1.1 |McGraw-Hill Ryerson Biology 12
1.1 |McGraw-Hill Ryerson Biology 12
Unit 1: Biochemistry
Chapter 1: The Molecules Of Life
Section 1.1: CHEMISTRY IN LIVING SYSTEMS
- All matter is composed of elements (substances that can't be broken down into simpler substances by ordinary chemical methods).
- About 92 elements serve as the building blocks of matter.
- Yet only six elements (carbon, hydrogen, nitrogen, oxygen, phosphorus, sulphur) are the chemical foundation for life's diversity.
- Carbon and hydrogen form the underlying structures of biological molecules. The other four provide particular properties to these molecules.
- An atom is the smallest particle of an element that retains the properties of that element.
- Each atom has a specific atomic mass (the sum of its protons and neutrons).
- All atoms of an element have the same number of protons however, the number of neutrons can vary. Atoms of the same element with different numbers of neutrons are called isotopes. For example, carbon has three common isotopes: carbon-12 has 6 neutrons (most abundant), carbon-13 has 7 neutrons and carbon-14 has 8 neutrons.
- Some isotopes are unstable. This means that their nucleus decays and breaks down by emitting radiation in the form of subatomic particles or electromagnetic waves. These isotopes are referred to as radioisotopes. Carbon-14 is a radioisotope. Radioisotopes are used as diagnostic tools in medicine. Radioactive glucose is usually injected into the patient and its movement is tracked. Cancerous cells take in more glucose than normal cells. When performing a scan you will be able to see if cancerous cells are breaking down the radioactive glucose at a faster rate than normal cells. This method is used to diagnose cancerous tumours.
Studying the Interactions of Molecules
- For most biological studies, elements are not considered in the form of individual atoms but, rather as components of molecules.
- Molecules are substances composed of more than non-metal atoms which are covalently bonded.
- Organic molecules are carbon-containing. Its carbon atoms are usually bonded to each other and hydrogen. Organic molecules can also include nitrogen, sulphur and phosphorus atoms.
- Biochemistry is the study of the activity and properties of biologically important molecules. It forms a bridge between chemistry and biology.
Interactions Within Molecules:
- Intramolecular forces are the forces that hold atoms together within a molecule. They are generally thought of as the chemical bonds within the molecule.
- Covalent bonds are the bonds within a molecule. They form when the electron shells of two non-metal atoms overlap, causing the valence electrons of each atom to be shared. Each atom has access to the electrons in the bond as well as its other valence electrons. This way both atoms obtain a full valence shell.
- Depending on the atom, it may attract electrons more strongly than other atoms. This is called electronegativity. Oxygen, nitrogen and chlorine have high electronegativity. While, hydrogen, carbon and phosphorus have lower electronegativity.
- When 2 atoms on different ends of the electronegativity spectrum share electrons in a covalent bond, the electrons will be more attracted to the atom with higher electronegativity. Electrons have a negative charge, causing the atom with higher electronegativity to have a partial negative charge (δ−) and the atom with a lower electronegativity to have a partial positive charge (δ+). This unequal sharing of electrons creates a polar covalent bond.
- When atoms with similar electronegativities form covalent bonds, the electrons are shared fairly equally between the atoms. These bonds are considered non-polar.
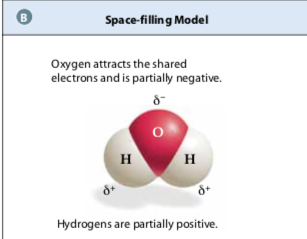 2 hydrogen atoms each share a pair of electrons with oxygen, forming covalent bonds in H2O. Since Oxygen is more electronegative than hydrogen, there is a partial negative charge on the oxygen and a partial positive charge on the hydrogens.
2 hydrogen atoms each share a pair of electrons with oxygen, forming covalent bonds in H2O. Since Oxygen is more electronegative than hydrogen, there is a partial negative charge on the oxygen and a partial positive charge on the hydrogens. Interactions Between Molecules:
- Intermolecular (occurring between atoms of different molecules) forces may form between molecules or even different parts of the same molecule if the molecule is very large.
- Intermolecular forces are much weaker than intramolecular ones. They determine how molecules interact with each other and different molecules, making them of vital importance in biological systems.
- Intermolecular interactions are usually attractive forces, making molecules connect. However, because of their lack of strength, they can be broken fairly easily, if sufficient energy is supplied. As a result, intermolecular forces are responsible for the physical properties of substances.
- There are two types of intermolecular forces which are particularly important for biological systems: hydrogen bonding and hydrophobic interactions.
HYDROGEN BONDING:
- A water molecule has two polar O-H bonds making it a polar molecule. It has a slightly positive end and a slightly negative end.
- Slightly positive hydrogen atoms of one water molecule are attracted to slightly negative oxygen atoms of another water molecule. This is a type of intermolecular attraction called a hydrogen bond.
- Hydrogen bonds are weaker than ionic or covalent bonds.
- A hydrogen bond can occur between or within the same molecule.
- Since cells exist in an aqueous environment, it is very important for hydrogen bonding to occur between biological molecules and water.
- Although hydrogen bonds are weak, many hydrogen bonds added together can be very strong.
- Hydrogen bonds between molecules in cells help maintain the structure and function of the molecules. For example, the 3D shape of DNA (stores an organisms genetic information) is maintained by numerous hydrogen bonds. The breaking and reforming of hydrogen bonds play an important role in how DNA functions in the cell.
