diffuses into the plasma (Figure 18.14a). This process keeps the concentration of Y inside the tubule epithelial cell low. Carrier proteins for Y are located on the apical membrane. Because the concentration of Y inside the cell is low, this solute moves into the cell by facilitated diffusion. Therefore, the net movement of Y is from the tubular fluid to the plasma.
Water Reabsorption Water diffusion is based on differences in osmolarity. As solutes such as X and Y are actively reabsorbed, they increase the osmolarity of the plasma while decreasing the osmolarity of the tubular fluid. Therefore, water diffuses down its concentration gradient to a region of greater osmolarity can diffuse into the plasma. (Figure 18.14b). Stated another way, water reabsorption follows the active reabsorption of solute. For water to follow solute, water must be able to permeate the tubule epithelium. Although water can permeate most plasma membranes in the body, we will see that some plasma membranes in the renal tubules are impermeable to water.
Passive Reabsorption of Solute Substance Z is passively reabsorbed via diffusion (Figure 18.14c). For this process to occur, two conditions must be satisfied: The concentration of Z must be greater in the tubular fluid than in the plasma, and Z must be able to permeate the plasma membranes of the tubule epithelium and the capillary endothelium. The tubular fluid, which came from plasma, has a higher concentration of Z than fluid in the capillary lumen, because most water that is filtered is reabsorbed as described earlier. As water leaves the lumen of the tubules and enters the plasma, the tubular concentration of Z increases, whereas the plasma concentration of this solute decreases. Therefore, Z diffuses down its concentration gradient from the tubular fluid to the plasma. Stated another way, reabsorption of molecules that can permeate membranes follows water reabsorption.
Transport Maximum
When solutes are transported from filtrate to plasma across the tubular epithelium by carrier proteins or pumps, those modes of transport can become saturated; that is, when solute concentration is high enough, all carrier proteins and pumps are occupied, and the system is operating at transport maximum (Tm). The transport maximum is best understood for substances that are normally 100% reabsorbed in the renal tubules, such that none of the substance is excreted in the urine. When the plasma concentration of the solute increases, however, more solute filtered is at the glomerulus, causing an increase in the concentration of solute in the filtrate. At a certain plasma concentration of the solute, the corresponding filtrate concentration saturates the carrier proteins in the tubules, and some of the substance appears in the urine. The plasma concentration of solute at which “spillover” into the urine occurs is called the renal threshold. As the plasma concentration rises above the renal threshold, the rate at which the solute is excreted in the urine becomes progressively higher. A well-known example of transport maximum occurs in the active reabsorption of glucose.
Glucose, which is freely filtered from the glomerulus, is normally completely reabsorbed by active transport in the proximal tubule, so no glucose is excreted in the urine.
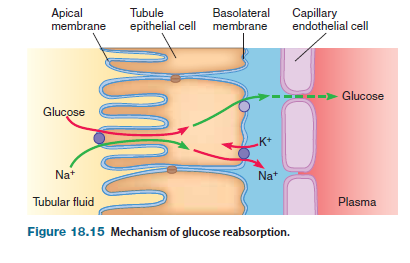
Glucose is actively transported across the apical membrane by a sodium-linked active transport; that is, glucose is cotransported with sodium ions from the tubular fluid into the epithelial cell. Such cotransport concentrates glucose inside the epithelial cell. A carrier protein for glucose is located on the basolateral membrane. Because glucose is found in high concentration inside the epithelial cell, this solute is transported by this carrier down its concentration gradient into the peritubular fluid, where it can diffuse into the plasma.
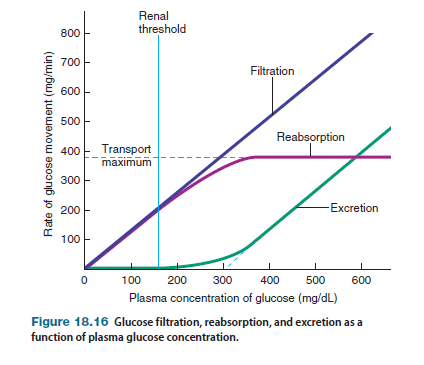
Figure 18.16 shows the relationship between the plasma concentration of glucose and the handling of glucose by the kidneys. The transport maximum for glucose reabsorption is 375 mg/min. The normal plasma glucose level is 80–100 mg/dL. Given that GFR is 125 mL/min (=1.25 dL>min), the filtered load of glucose when its plasma concentration is 100 mg/dL is
GFR Pglucose = 1.25 dL/min 100 mg/dL = 125 mg/min
which is well below the transport maximum of 375 mg/min. Therefore, all the glucose is reabsorbed, and none is excreted in the urine. However, if glucose levels increase, eventually the amount of glucose in the filtrate will exceed the capacity for reabsorption, and some glucose will be excreted in the urine. The “theoretical” renal threshold for glucose—the plasma concentration at which the amount of glucose in the filtrate exceeds the transport maximum, and at which glucose appears in the urine can be calculated as follows:
GFR * renal threshold = transport maximum
renal threshold = transport maximum/GFR
renal threshold = 375 mg/min/1.25 dL>min = 300 mg>dL
However, the true renal threshold for glucose is 160–180 mg/dL. At these plasma levels of glucose, the filtered load of glucose is approximately 225 mg/min. Although this value is considerably lower than the transport maximum for glucose, some glucose molecules in the filtrate avoid contact with carrier proteins and, therefore, are excreted in the urine even though the carrier proteins are not 100% saturated. (Notice the nonlinear relationship between glucose reabsorption and excretion rates and plasma concentration of glucose as the rates approach the maximum in Figure 18.16.)
