Yr 1 - Physical
Summary and key points of CH1:
Within nucleus there is a strong nuclear force that acts in short distances, and is stronger than electrostatic forces that occurs between protons and electrons
Relative atomic mass: Average mass of an atom relative to 12C, taking into account of naturally occurring isotopes
Mass spectroscopy: To determine the different isotopes that make up the an element [relative abundance (%) on y axis and m/z (mass to charge ratio on x axis)]
What conditions are put in place for mass spectrometry? The whole process is put in a vacumm to avoid the ions from colliding with air molecules
Ionisation: Electron spray (sample dissolved in volatile solvent, forced through hollow needle, and each particle gains a proton - takes place in gaseous state) OR electron impact when a higher energy electron is fired at an atom, knocking out one electron.
Acceleration: when the positive ions are attracted to a negative plate and are accelerated towards them (all with the same kinetic energy)
Ion drift: where positively charged ions move towards (attracted towards) negative plate (lighter ions travel faster, heavier ions travel slower) towards detector
Detection: Where each positively charged ion gains an electron (which causes a current to flow, that is proportional to the abundance)
Atomic orbitals are considered a cloud of negative charge
Ionisation energy = the amount of energy required to remove one mole of electrons from one mole of atoms in its gaseous state
Overall, IE depends on nuclear charge, distance between nucleus and outermost electrons and shielding.
Summary and key points of CH2:
Relative molecular mass => adding relative atomic masses of all atoms in molecule // Relative formulae mass of ionic compounds is used, because they don’t exist as molecules.
1 dm cubed contains 1000 cm cubed
Ideal gas equation:
PV = nRT
Pressure → pascals
Volume → m cubed
n → moles
R → gas constant (8.31 JK/mol)
T → temperature (Kelvin) => to get to degrees to kelvin +273
Emperical formulea, is the simplest whole number ratio of atoms of each element in a compound
Molecular formulea is the actual number of atoms of each element in one molecule of compound
Atom economy = mass of desired product (Mr of desired product)/ mass of reactants (Mr of reactants) X 100
Tells us in theory how many atoms are being wasted (how much of reactant atoms end up as product)
Percentage yield = Moles of product (mass of product)/ Theoretical moles (theoretical moles) X 100
Yield tells us the practical efficiency of process
practical process of reaction [using equipment, some of product left in equipment]
Maybe reaction didn’t go to completition
Reversible reactions
Summary and key points of CH3:
Ionic bonding: Ionic compounds = Ionic lattice, when electrons are transferred between atoms to form ions and these ions are attracted together by electrostatic forces of attraction. (giant structures = high melting point, conduct electricity in molten state ions free to move, brittle and shatter easily because when struck this distorts the layer of ions, causing like ions to interact and repel)
Covalent bonding: Share pair of electrons with other atom [Giant covalent = macromolecules Vs simple covalent]
Co-ordinate bonding (dative bonding): when one atom provides both electrons, one atom accepts a pair of electrons (electron deficient) and one atom provides a pair of electrons (lone pair)
Dative bonding is represented with an arrow from the lone pair to the electron deficient pair (with all bonds the same length and strength + a charge is written on top)
Metallic bonding: Metallic latice of positive ions and a sea of delocalised electrons.
Good conductors of electricity (sea of delocalised electrons move)
Good conductors of heat (sea of delocalised electrons + the positive ions that vibrate vigourously)
The greater number of delocalised electrons = stronger electrostatic force of attraction, the smaller size of ion, the closer electrons are to positive nucleus’
BRITTLE: When the metal is struck, this causes the ions to move position such that ions with like charges are next to each other, and this causes repulsion
MALLEABLE: Metals contain giant lattice arranged in regular layers, these ions are surrounded by delocalised electrons, so when draw the layers can slide over each other and the delocalised electrons move around next to the positive ions
Electronegativity: is the power of an atom to attract a pair of electrons towards itself in a covalent bond.
