Yr 2 - Organic Chemistry
Section 25:
Section 26 - Compounds containing the carbonyl group
A carbonyl group contain C = O, such as aldehydes and ketones.
The physical properties:
The carbonyl group is strongly polar with the carbon being delta positive and the oxygen being delta negative.
These forces means these compounds have a higher boiling point than alkanes, but lower boiling point than hydrogen bonding present compounds
Shorter chain carbonyl containing compounds mix with water, because the hydrogen bonding in the water and the oxygen on carbonyl compound forms a bond.
With increased length decreased ability of carbonyl compound mixing with water.
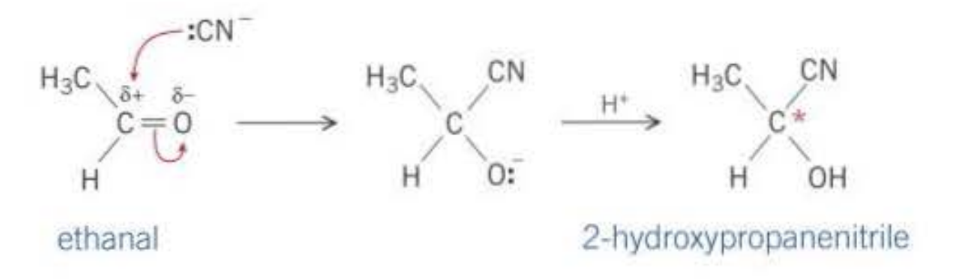
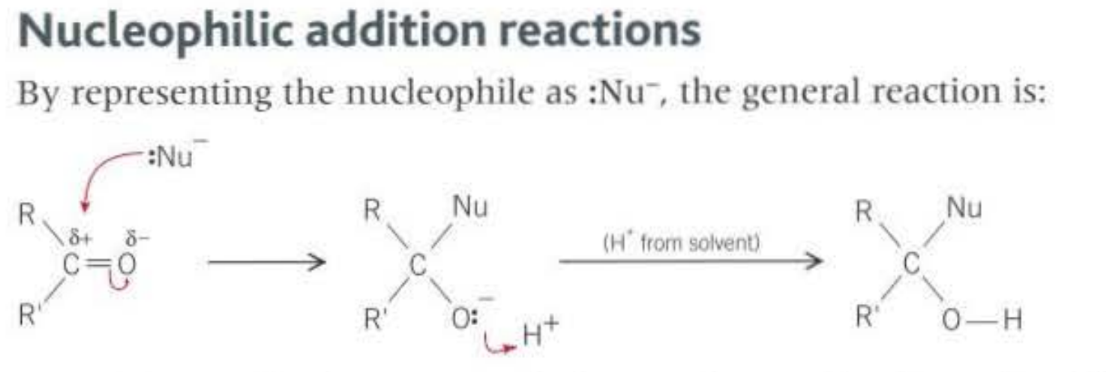
The C=O bond is highly reactive, this is because of the large difference in the electrongeativity between the carbon and the oxygen. This causes the carbon to be slighly positive, so nucleophiles can attack (most reactions nucleophillic addition)
Carboxylic acids:
They contain a carbonyl group and a hydroxy group (which is also in alcohols), but the difference is that the OH in carboxylic group is more acidic than OH in alcohols.
Esters:
They have a functional group of COO
The OH from the alcohol and the H from the carboxylic acid
To name an Ester: The alchol first then the carboxylic acid
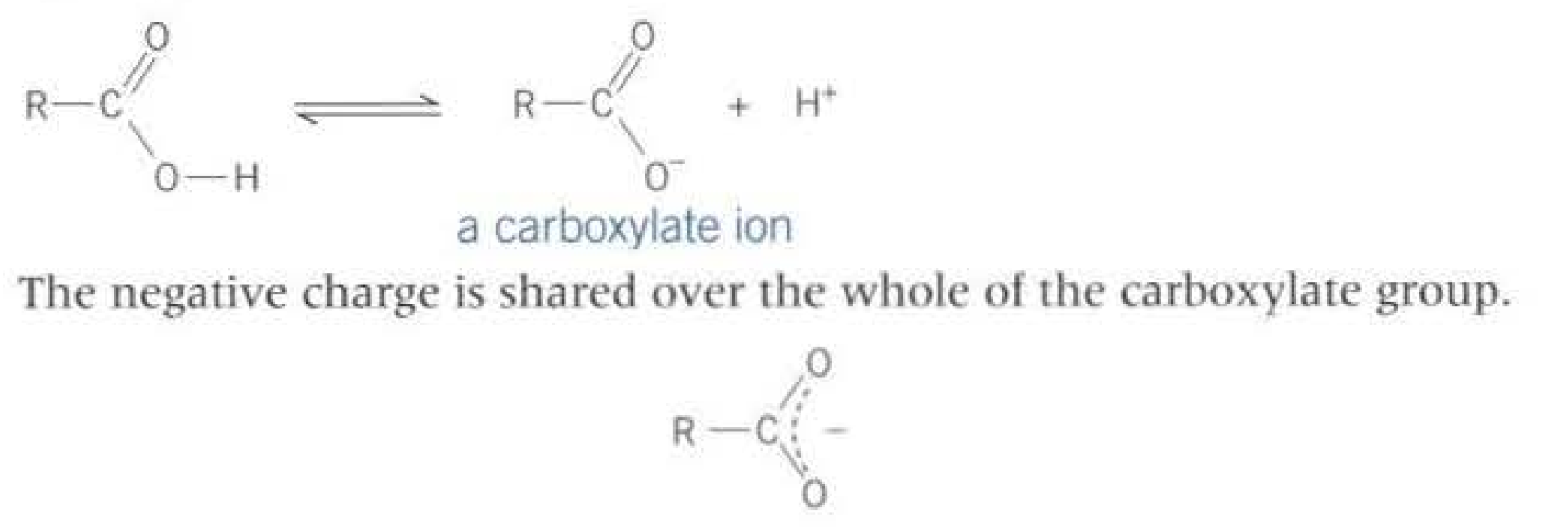
The picture above shows delocalisation which makes the ion more stable.
