Free Radical Substitution
Free Radical Substitution
Alkanes are interactive molecules due to it being:
Non-polar: because carbon and hydrogen atoms have a very similar electronegativity.
Bonds are relatively strong: takes a lot of energy to break.
Free radical is any special with an unpaired electron. The unpaired electron is represented by a dot. Radicals are highly reactive species.
Free Radical substitution involves:
halogens
Occurs in the presence of UV light
Halogens form free radicals which substitute one or more hydrogen atoms
Three steps: initiation, propagation and termination
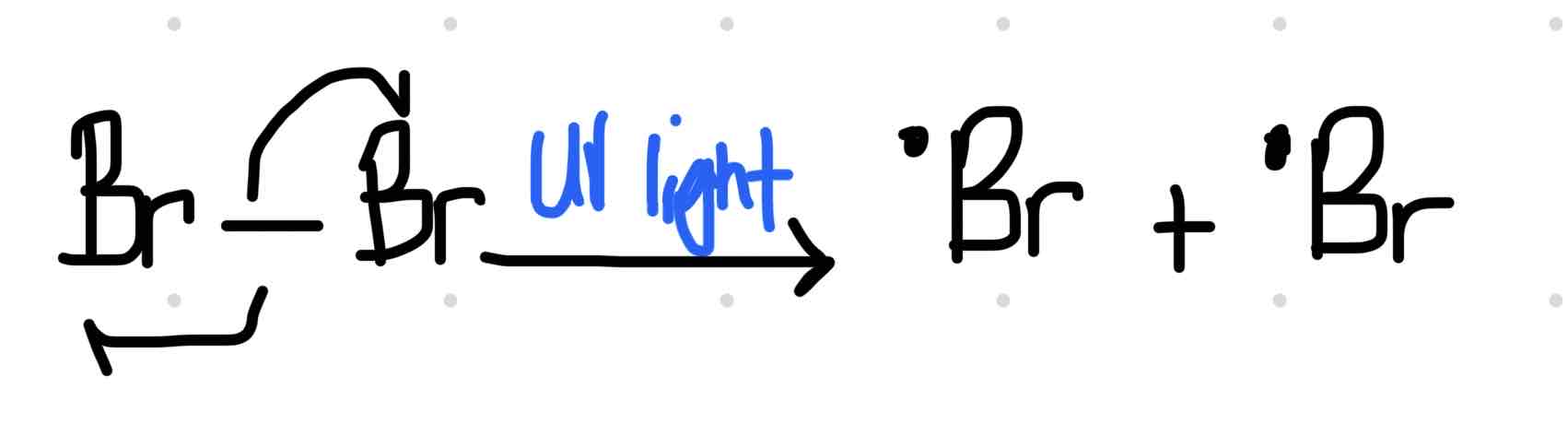
Initiation
Formation of free radicals, due to the presence of UV light, which causes the single covalent bonds to break by homolytic fission. A single covalent bond consist of a pair of electrons. When the bond breaks like this one electron goes to the each halogen atom.