Electronegativity depends on the nuclear charge, the distance between the outermost shell and nucleus, shielding of electron shells
Electronegativity links to polarity, with larger difference in polarity between two atoms (greater polarity = greater ionic characteristics)
Intermolecular forces of attraction:
Van der vaals (all molecules, depends on the random movement of electrons)
Dipole-dipole (depends on the polarity between different atoms)
Hydrogen bonding (O, F, N)
Summary and key points of CH 4:
Exothermic reactions release energy to the surrounding (- enthalpy, and MEXO => Making bonds is exothermic)
Endothermic reactions take in energy from surroundings (+ enthalpy, and BENDO => breaking bonds is endothermic)
Enthalpy = the heat change measured at constant pressure (standard conditions => 100kPa, 1mol/dm3, 298K)
For example, it may be strange to say heat change under constant temperature, this means measuring the enthalpy of the starting reactants at 298K and waiting until the reaction comes back to 298K
For example, pressure => the heat given out when gas is formed is used to push away the atmosphere, so if the pressure is higher than atmospheric pressure, then more energy required to remove the air particles, so less heat change
Standard states (S, L, G) => since more energy is required to change to turn liquid to gas.
Standard enthalpy of formation: the amount of energy required to form one mole of substance from its constituous elements in standard states, under standard conditions.
Standard enthalpy of combustion: the amount of energy required to burn 1 mole of fuel in complete oxygen (complete combusion) under standard conditions.
Heat => Total energy of all particles, energy of every particle - dependent of all particles // Temperature => the average kinetic energy of particles, independent of the amount of particles
Enthalpy change: But remember final answer in kJ/mol
Enthalpy change (J) = mass (grams) X SHC (J/ K g) X Change in temperature (K)
Flame calorimeter: has spiral chimney made from copper (copper can provide high thermal conductivity), flame is enclosed (reduces effect of convection), flame burned in pure oxygen rather than air (allow for complete combustion)
Polystyrene cup, good insulator prevents heat loss, by plotting a cooling curve (measure the temperature 4 minutes before, add the other reactant measure the temperature, then draw two lines of best fit, extrapolate, so can find the temperature rise at the point of adding)
Hess law states that the enthalpy change of a chemical reaction is the same whatever the route taken from the reactants to products.
Bond dissociation enthalpy: the enthalpy change to break covalent bonds with all species in gaseous state.
Mean bond Enthalpy: The average value of bond dissociation enthalpy for a given type of bond taken from a range of different compounds
Summary and key points CH5: Kinetics, the study of the factors affecting rates of chemical reactions.
Collision theory: for a reaction to take place two particles must collide with enough activation energy and with correct orientation.
Factors effecting rate of reaction: temperature, pressure, concentration, presence of catalyst, increasing surface area.
Activation energy: Minimum amount of energy required to start a reaction.
The species that exists at the top of the curve on enthalpy diagram is called transition state (activated complex)
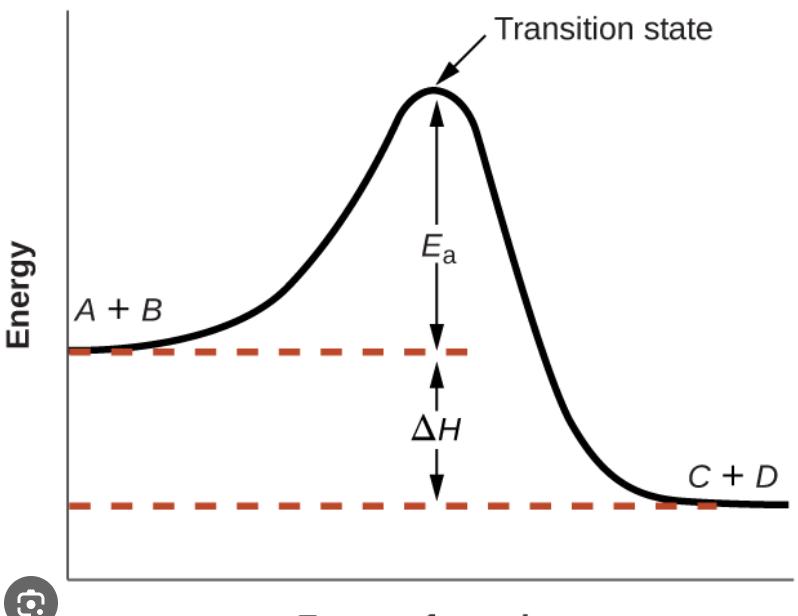
Everything about Maxwell-Boltzman distribution: Tells the distribution of energy among particles.
