13.2 Genomes and chromosomal abnormalities
Identify normal and simple, abnormal karyotypes
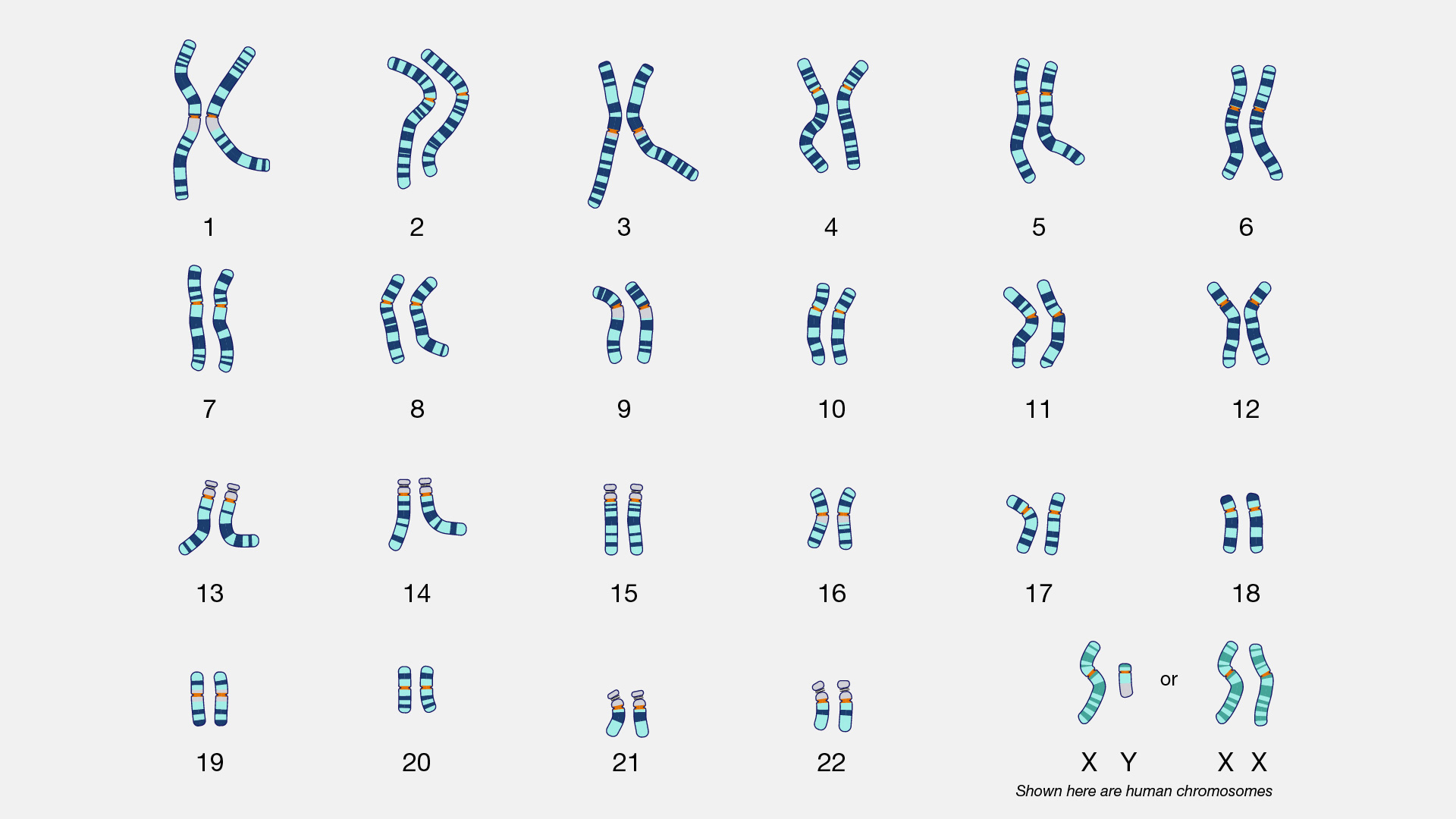
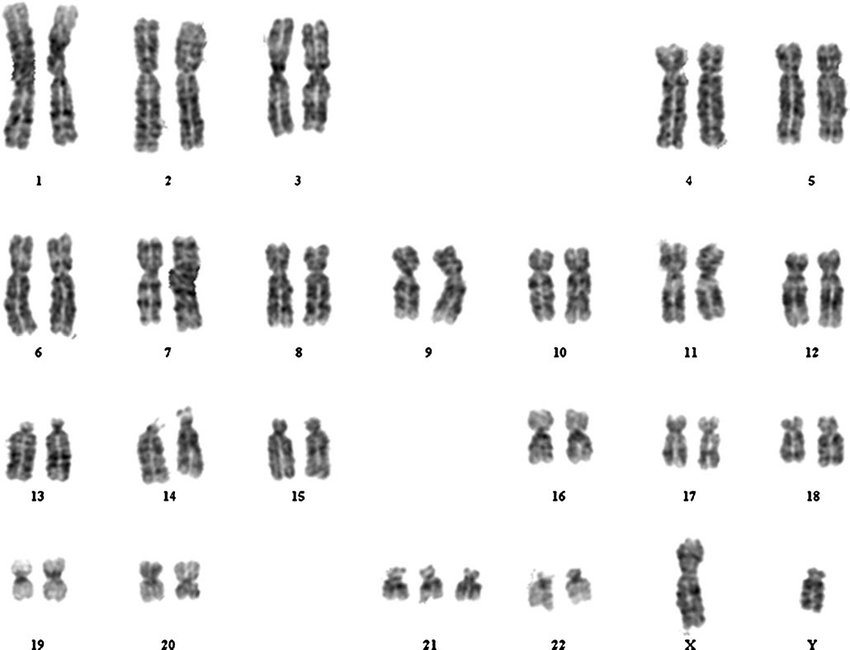
Normal karyotype
46 chromosomes, 23 pairs: 22 autosomes + 1 sex pair
Female: 46, XX
Male: 46, XY
Karyotyping technique
G-banding using Giemsa stain during mitosis
Sample sources: blood, amniotic fluid, chronic villus, tumour biopsy
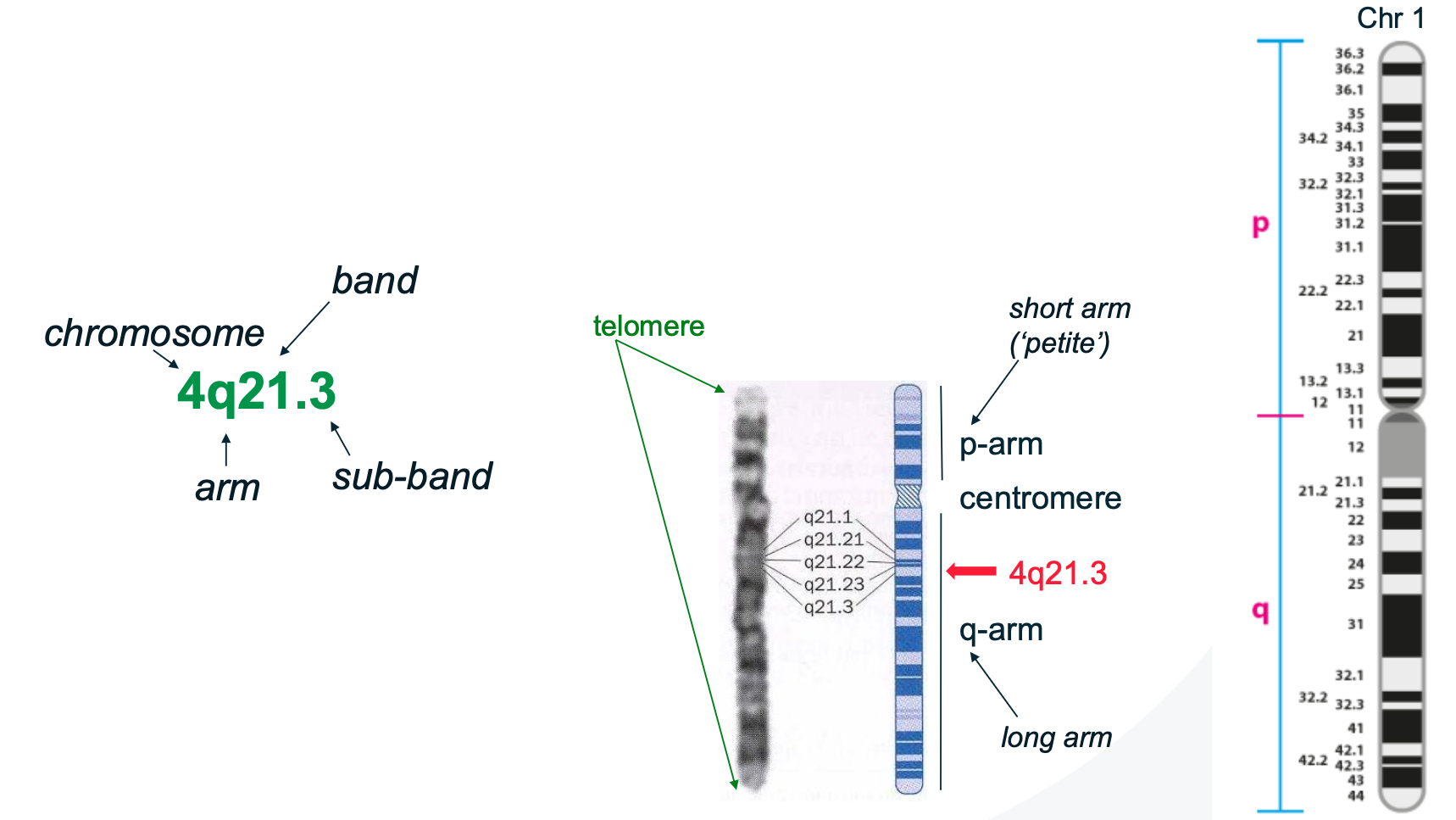
G-banding ‘addresses’ for chromosomes
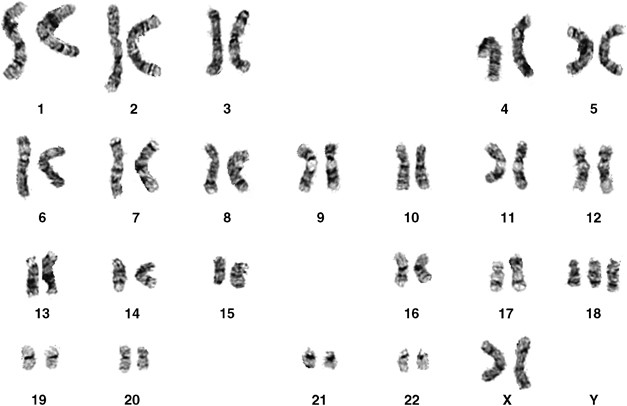
Abnormal karyotype examples
Karyotype | Description | Condition |
47, XX/XY, +21 | Trisomy 21 (3 chromosomes for pair 21) | Down syndrome |
47, XX/XY, +18 | Trisomy 18 (3 chromosomes for pair 18) | Edward’s syndrome (usually lethal in first year) |
47, XX/XY, +13 | Trisomy 13 (3 chromosomes for pair 13) | Patau syndrome (usually lethal within a few months of birth) |
45, X | Monosomy X (Only 1 X no XX or XY) | Turner syndrome |
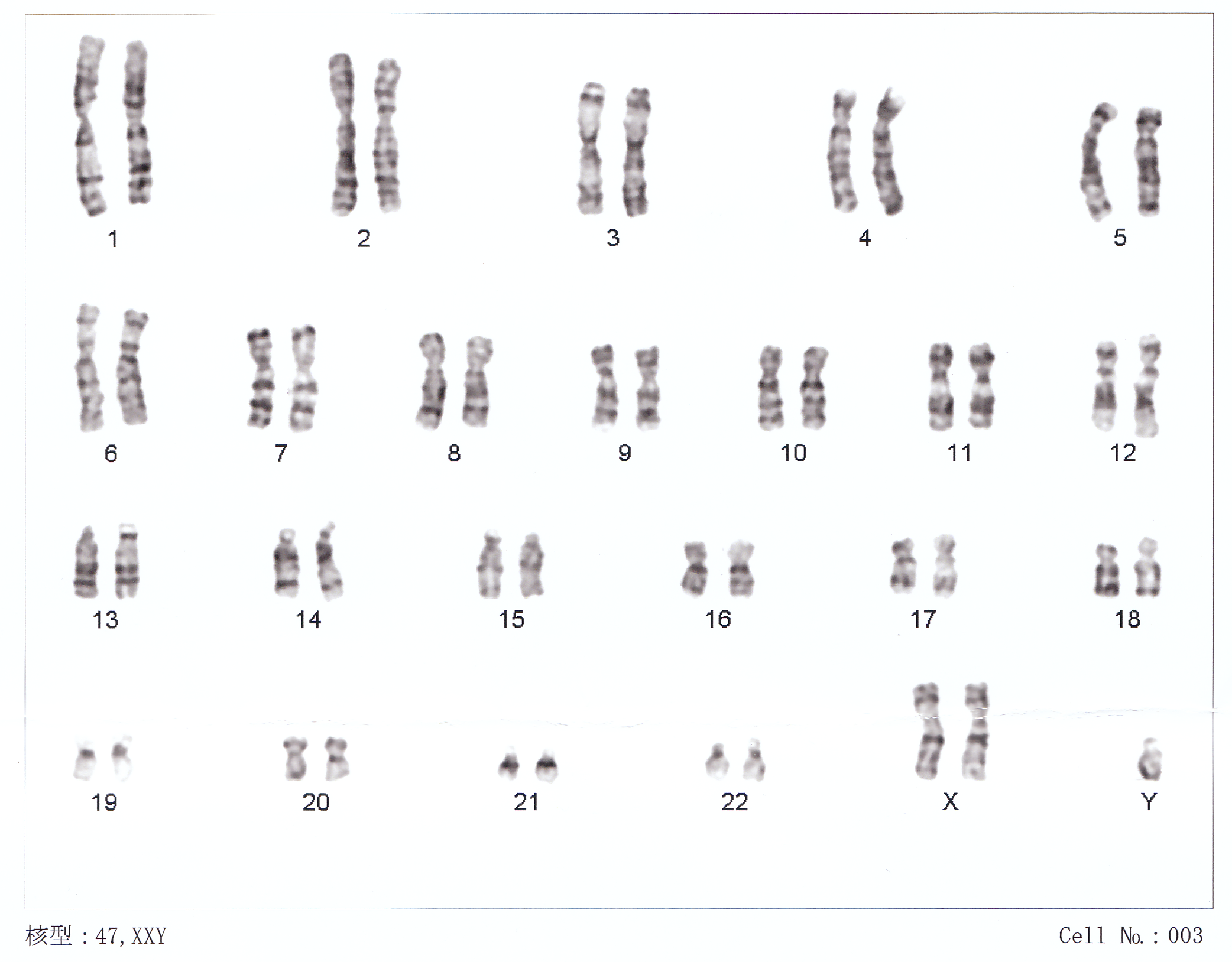
47, XXY | Extra X in males | Klinefelter syndrome |
69, XXX | Triploidy (Triple chromosomes instead of pairs) | Usually lethal |
46, XX, t(1;22) | Unbalanced translocation | Chromosomal abnormality |
Aneuploidy: missing or extra chromosomes
Trisomy 21 (Down syndrome)
Trisomy 18 (Edward’s syndrome)
Trisomy 13 (Patau syndrome)
