Hydrocarbons
{{{{
\ <<1. decarboxylation reaction: When sodium salt of fatty acid in the presence of sodium hydroxide and soda lime (3:1) ratio gives one less carbon than the reactant. The product formed is an alkane. It is directly proportional to the stability of the anion.<<
<<2. Kolbe’s electrolysis: By the electrolysis of aqueous concentrated sodium or potassium salt of carboxylic acid then symmetrical alkane is formed at the anode.<<
\
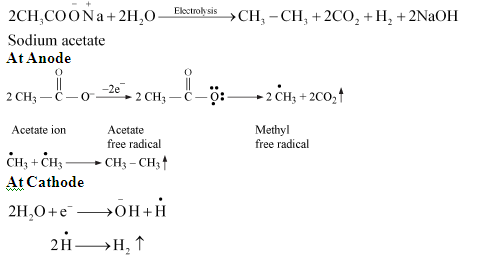
\ <<3. From alkyl halide:<<
<<i) Reduction of alkyl halide: it gives alkane. the reducing agents are Red P/ HI, Zn/HCl, Zn/Ch3cooh, Al-Hg/ethanol<<
<<ii) Wurtz reaction: when an alkyl halide is heated with sodium in presence of dry ether, a symmetrical alkane is formed.<<
<<iii) Frankland reagent: when alkyl halide is heated with zinc in an inert medium, then alkane is obtained.<<
<<4. From unsaturated compounds: In the presence of Ni/H2 when alkyne/alkene reacts it forms alkane. This reaction is called Sebetian Sandmeyer reaction.<<
<<5. From Grignard reagent:<<
<<6. Fittig reaction:<<
<<7. Wurtz-Fittig reaction:<<
<<8. Correy-House synthesis:<<
<<9. From G.R:<<
<<10. From metal carbides:<<
<<11. From reduction of -oh, -cooh, -c=o, -c=o-oh in presence of red P/HI:<<
\ \ \ \