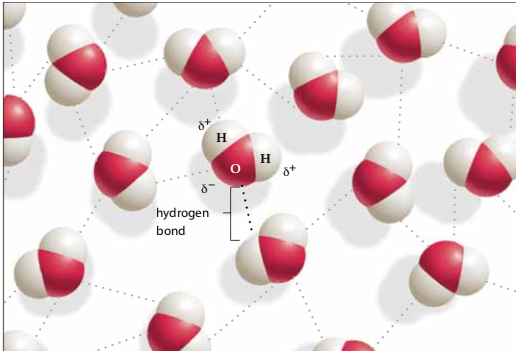 Hydrogen bonds are represented with dotted lines to distinguish it from a stronger covalent bond.
Hydrogen bonds are represented with dotted lines to distinguish it from a stronger covalent bond.HYDROPHOBIC INTERACTIONS:
- When non-polar molecules (motor oil, cooking oil, etc) interact with polar molecules, they don't mix. Instead, the non-polar molecules clump together. Hence the saying, "oil and water don't mix".
- If the molecules had human characteristics, it would be as if the non-polar molecules were frightened of the polar molecules.
- Thus, non-polar molecules are hydrophobic ("water-fearing") in their interactions with water molecules.
- Polar molecules, however, form hydrogen bonds with water, making them hydrophilic ("water-loving").
- The clumping together of non-polar molecules in water is referred to as the hydrophobic effect.
Ions in Biological Systems
- Atoms can obtain steady valence shells by gaining or losing electrons. They don't always have to share them.
- When an atom or group of atoms, gain or lose electrons, they acquire an electric charge and become ions.
- When atoms acquire a positive charge they become cations.
- When atoms acquire a negative charge they become anions.
- Ions can be composed of one element such as Na+ (sodium), or several elements, such as HCO3– (bicarbonate).
- Ions serve an important role in living systems. For example, hydrogen ions, H+, are critical to the biological process of cellular respiration. Sodium ions, Na+, are part of the transport mechanisms that enable specific molecules to enter cells.
- For biological processes in the cell, substances that form ions are never recognized as ionic compounds. Since the cell is in an aqueous environment, almost all ions are considered free. For example, sodium chloride (NaCl(s)) dissociates into (Na+(aq) and Cl–(aq)) since they dissolve in water.
Functional Groups Determine the Properties of a Molecule
- Organic molecules that are made up of only carbon or hydrogen atoms are called hydrocarbons.
- Properties of hydrocarbons include not dissolving in water, having relatively low boiling points, being flammable and being non-polar.
- Covalent bonds between carbon and carbon or carbon and hydrogen are "energy-rich".
- The breaking of these bonds creates a vast amount of energy. Thus, hydrocarbons are good fuels. They include acetylene, propane, butane and octane.
- Hydrocarbons share similar properties, but other organic molecules have a wide variety of properties because other atoms/groups of other atoms are attached to the organic molecules carbon-based structure.
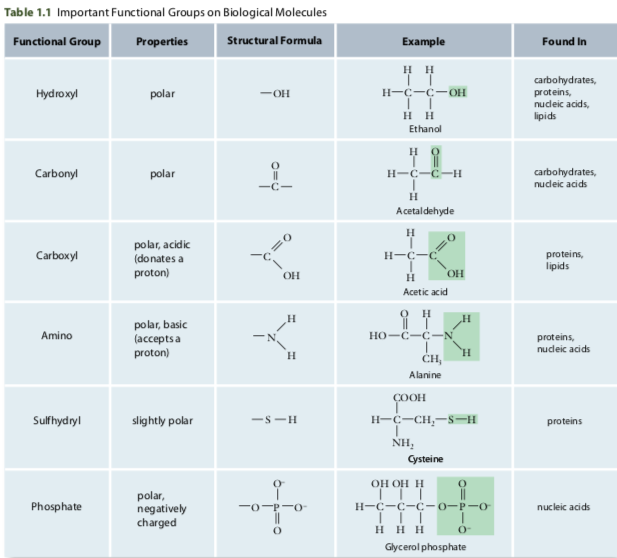
- A cluster of atoms that always behaves in a certain way is called a functional group. They contain oxygen, nitrogen, phosphorus or sulphur.
- Certain chemical properties are associated with certain functional groups. Hydroxyl or carbonyl groups found on a molecule will make the molecule polar. While, carboxyl will make a molecule acidic, resulting in it releasing a hydrogen atom for another molecule. Therefore, many functional groups participate in hydrogen bonding.
Structures and Shapes of Molecules
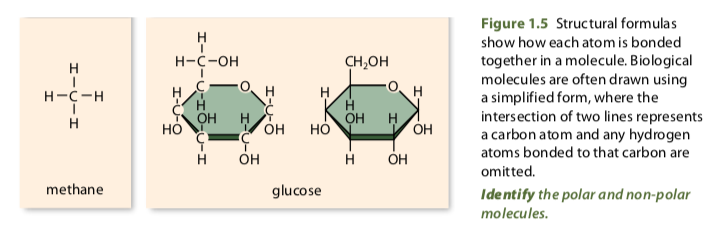
- Molecular formulas show the number of each type of atom in an element or compound.
- Structural formulas show how different atoms of a molecule are bonded together.
- When showing molecules using structural formulas, a single line is used to represent a single covalent bond, double lines represent a double bond and triple lines represent a triple bond.