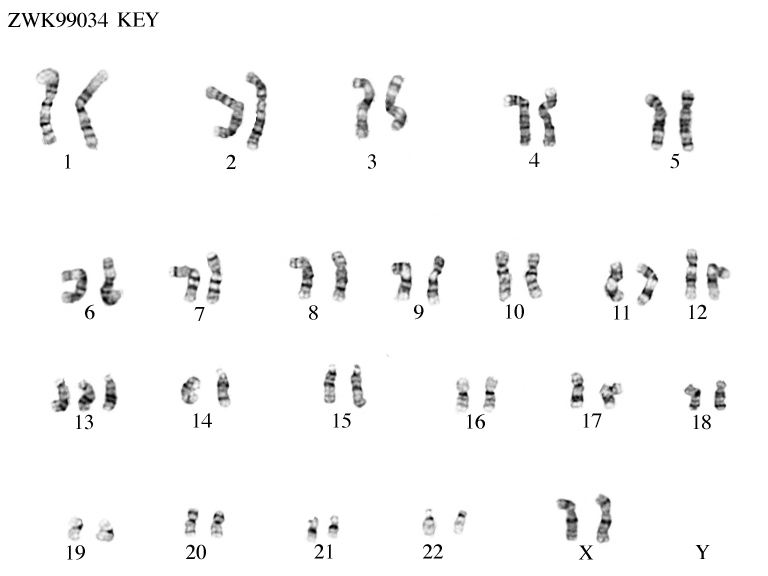
Monosomy X (Turner syndrome)
Klinefelter syndrome
Triploidy

Limitations of traditional chromosome analysis
Traditional karyotyping
Visualisation of chromosomes at metaphase using Giemsa stain (G-banding)
Allows for the identification of:
Chromosome number
large structureal abnormalities
Banding pattern
Sex chromosomes
Common sample types:
Peripheral blood (adults)
Chorionic villi or amniotic fluid (prenatal)
Tumour biopsies (cancer cytogenetics)
Key limitations
Low resolution: Cannot detect small deletions, duplications or point mutations; resolution limited to changes less than 5-10Mb
Requires dividing cells: Only metaphase chromosomes can be visualised, so cells must be cultured and arrested—which takes several days
Misses mosaicism: If only a subset of cells is abnormal, and not sampled in culture, it may go undetected
Not gene-specific: Cannot detect specific gene mutations (e.g. CFTR in cystic fibrosis, BRCA1/2)
Labor intensive and time-consuming: Requires skilled technicians and prolonged cell culture
Modern complementary techniques
Technique | Description | Strength |
FISH (Fluorescent in Situ Hybridisation) | DNA probes hybridise to specific gene regions | Detects specific sequences, doesn’t require mitotic cells |