In untreated diabetes mellitus, plasma glucose levels are elevated, a condition called hyperglycemia, and may be several times the normal value. When the concentration of glucose exceeds the renal threshold, glucose appears in the urine. Before modern methods for detecting glucose in urine were invented, a common test for diabetes mellitus was to taste the urine for sweetness. Elimination of glucose in the urine also produces diuresis (increased water loss or urine volume). Recall that water moves across epithelium by osmosis, and generally water movement follows active transport of solute. Normally the actively reabsorbed glucose would be followed by passive reabsorption of water. However, when glucose is not reabsorbed but rather remains in the tubular fluid, it exerts an osmotic force that causes water to stay in the renal tubules and ultimately be eliminated from the body in urine. As a consequence, early symptoms of diabetes include excessive thirst and urination. As well as suffering the acute effects of diabetes on urine function, 20–30% of people with diabetes develop renal disease. The high plasma glucose levels of diabetics, working through mechanisms as yet unknown, damage the nephrons, inducing diabetic nephropathy. Over time, the disease may eventually progress to chronic renal failure and end-stage renal disease.
Secretion
In tubular secretion, molecules move from the plasma of peritubular capillaries into the renal tubules to become part of the filtrate. Secretion follows the same basic processes as reabsorption and involves the same barriers, except that movement goes in the reverse direction. Some substances diffuse from plasma into the filtrate, whereas others are actively transported. Secretion by active transport requires either that proteins in the basolateral membrane actively transport the solute from interstitial fluid to inside the epithelial cell, or that proteins in the apical membrane actively transport the solute from inside the epithelial cell into the filtrate. Among the substances actively secreted by the renal tubules are ions, such as potassium ions and hydrogen ions; waste products, such as choline and creatinine; and foreign substances, such as the antibiotic penicillin. The end result of secretion is an increase in the quantity of solute excreted in the urine, which decreases the solute’s plasma concentration.
Regional Specialization of the Renal Tubules
Because the properties of the tubule epithelium vary from region to region along the length of the tubule, both the substances transported and the mechanisms of transport differ in different regions of the tubule (Table 18.2). In the following sections, we look at transport in different regions of the renal tubules.
Nonregulated Reabsorption in the Proximal Tubule
In the proximal tubule, mechanisms of reabsorption are so efficient that 70% of the sodium and water that are filtered is reabsorbed by the time tubular fluid reaches the beginning of the loop of Henle. Solute and water reabsorption are coupled such that the process is iso-osmotic; that is, no change in solute concentration occurs ineither the plasma or the filtrate. Some solutes (including glucose) are virtually 100% reabsorbed by this time. For this reason, the proximal tubule is said to function as a mass absorber whose function is to reabsorb the bulk of filtered solutes and water, thereby preventing their loss from the body. This reabsorption of solutes in the proximal tubule is an ongoing process that generally is not regulated. The epithelium of the proximal tubule has a number of features that facilitate mass absorption (Figure 18.17a). First, the apical membrane is highly folded into many microvilli, a conformation known as a brush border; such folding increases the total surface area of the apical membrane, which facilitates transport. Second, transporting cells possess large numbers of mitochondria, which supply the large quantity of ATP necessary to drive active transport. Finally, the tight junctions between epithelial cells have a relatively high permeability to small solutes and water, making this a “leaky” epithelium. This high permeability at the tight junctions facilitates the diffusion of solutes and water between cells (called paracellular transport), further enabling the epithelium to transport large quantities of materials.
Regulated Reabsorption and Secretion in the Distal Tubule and Collecting Duct
In contrast to the proximal tubule, the distal tubule and the collecting duct are specialized to allow regulation of reabsorption and secretion. In the epithelium of these tubules, the brush border is much less prominent than in the proximal tubule, or is even lacking altogether.
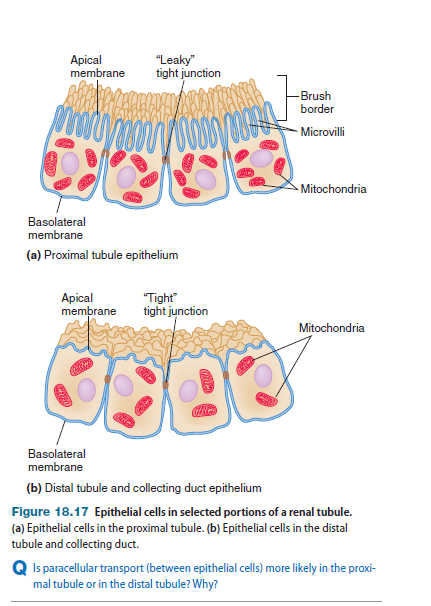
In addition, epithelial cells have fewer
mitochondria, and tight junctions are far less permeable, making
this an example of a “tight” epithelium. Furthermore, tubule epithelial
cells have receptors for hormones that regulate the transport of
water and several solutes, and water reabsorption does not always
follow solute reabsorption. In the distal tubule and collecting duct,
for example, sodium reabsorption is stimulated by aldosterone, a steroid
hormone secreted by the adrenal cortex, and inhibited by atrial
natriuretic peptide, a peptide hormone secreted by the atria of the
heart. In these tubular segments, water reabsorption is stimulated
by antidiuretic hormone (ADH), a peptide hormone secreted by the
posterior pituitary. Actions of these hormones and others in fluid and
electrolyte balance are described later in this book (in Chapter 19).
Water Conservation in the
Loop of Henle
The loop of Henle within juxtamedullary nephrons is specialized to create
an osmotic gradient in the renal medulla, such that the fluid in the
outer portion of the medulla (near the cortex) is at a lower osmolarity