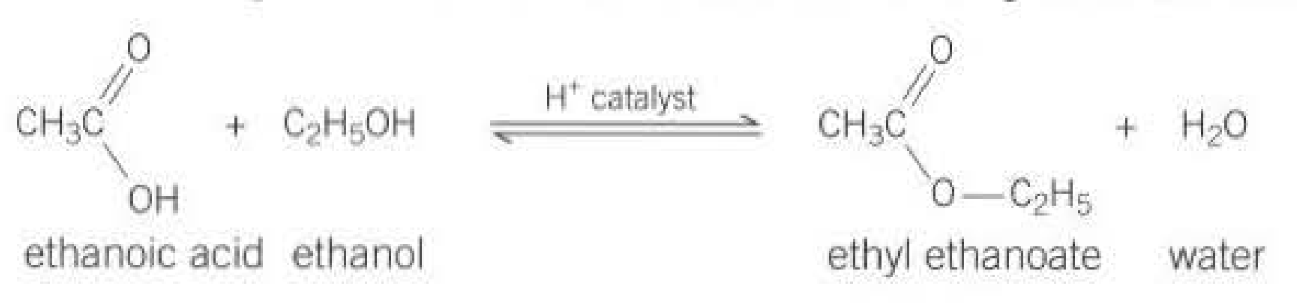
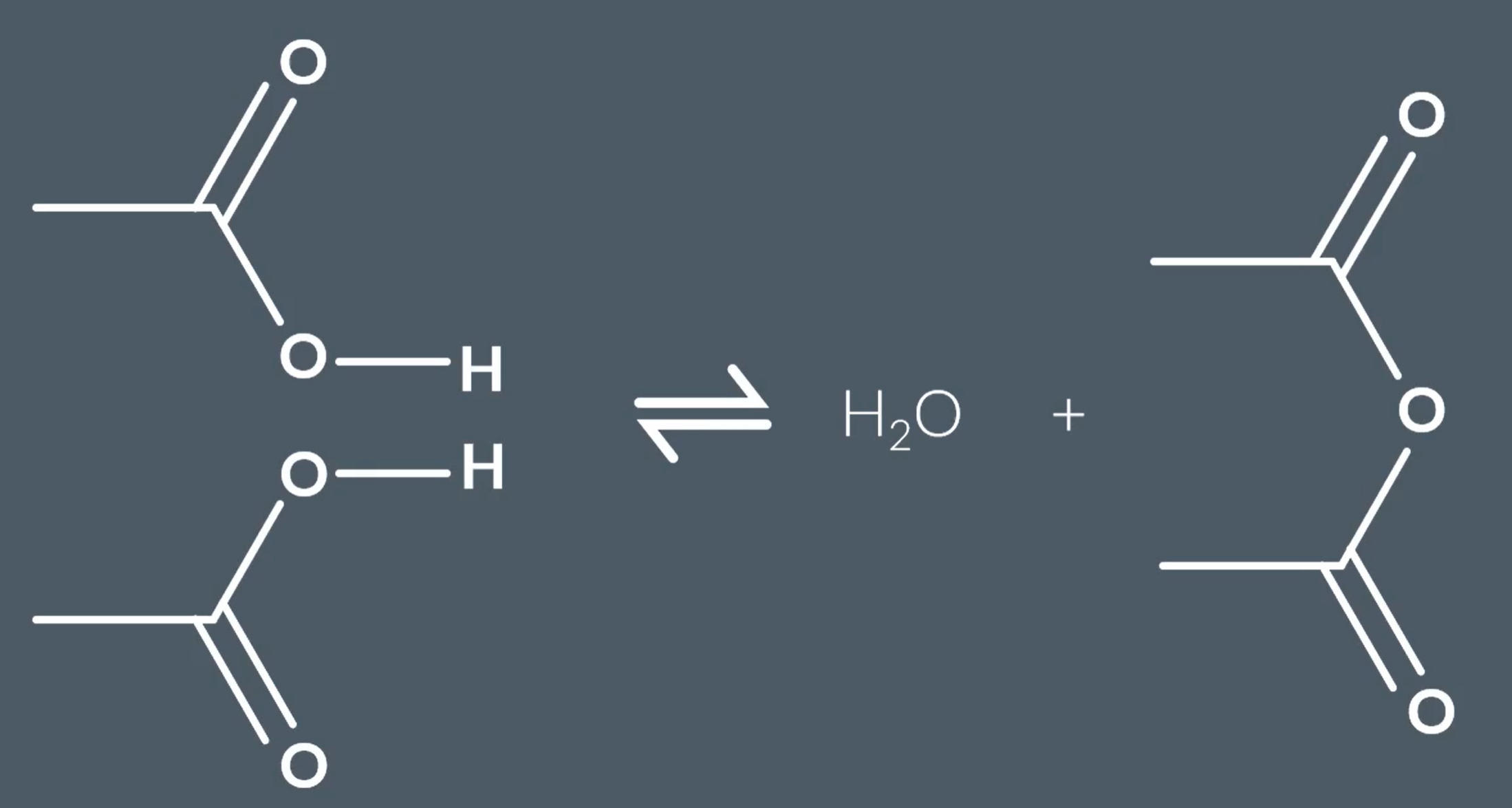
Formation of Esters:
Require a strong acid catalyst
It is a reversible reaction
Requires carboxylic acids and alcohols
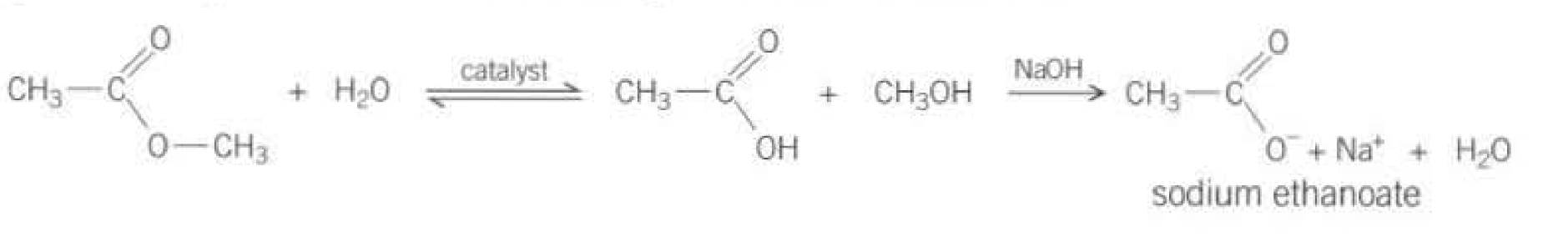
For the hydrolysis reaction using an acid:

For the hydrolysis reaction using a base:
The salt of the acid is formed rather than the acid itself
The last step means there is no equlibrium set, so the reaction goes to completion
Uses of Esters:
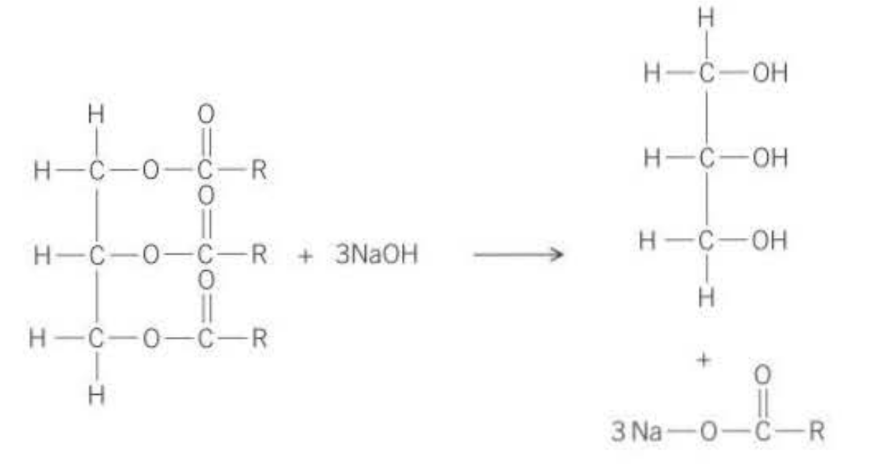
In fats and oils, there is ester components. The glycerol (propane-1,2,3-ol) and 3 fatty acids (carboxylic acids) react to form ester bonds.