Chromosomal microarray (CMA) | Detects copy number variations at much higher resolution than karyotyping | Can detect microdeletions/duplications missed by G-banding |
Whole genome sequencing | Reads entire DNA sequence | Detects point mutations, indel, CNVs, structural variants |
Sex chromosome abnormalities and sex determination
Basic genetics of sex determination
Determined by the presence of the SRY gene (Sex-determining region Y)
Located on Y chromosome
Triggers differentiation of the bipotential gonads into testes
Without SRY → ovaries develop
Phenotypic sex is determined by:
Sex chromosome composition
Gene expression patterns
Hormonal signalling
Common sex chromosome aneuploidies
Turner syndrome — 45,X
Monosomy X – only viable monosomy in humans
Phenotypic sex: Female
Clinical features:
Short stature
Webbed neck, broad chest
Primary amenorrhoea, streak ovaries → infertility
Normal intelligence, may have subtle learning difficulties
Increased risk of cardiac defects, horseshoe kidney
Klinefelter syndrome — 47,XXY
Extra X chromosome in male
Phenotypic sex: Male
Clinical features:
Tall stature, small testes
Gynecomastia (man boobs)
Infertility due to azoospermia (absences of motile sperm)
Feminine body fat distribution
XYY syndrome — 47,XYY
Male with an extra Y chromosome
Usually normal phenotype
Tall stature