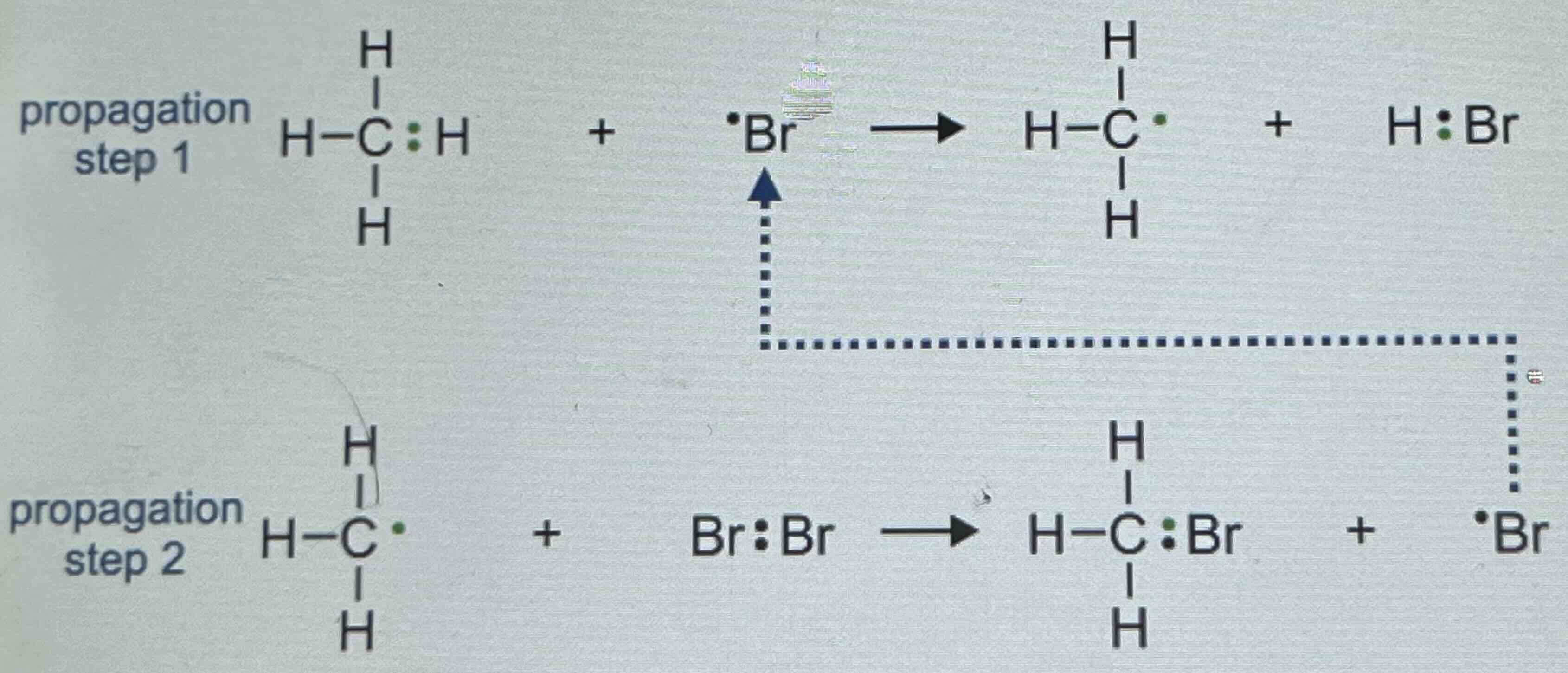
 Propagation
Propagation
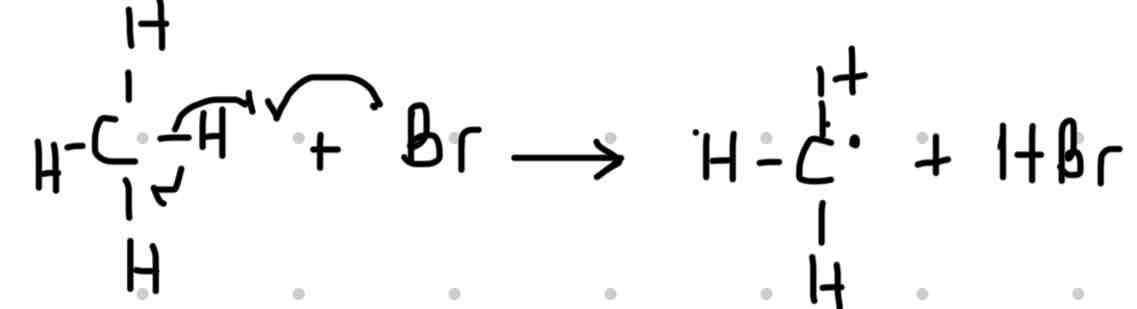
Step 1: Bromine free radicals which is highly reactive reacts with a methane molecule. The é in the bromine free radicals takes the hydrogen atom plus one electron. This reaction produces hydrogen Bromide and methyl free radicals.
 Step 2: The methyl free radicals reacts with a bromine molecule. This produces bromethane plus another bromine free radical.
Step 2: The methyl free radicals reacts with a bromine molecule. This produces bromethane plus another bromine free radical.
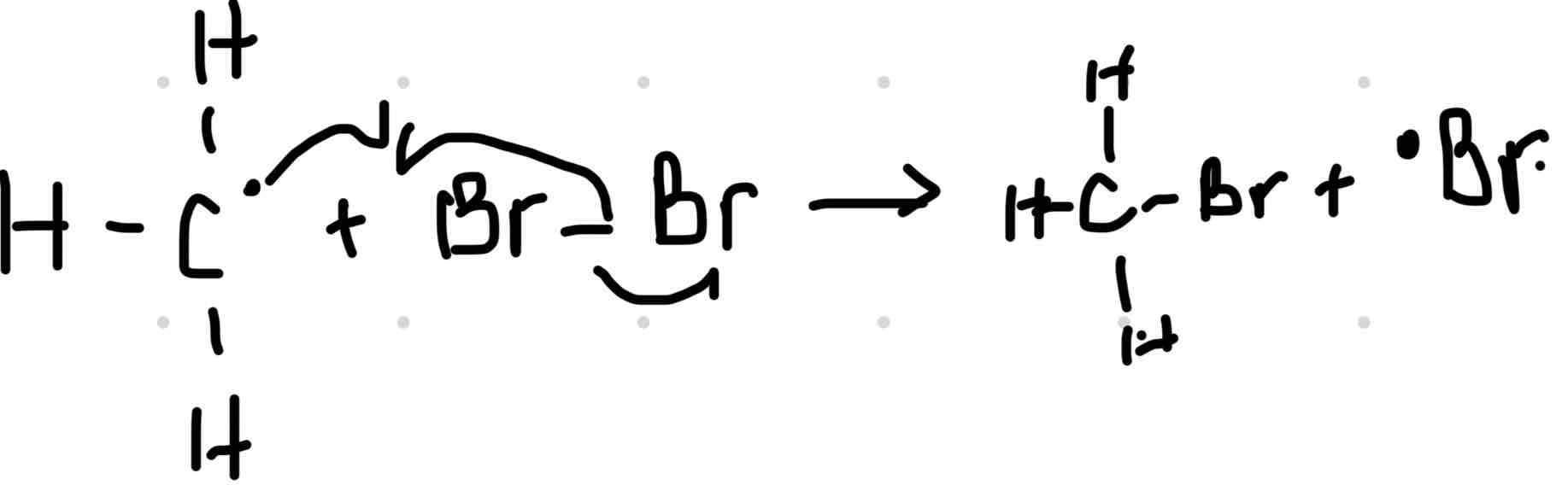 Propagation Steps 1 and 2 can chain reaction is formed. The bromine free radicals takes formed in Step 2 can go back and react with methane in step 1. This reaction will continue until final stage takes place which is called termination. Once a radical is present, this stage is a continuous process.
Propagation Steps 1 and 2 can chain reaction is formed. The bromine free radicals takes formed in Step 2 can go back and react with methane in step 1. This reaction will continue until final stage takes place which is called termination. Once a radical is present, this stage is a continuous process.
Termination
Two free radicals reacts with each other to form a molecule with no unpaired reaction. This is now a stable molecule and no longer takes place in the reaction.
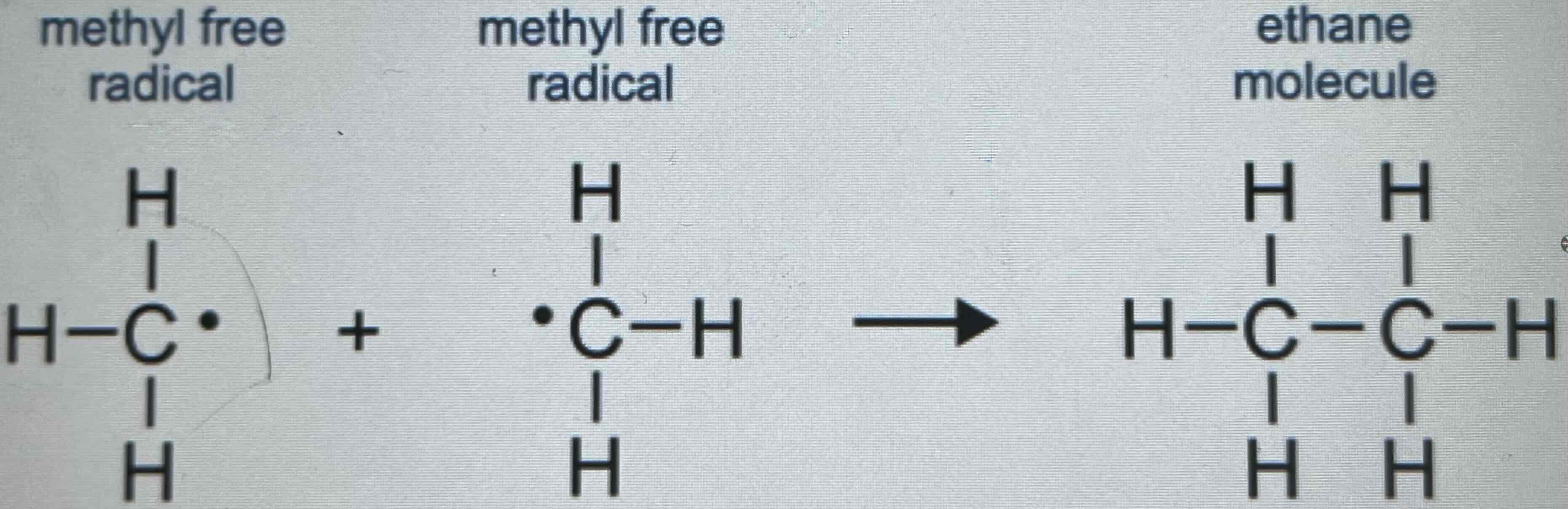
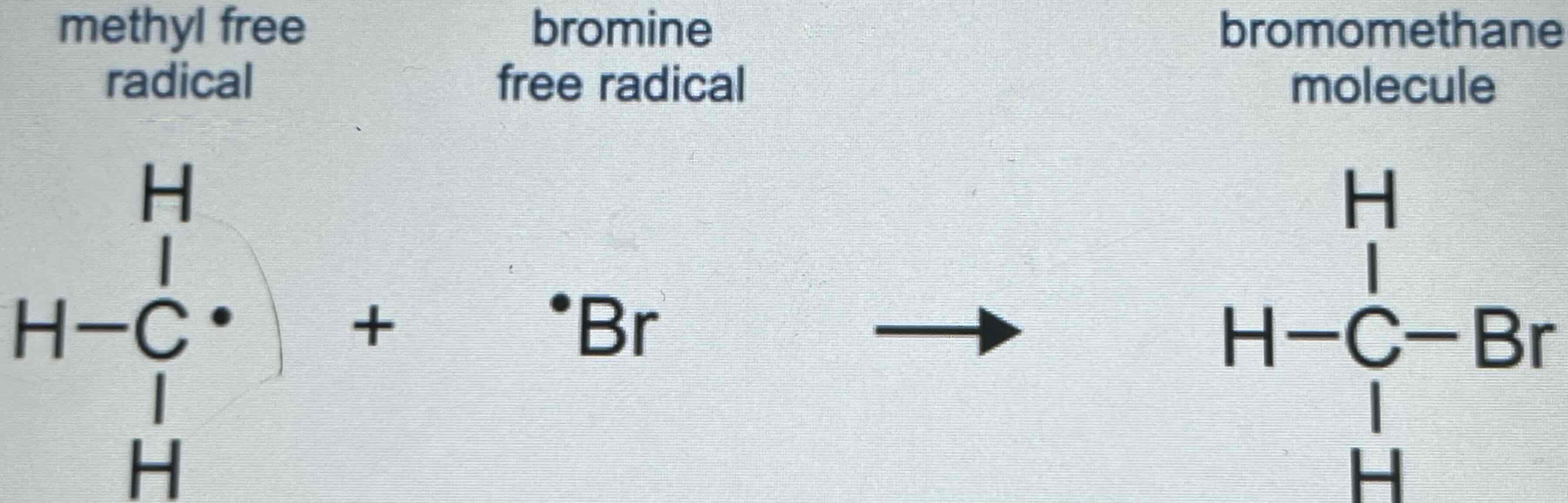
Three Possible Reactions in Termination
1. Two bromine free radicals can form a bromine molecule.