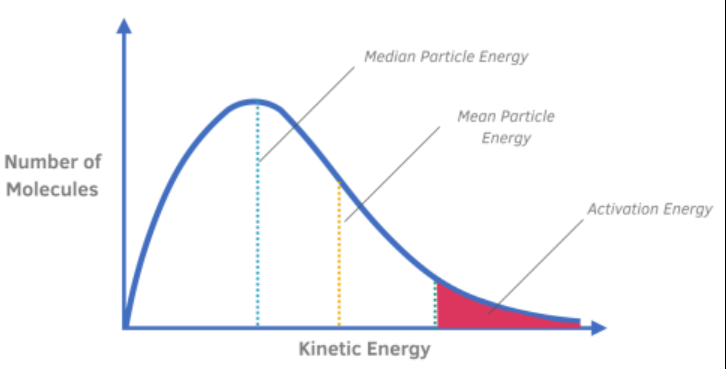
No particles have zero energy // Most particles have energy around peak (median particle energy) // Median particle energy and mean energy is not the same // The area under the curve represents the number of particles
With higher temperatures, the curve will be smaller and shifted to the right => so more particles with activation energy so the rate of reaction increases (more successful collisions take place)
With a catalyst present this lowers the activatation energy, so greater proportion of particles with activation energy (increasing the rate of reaction)
Catalytic converters: catalyst forms weak bonds with the metal atom (absorption, holding the gases in right position for them to react) // desorption, where the metal atoms break away from the catalysts.
Summary and key points of CH6:
Dynamic equilibrium: the rate of the forward direction is equal to the rate of backward direction
When Equilibrium is establised all the reactants don’t have to have same concentrations they can have varied.
Le chatelier’s principle: If a system in equilibrium is disturbed the equilibrium moves in the direction that reduces the disturbance
Catalyst has no effect on the position of equilibrium (increases the rate of forward and backward reaction)
Equilibrium constant: Kc (concentration in mol/dm cubed)
A + B → C + D
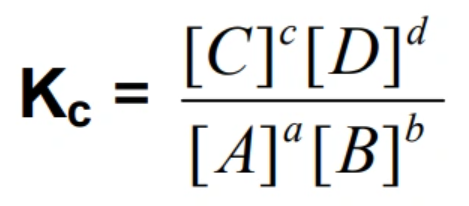
Kc is only effected by temperature AND Kc > 1 products dominate over reactants AND Kc < 1 reactants dominate over products
Summary and key points of CH 7: OILRIG
Oxidation states rules:
Uncombined elements have oxidation state of 0
Hydrogen is +1, except in metal hydrides (NaH) => where group 1 or group 2 with hydrogen bonded.
Group 1 is always +1
Group 2 is always +2
Aluminium always +3
Oxygen is -2, except in peroxides where it is -1 (H2O2), and OF2 where it is +2
Flourine is always -1
Chlorine is -1, except when in compounds with F and O it has positive oxidative state

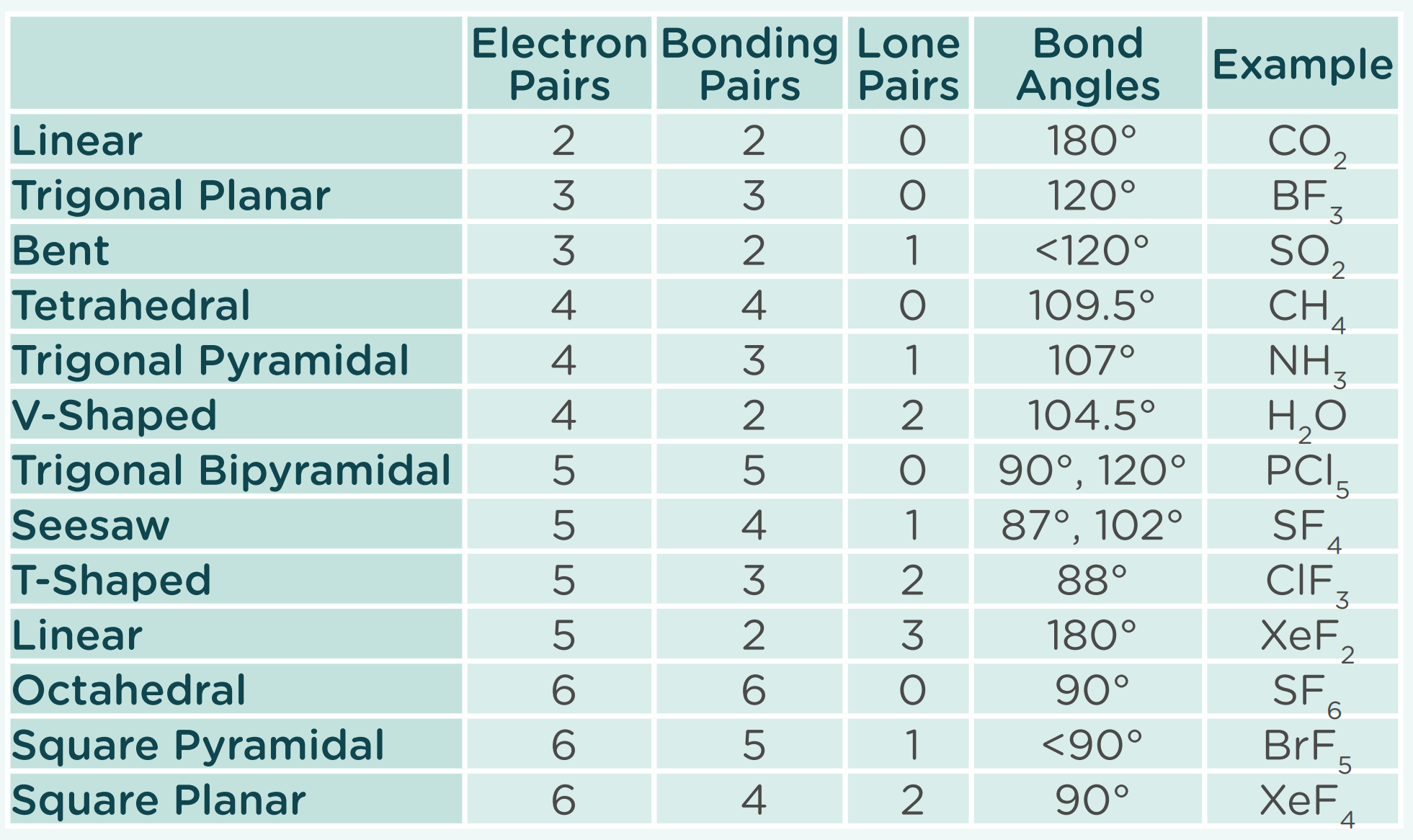
Linear x2
Trigonal planar
Bent
Tetrahedral
Trigonal pypiramidal
v-shaped
trigonal bipyramidal
seesaw
T-shaped
Linear
Octahedral
square pyramidal
square planar
2BP 0LP
3BP 0LP
2BP 1LP