May have mild learning or behavioural difficulties
Triple X syndrome — 47,XXX
Female with an extra X
Often undiagnosed
Usually normal phenotype
May be taller than average; occasional developmental delays
Why are the effects milder than autosomal aneuploidies
X-inactivation: All but one X chromosome is inactivated in each cell → reduces dosage imbalance.
Y chromosome has few genes, many not essential for somatic development.
Thus, sex chromosome aneuploidies often have less severe developmental impact than autosomal ones.
Describe: Trisomy, Monosomy, Triploidy, Translocation
Trisomy
Definition: Presence of 3 copies of a specific chromosome (instead of the normal 2)
Caused by non-disjunction during meiosis
Most common:
Trisomy 21 (Down syndrome): Only trisomy compatible with adult life
Trisomy 18 (Edwards): Severe congenital anomalies; <1-year survival
Trisomy 13 (Patau): Midline defects; survival typically <6 months
Effects due to gene dosage imbalance: having 3 copies of dosage-sensitive genes
Monosomy
Definition: Loss of one chromosome (45 chromosomes total)
Only viable case: Monosomy X (Turner syndrome)
Autosomal monosomies: Lethal very early in embryogenesis
Triploidy
Definition: Three complete sets of chromosomes (69 total)
Mechanisms:
Dispermy: Fertilisation of one egg by two sperm
Diploid gametes due to meiosis failure