They can be hydrolysed using sodium hydroxide. Both products from hydrolysis are useful, the glycerol and mixture of sodium salts of the three acids that formed part of the ester. The salts are soaps. The glycerol (contains OH bonds so very soluble in water) is used as a solvent, in many medicines and in food industry (found in toothpaste)
The RCOO- and the Na+ with dissociate as they are ionic. The long hydrocarbon chain which is non-polar and the COO- is polar and ionic. Where the hydrocarbon will mix with the grease and the COO- mixes with the water. Therefore is present in cleaning agents.
Acylation:
Is the process when acyl group is introduced into another molecule.
Acid anhydrides are when two of the acyl groups // carbonyl groups are bonded to the same oxygen.


Both Acyl chlorides and acyl anhydrides attract nucleophiles, however acyl chloride contains chlorine which is more electronegative so withdraws electrons from carbonyl group making the carbon more positive, enabling the nucleophile to attract the carbon more easily.
The following Nucleophiles:
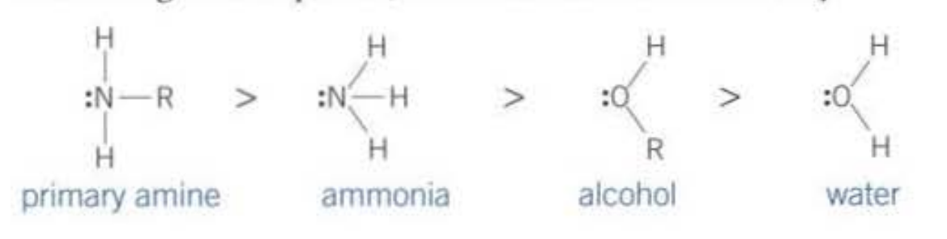
Primary amine → N-substituted amide
Ammonia → Amide
Alcohol → Ester
Water → Carboxylic acid
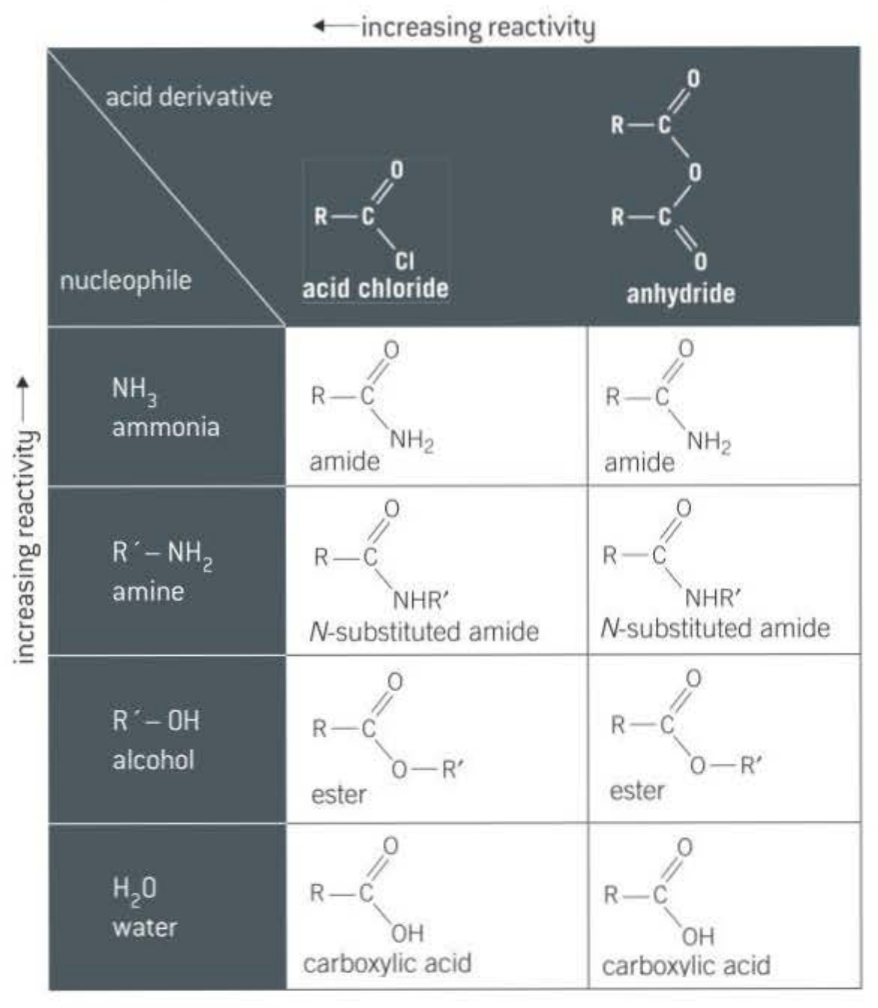
Eventhough Acid chlorides are more reactive, acid anhydrides are preferred.
It is cheaper
It is less corrosive
It does not react with water as readily
Acid chlorides produce HCl as a py-product which is not safe compared to acid anhydrides that form carboxylic acid as a by-product (more safer)
The reaction with acyl chlorides is highly exothermic.
Aromatic chemistry:
Key questions: refer to textbook for answers
27.1 Introduction to Arenes:
What are Arenes?
Describe the bonding and structure of Benzene:
Explain the evidence behind bonding and structure:
Why is Benzene stable?
Explain why the carbon-carbon bonds are of equal length?
