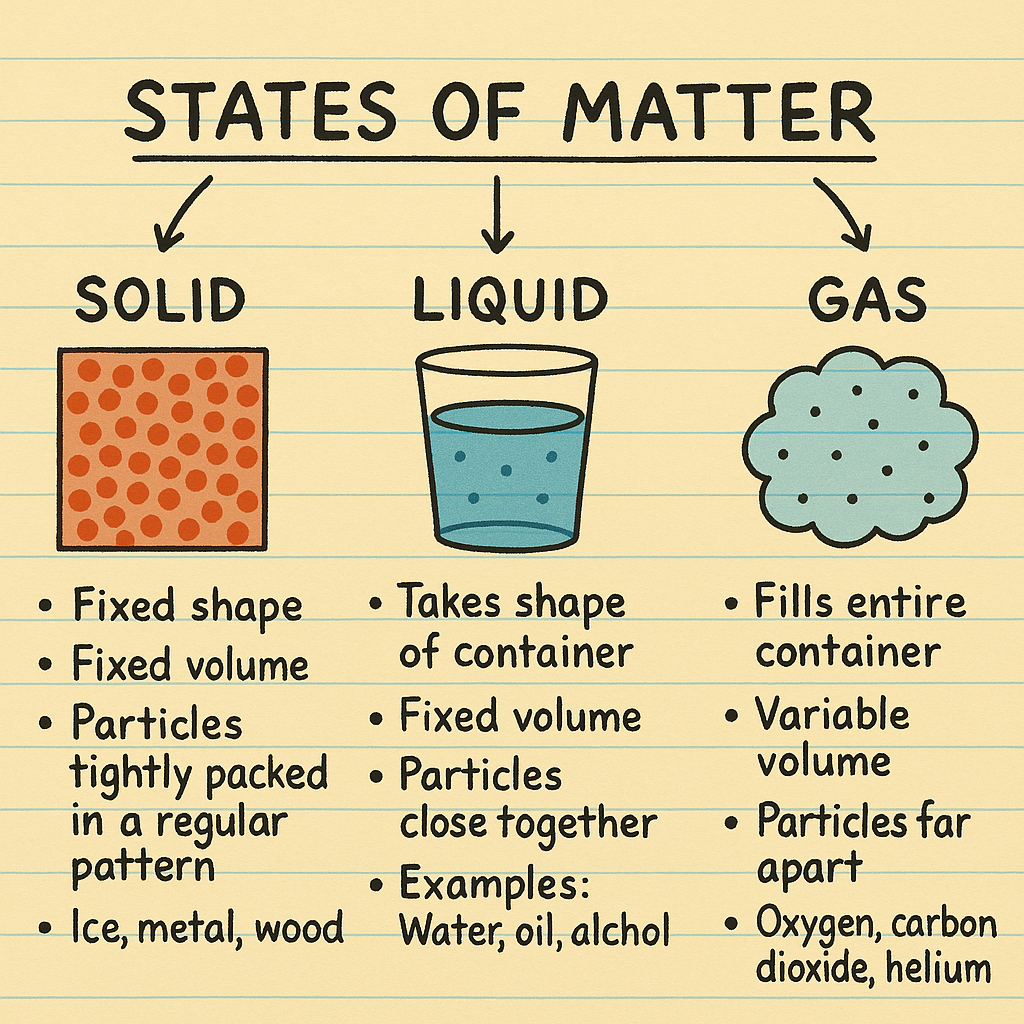
solids, liquids, gases
🌡 States of Matter: Solids, Liquids & Gases
🧊 Solid
Shape: Fixed
Volume: Fixed
Particle Arrangement: Tightly packed in an orderly structure
Movement: Vibrate in place
Compressibility: Not easily compressed
Examples: Ice, metal, wood
💧 Liquid
Shape: Takes shape of container
Volume: Fixed
Particle Arrangement: Loosely packed, can slide past each other
Movement: Moderate movement
Compressibility: Slightly compressible
Examples: Water, oil, alcohol
💨 Gas
Shape: Fills entire container
Volume: Variable
Particle Arrangement: Far apart, no fixed arrangement
Movement: Rapid and random
Compressibility: Easily compressed
Examples: Oxygen, carbon dioxide, helium
Diagram