Outcomes:
Most cases miscarry in the first trimester
Rare live births → die within days
Translocation
Definition: Rearrangement of chromosome segments between two non-homologous chromosomes
Types:
Balanced translocation: No genetic material lost or gained → often asymptomatic
Unbalanced translocation: Additional or missing genetic material → may cause birth defects or miscarriage
Connect aneuploidies with errors in meiosis and mitosis
What is aneuploidy
Any deviation from the normal number of chromosomes (46)
Arises from non-disjunction events: Failure of chromosome separation
Key mechanisms:
Meiosis I non-disjunction
Failure of homologous chromosomes to segregate
All resulting gametes abnormal: 2 with n+1, 2 with n-1
Meiosis II non-disjunction
Failure of sister chromatids to separate
Results in 2 normal gametes, 1 with n+1, 1 with n-1
Mitotic non-disjunction (post-zygotic)
Occurs after infertilisation
Leads to mosaicism: mixture of normal and abnormal cells
Clinical severity depends on:
Proportion of affected cells
Tissues involved
Timing of mutation during development
Trisomy 21 origins (most cases of trisomy)
~85% of cases from maternal non-disjunction
~65% meiosis I
~20% meiosis II
Remaining cases:
~5% paternal meiosis
~5% mitotic error (mosaic)
~4% Robertsonian translocation