27.3 Reactions of Arenes:
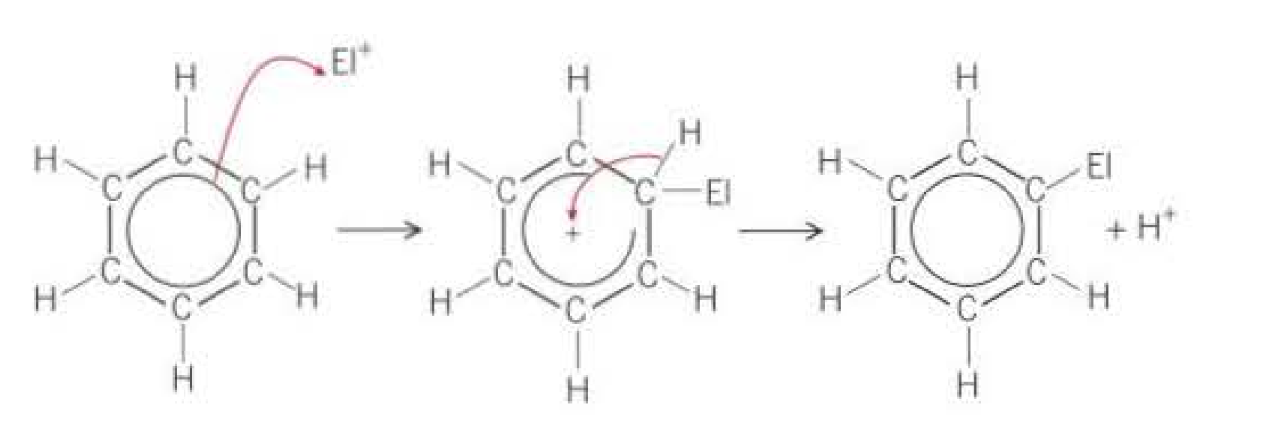
Why does Arenes undergo substitution reaction more readily than addition reactions?
Outline the mechanism: (Electrophillic substitution)
Nitration formuleas: When generating the electrophile (NO2+) is called the nitronium ion, the reagents and conditions are: CONC sufuric acid, CONC nitric acid
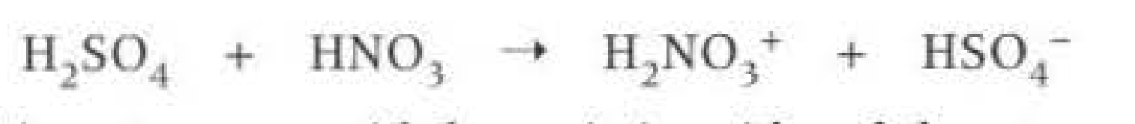

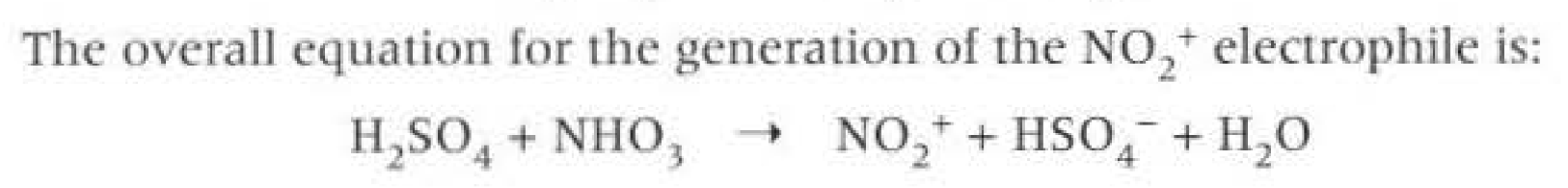
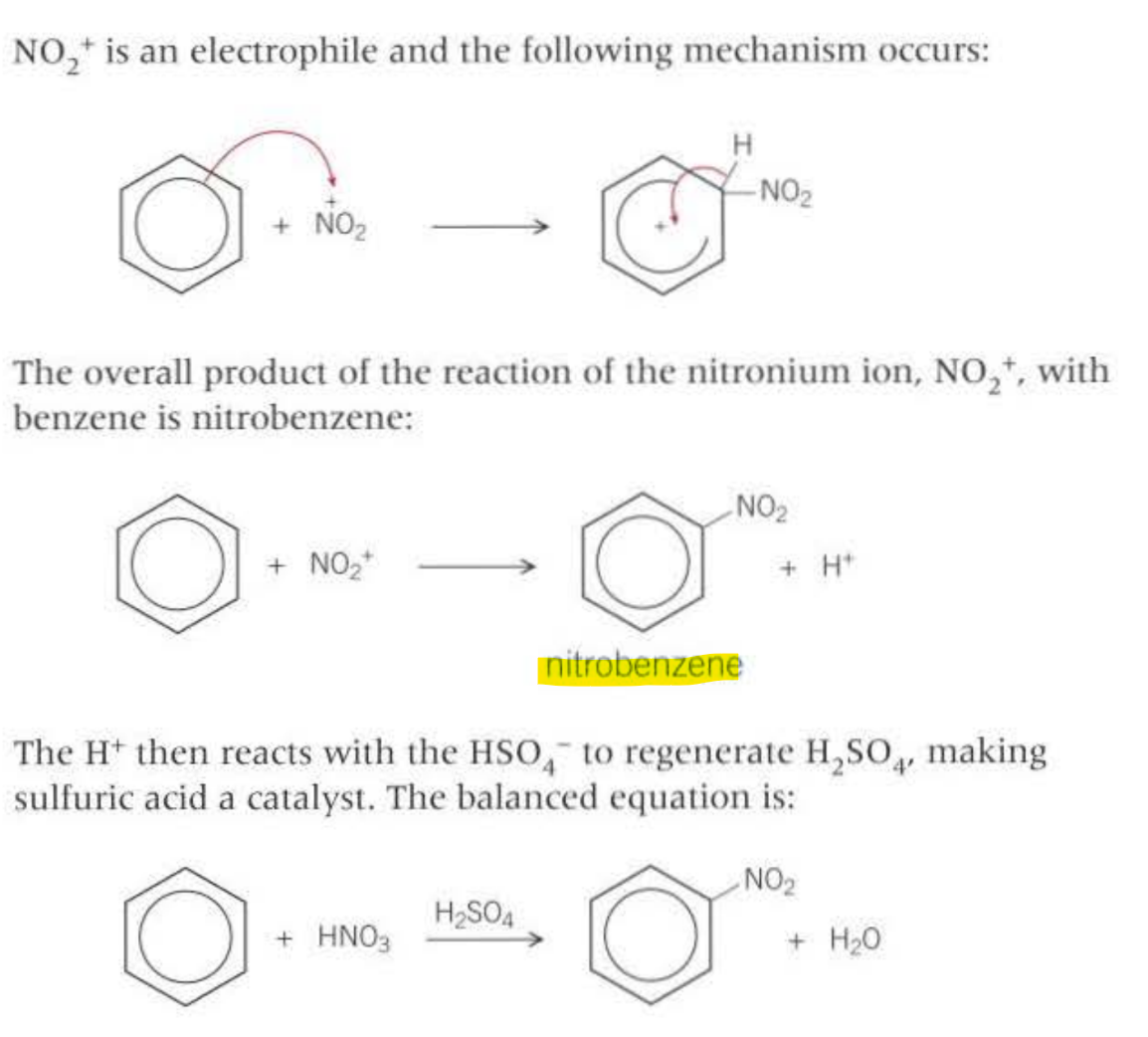
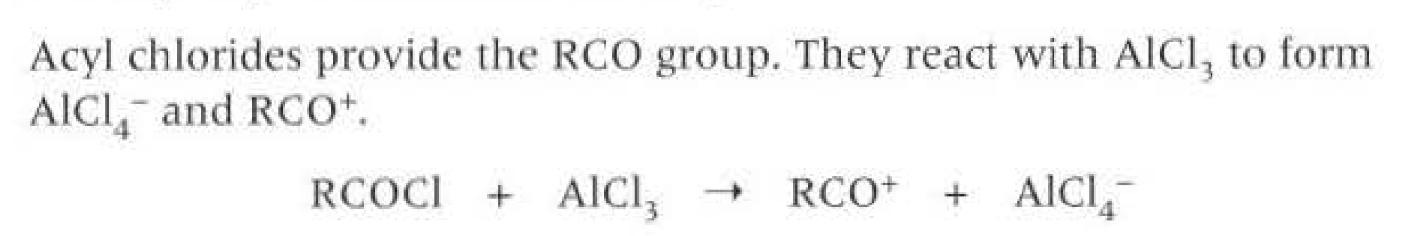

28 Amines:
28.1
What are amines?
They are Ammonia, where the hydrogen is replaced by one or more alkyl groups
Describe the shape of ammines, the boiling point compared to alcohols, the solubility and reactive properties (base + nucleophile)?
