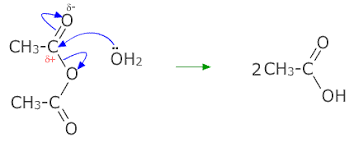
Acyl chlorides and acid anhydrides
Acyl chlorides
Made by reacting a carboxylic acid with SOCl2
e.g.
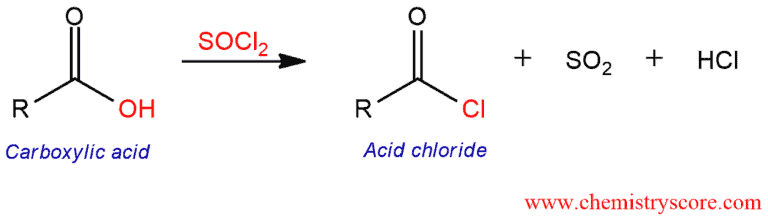
Very reactive
Acyl chlorides have polar bonds
They attract nucleophiles to the delta positive on the carbon of the C=O
e.g. H2O, NH3, OH group on alcohols -CN
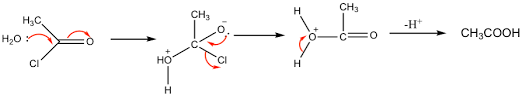
H2O adds to the acyl chloride forming a dative covalent bond. The double bond breaks - addition stage
The double bond reforms and the chlorine leaves as Cl- - elimination stage
A proton leaves
Overall: nucleophilic addition-elimination
Acid anhydrides
Made by dehydration of 2 carboxylic acid molecules
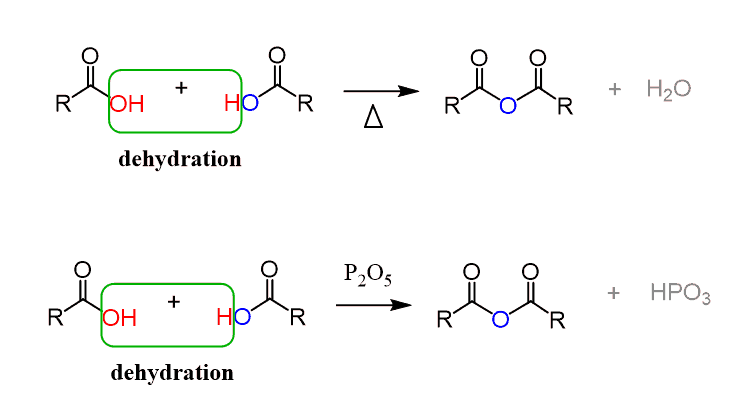
Not as reactive as acyl chlorides
Reaction with water: