Crystallization, recrystallization, melting point analysis and TLC.
Crystallization
- Crystallization is the formation of a solid in crystalline form from the gas or melt phase.
- The process occurs by either supersaturating a concentrated solution, lowering the temperature, or evaporating the solvent.
- In this method, most contaminants usually remain in the solution.
- Crystallization is often used as a purification step, e.g. in sugar production.
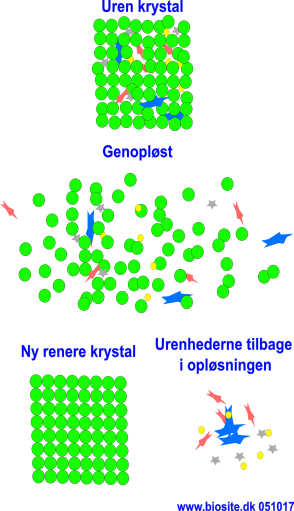
Recrystallization
Cleansing of crystalline matter.
- Redissolution and recrystallization.
- The purpose is to increase the purity of an already crystallized substance.
- Increasing the purity, the impurities remain in the mother liquor (i.e. the remaining solution).
Redissolve crystal by heating and subsequently restore by cooling.
The yield percentage decreases, as the mother liquor has a higher saturation of the desired substance.
Evaporation and cooling can crystallize out if recrystallization, i.e. that more of the desired substance comes in, but the impurity increases.
The crystals from the recrystallization can be dissolved again and returned to the primary crystallization.
Melting point analysis
- Melting point analysis is a method of determining the temperature point at which a substance changes from solid to liquid.
- The analysis is used to determine the purity of a sample.
- Impurities in the molecular lattice lower the melting point.
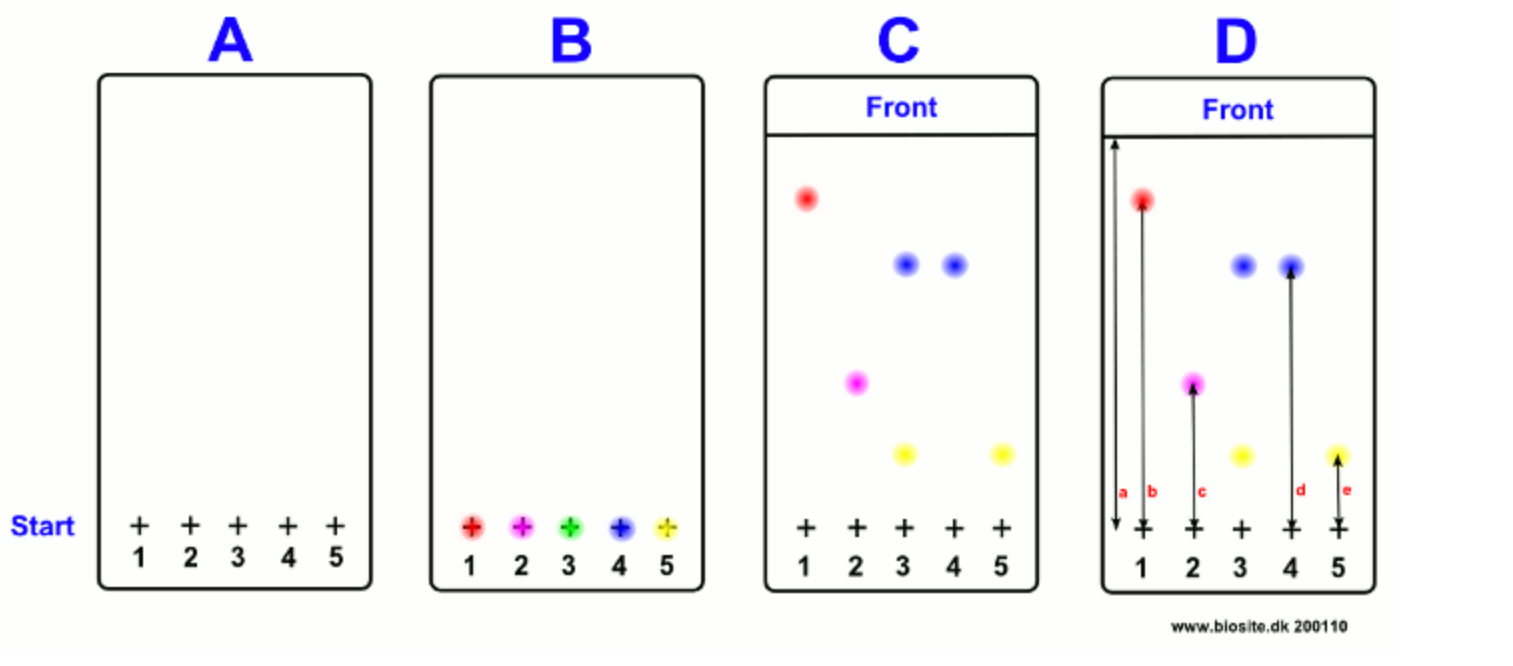
Thin Layer Chromatography (TLC)
Stationary: Stationary plate/paper
Mobile phase: movement running fluid
- TLC can be used on many different substance mixtures such as colour mixtures, amino acids, lipids, alkaloids and mycotoxins.
- Chromatographic separation technique where a plate is used either of glass or strong aluminium foil.
- The plate on one side is covered with a powder which has different properties in relation to which substance classes you want to separate.
- On the lower part of the plate, attachment points are marked, usually with a soft pencil, where a few drops of the substance mixtures are then placed with a micropipette.
TLC method (simplified).
- Apply a solution of the substances to be separated into the lower part of a plate.
- Also, apply standard solutions of known substances.
- Place the plate upside down in a beaker with a suitable solvent at the bottom.
- Wait for the solvent to move up the plate.
- Remove the plate when the solvent is approximately 1 cm from the top edge of the plate.
- Mark the position of the liquid front with a pencil.
- Use a beaker with a lid and filter paper to saturate the air with the solvent vapours.
- Analyze the coloured spots that have moved differently up the plate.
- Identify the different dyes in the mixture using the location of the standard solutions on the plate.
Result processing of TLC.
To compare the running length of the spots, RF values are used. (Relative to Front or retardation factor).
- The RF value is determined by measuring the running distance of the spots.
- The running distance is measured by measuring from the individual spot to the centre and then dividing this length by the length of the liquid front.
2D-TLC is used for more complex mixture systems.
- First, the TLC plate is developed in a solvent mixture which separates the polar components, and then the plate is dried.
- The plate is then turned 90 degrees and developed in a solvent mixture which separates the non-polar components of the mixture.