28.2 Properties of amines as bases:
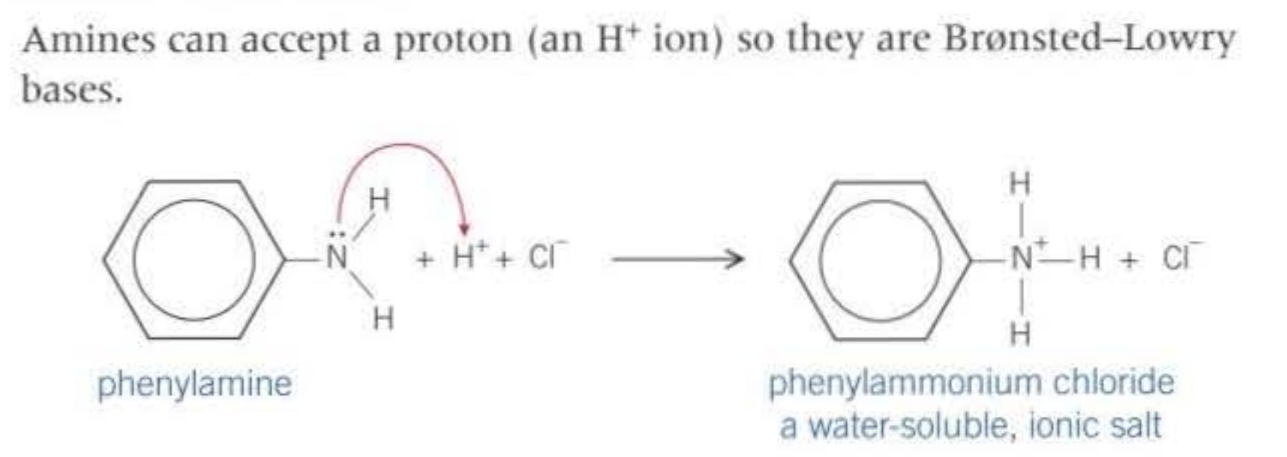
This salt reacts with water because the salt dissolves in water because both are polar.
To compare basic strength, use the positive inductive model, where with more alkyl groups, there is more electron density given to the Nitrogen on the NH3, so the nitrogen will be more delta negative so accept hydrogen more readily
So, secondary amines are more basic than primary amines
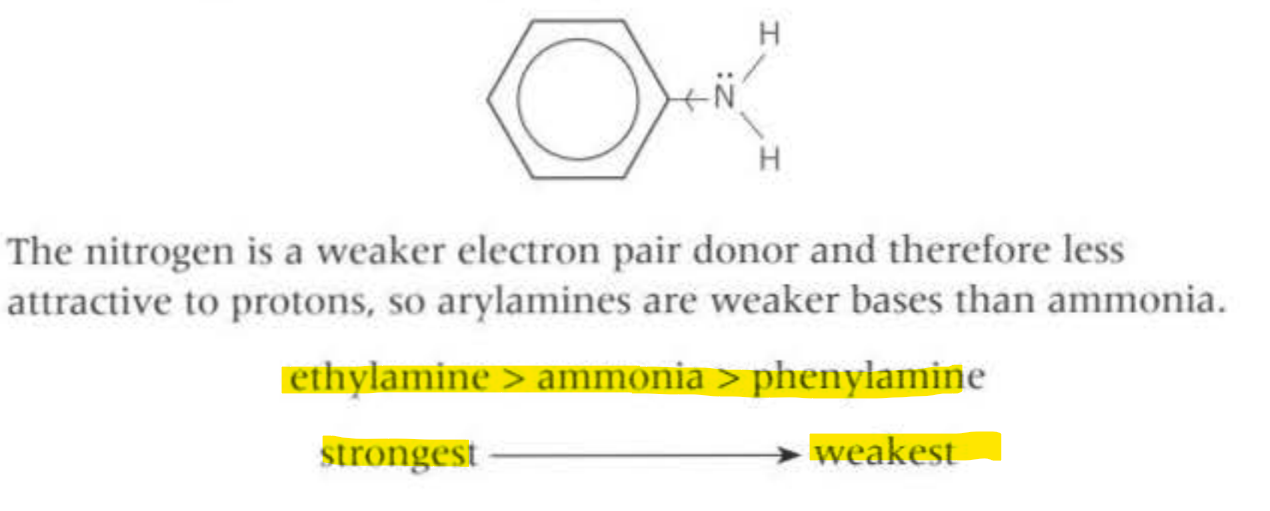
The key point is highlighted, the Phenylamine is the weakest, because the benzene ring withdraws electrons from the nitrogen making it more positive, so attracts the proton less than ethylamine, where the ethyl group forms a positive inductive effect.