2. Two methyl free radicals can form a molecule of ethane.
3. A methyl free radical and a bromine free radical can form a molecule of bromethane.
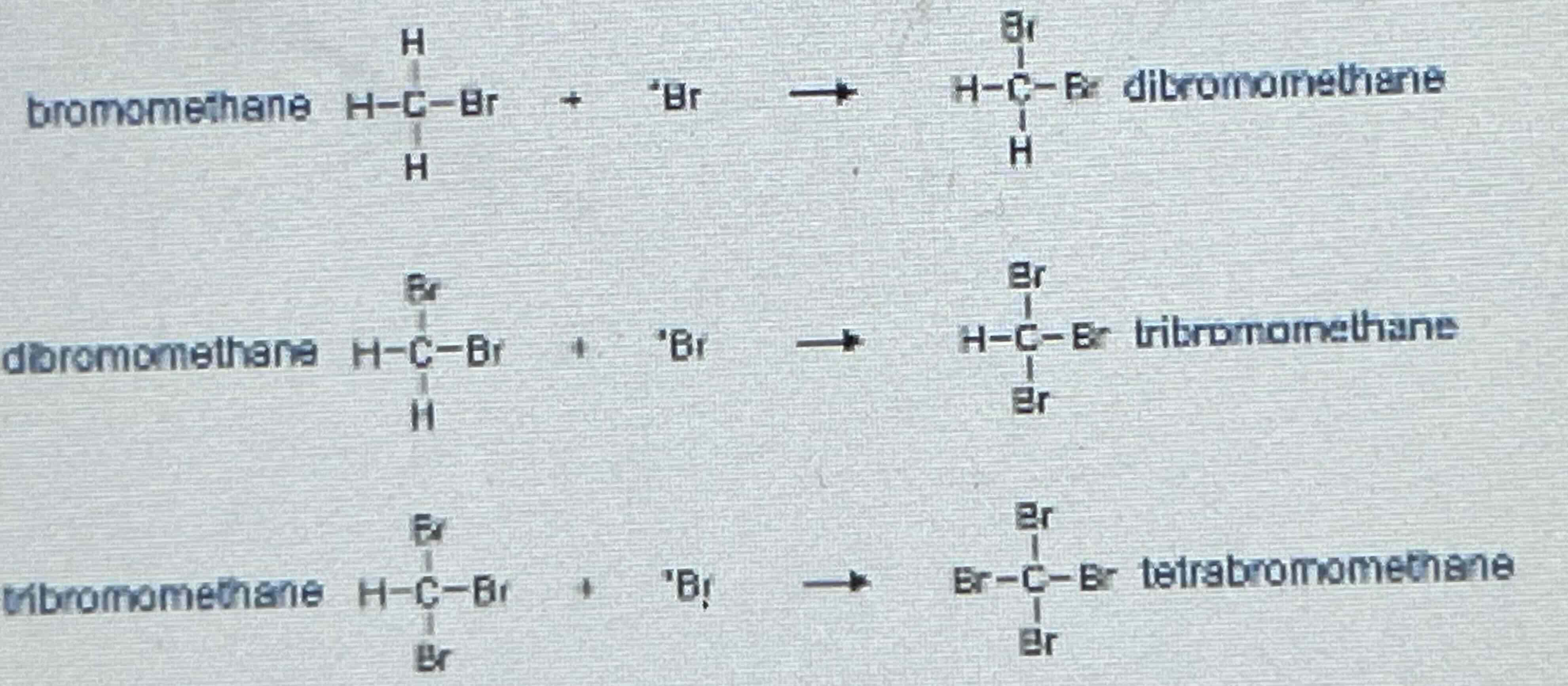
Side Products of Free radical substitution