The three ways to make primary amines:
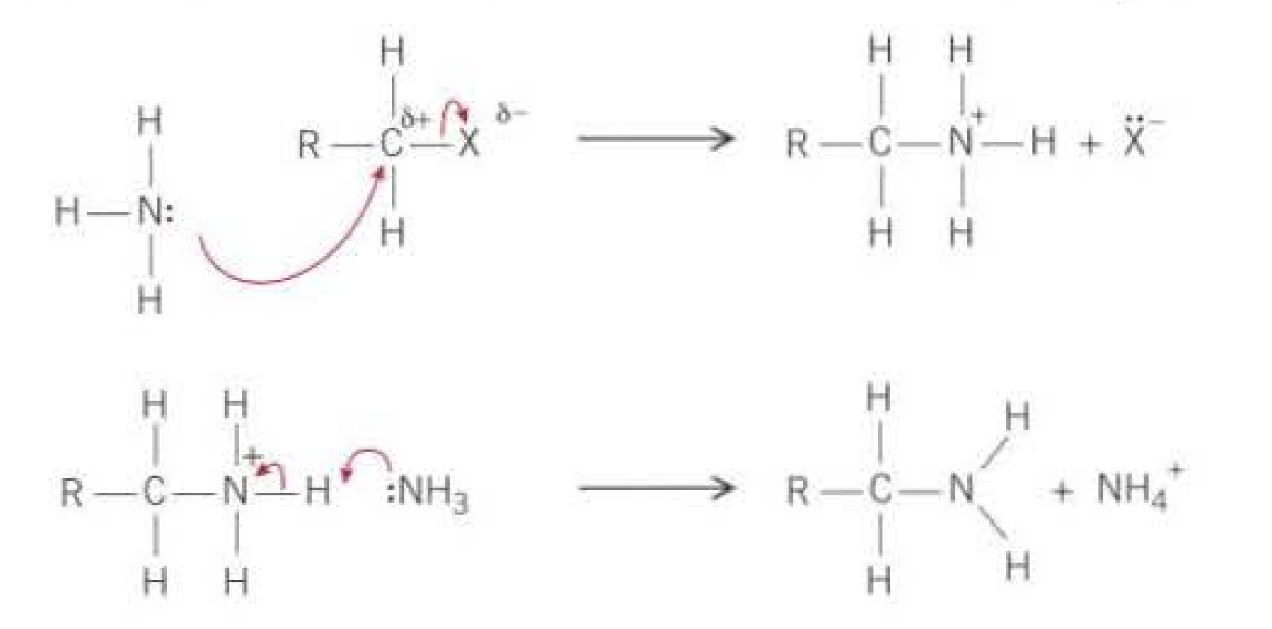
Nucleophillic substitution, of halogenoalkane and ammonia. When there is an excess of ammonia then the primary amine is formed. When there is an excess of the halogenoalkane then the quaternary ammonia salt is formed, used for conditioners
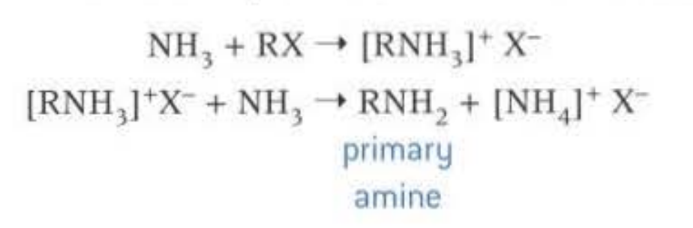

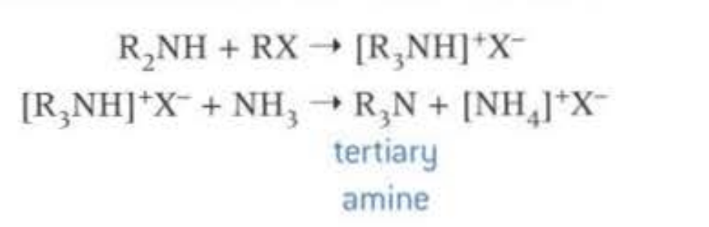
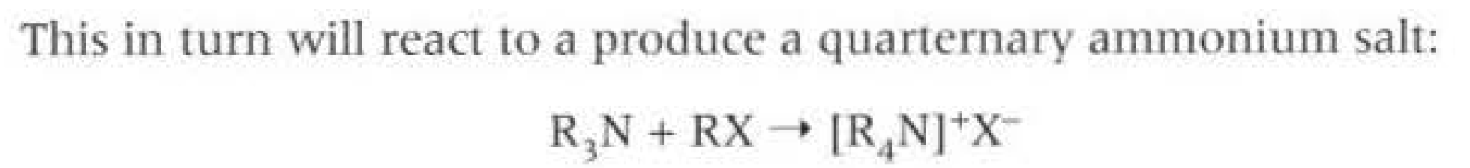
This causes a mixture of primary, secondary, tertiary and quarternary amines to be formed, so not a very efficient way of forming amine, as products have to be seperated via fractional distillation.
The mechanism:
Reduction of nitriles (C≡N)
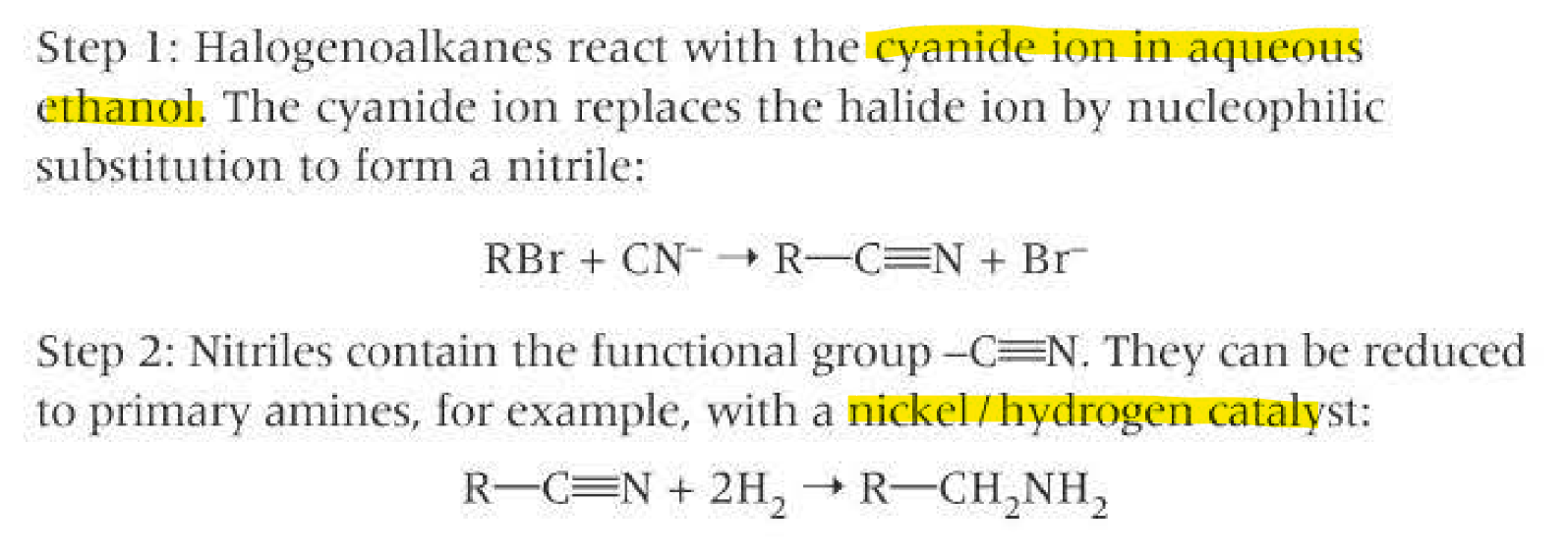
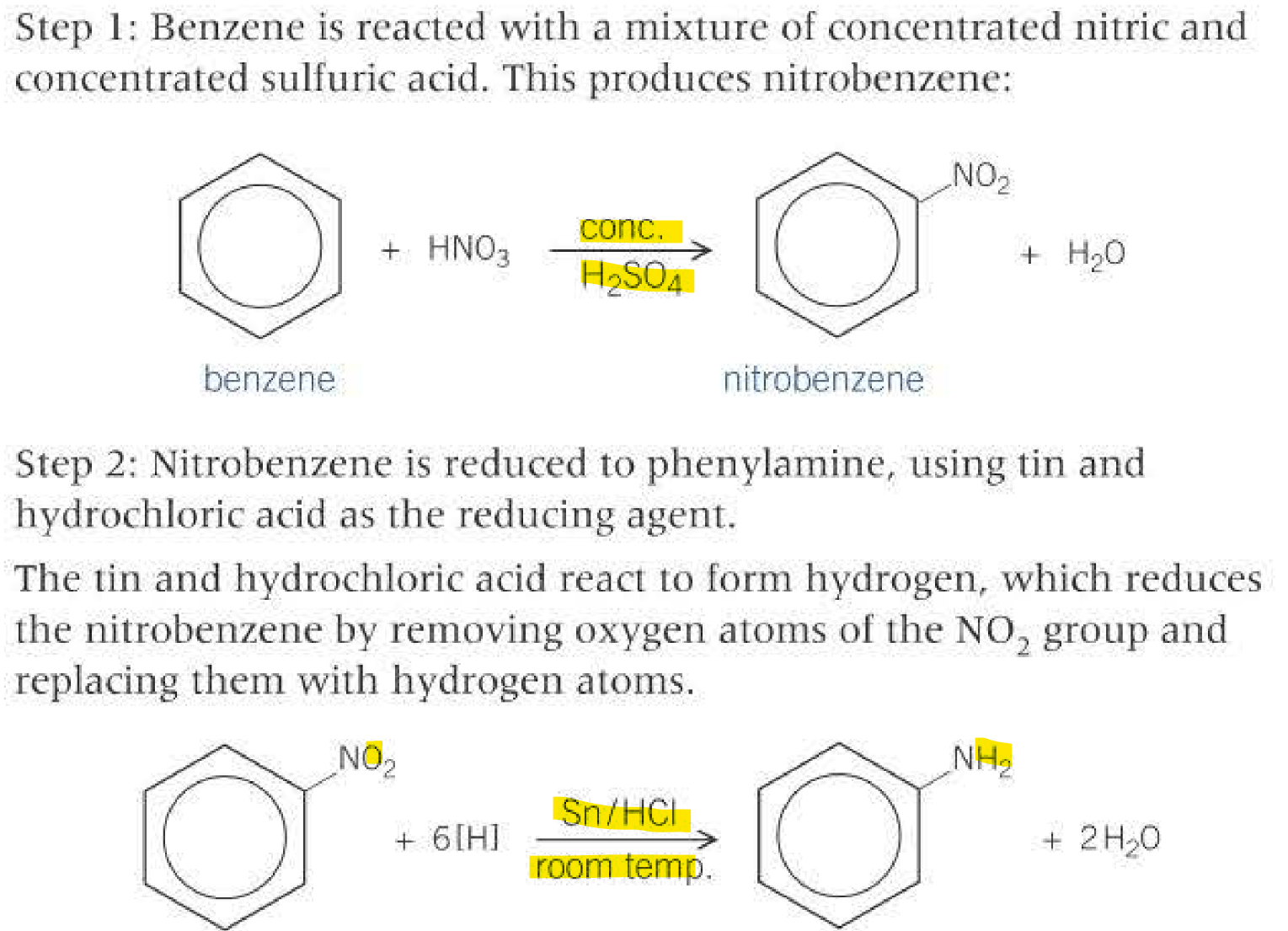
Phenylamine from benzene
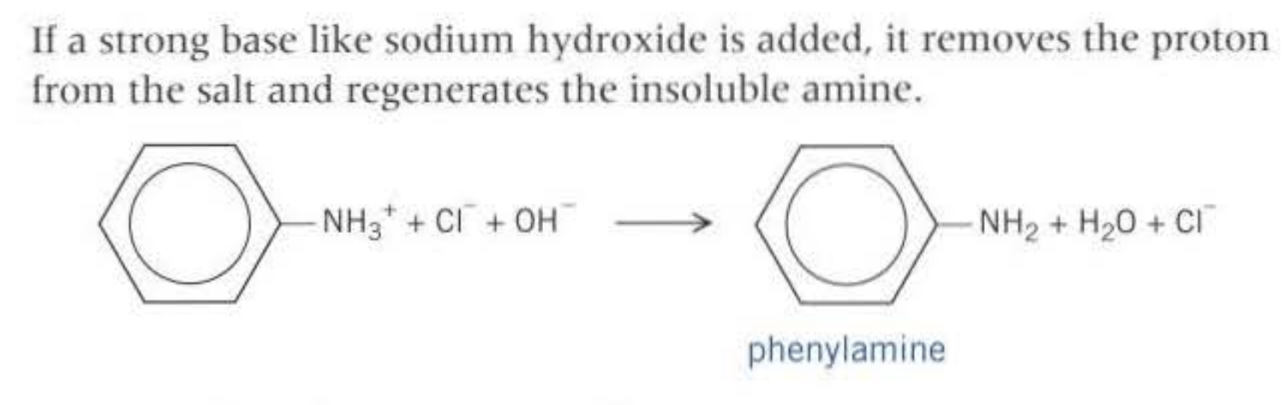
If HCl is used:NaOH is used to neutralise
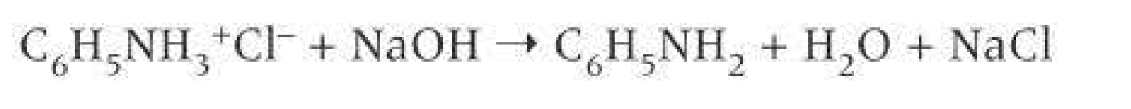
The uses of amines:
Quaternary amines: have long hydrocarbon chain and positively charged organic group, forming cations.
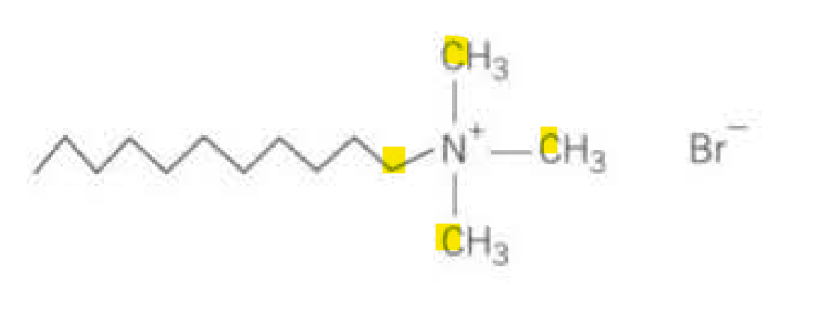
The positive charges on the cations attract to the wet surfaces, forms a coating that prevents the build-up of charge: so can form hair conditioners, detergents.
29 Polymerisation:
Condensation reaction: when two molecules react together and a small molecule is given off such as water of hydrogen chloride.
Example:
The OH from the carboxylic acids and the H from the alcohols leave and form water, and an ester is formed. To name an ester (the alcohol part then the carboxylic part such as ethyl ethanoate)
A polyester has the ester linkage -COO-, which is formed by when two functional Diols and dicarboxylic acid. Water is given off.
A polyamide has the amide linkage -CONH-, which is dicarboxylic group reacts with dicarboxylic acid and diaminoalkane react (which has two amine groups). Water is given off.
A polypeptide is also a polyamide => Made from amino acid repeating units.
Polyester, polyamide and polypeptide are recyclable, because they are hydrolysed by water.
Polyethene and polypropene is not recyclable, because they have only strong, non-polar C-H and C-C bonds. Formed via addition reactions.
Note: When it is the hydrolysis of a compound formed via condensation reaction, you can’t have water on its own - required to have NaOH/ HCl/ Na2CO3
CH 30 - Amino acids, proteins and DNA
Key summary from this chapter:
Amino acids have two functional groups, carboxylic acid and amine group
If the chiral carbon is bonded to 4 different groups, we call this geometric isomerism
The carboxylic acid has the tendency to loose a proton (acts as an acid) => deprotonated
The amine group has the tendency to accept a proton (acts as a base) => protonated
So, amino acids behave as a zwitterion, where they both have a positive (amine group) and a negative charge (carboxylic group), but overall molecule is neutral
They are ionic, so they dissolve well in water, but not in non-polar solutions
In strong acidic situations, the NH2 gains proton (protonated). In strong alkaline solutions, the COOH looses a proton (deprotonated)
Key idea, maintain this kind of equilibrium
When an amino acid reacts with a carboxylic acid, we have an amide linkage (in biology we call it peptide bond) => formed via a condensation reaction
To hydrolyse peptide (2 amino acids bonded together) and water and HCl (acts as a catalyst) and boil
Primary structure: The sequence of amino acids => held together by covalent bonds
Secondary structure: This forms the beta-pleated sheet and the alpha helix => held together by the hydrogen bonds between C=O and N-H
Tertiary structure: The alpha helix shape or beta-pleated sheet folds into three dimentions, through series of more hydrogen bonding, ionic bonding and disulfide bridge bonds
Enzymes are biological catalysts that speeds the rate of reaction without getting used up
Chemistry uses the lock and key model: Where the enzymes’ active site fits perfectly with the reacting molecule. Where it is bonded temporarily by the inter molecular forces. When bonded this promotes the electron movement within the substrate, which lowers the activation energy.
Properties of enzymes: Stereospecificity, meaning that the enzyme can only catalyse the reaction between one or the other sterioisomer.
Inhibtition of enzymes: describe competitive inhibitors (bind to the active site) and non-competitive inhibitors (bind to another area of enzyme other than active site)
The monomers of DNA are nucleotides
Nucleotide consists of ribose sugar, phosphate ion and a nitrogenous base (ATCG)
The phosphodiester backbone is formed via a condensation reaction, when the H on the phosphate group and OH from sugar make water a bond of C-O is formed
The shape of DNA is double helix, with the nitrogneous bases forming hydrogen bonding. Where C-G forms 3 hydrogen bonds and A-T forms 2 hydrogen bonds
Why is the DNA acidic? The phosphate group, P-O-H where the H is donated, forming a P-O negative.
The action of anti-cancer drugs:
Cisplatin is a square planar shape and has the following displayed formulea
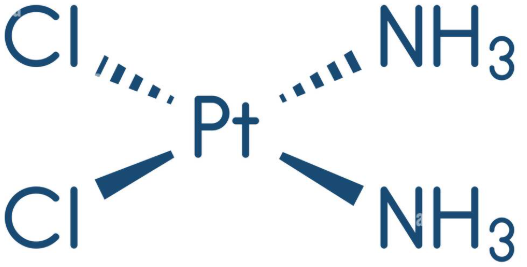
This works via a ligand substitution reaction, where when the cisplatin enters the chloride groups are replaced by water ligands, then these water ligands form hydrogen bonding between two adjacent guanime bases, this stops transcription and transulation for fastly dididing cells such as cancerous cells.