stoichiometry
formulas
the piramids
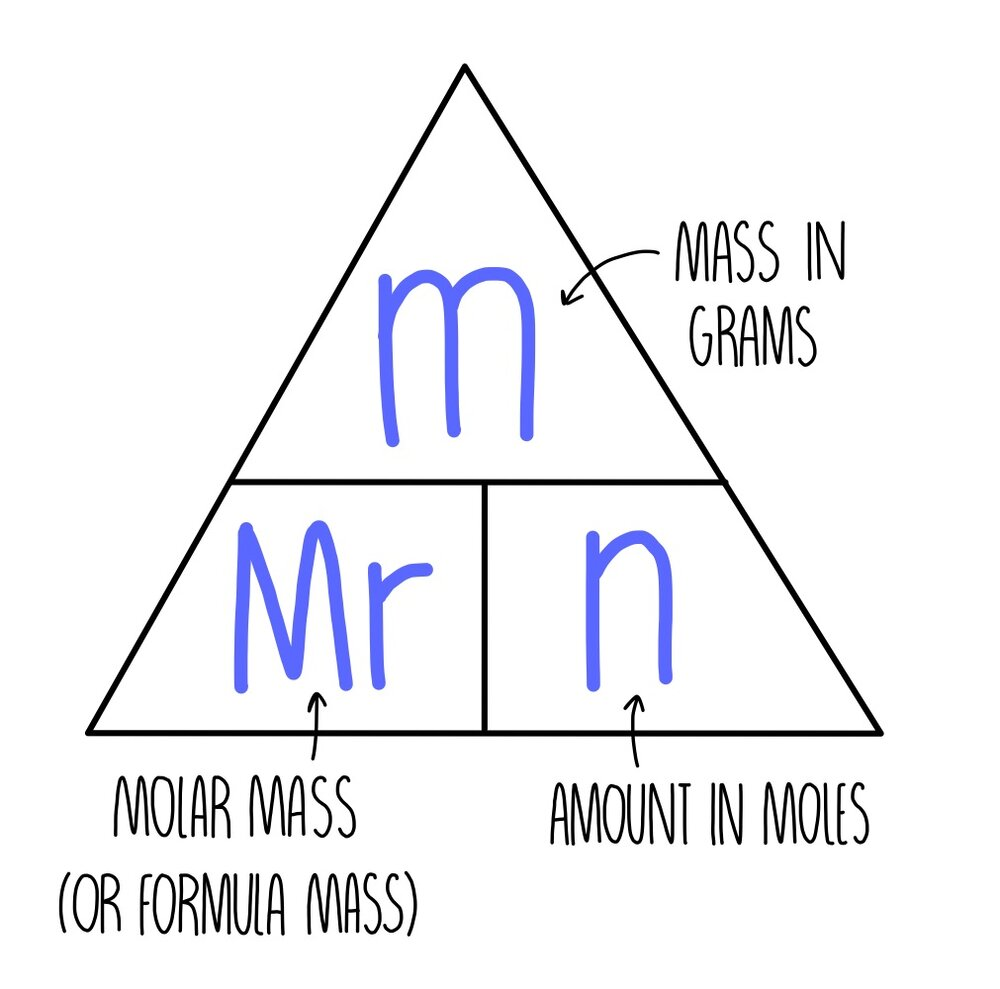
n = m : M
n - number of moles
m - mass [grams]
M - molar mass (from periodic table)
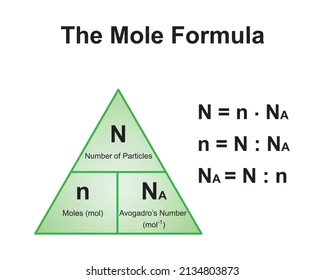
N = Nan → n = N : NA
N - number of molecules
NA - avogadros number ()
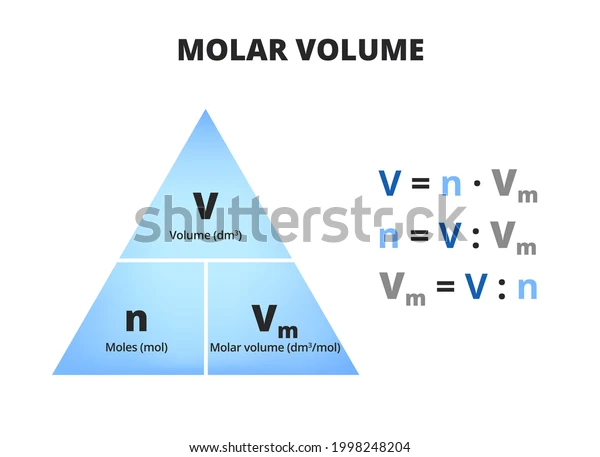
n = V : Vm
V - volume - mass : density
Vm - molar volume (a volume occupied by 1 mole) - in stp its ^^22,7^^dm^3/mol, in rtp its ^^24^^ dm^3/mol
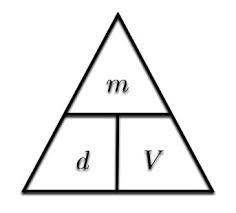
d = m : V
d = M : Vm
given temp and pressure
standard temperature and pressure (^^STP^^)
T = 0*C, 273K
Vm = 22,7dm^3/mol
P = 100kPa
room temperature and pressure ()
T = 25*C, 298K
Vm = 24dm^3/mol
P = 100kPa
empirical and molecular formula
empirical formula
- if % - change into grams
- grams into moles (m : M)
- divide everything you got by the smallest of the values
- round them up to whole numbers
- now put these numbers as the little ones
- done :grinning:
molecular formula
- find the M (molar mass) of the empirical formula
- divide the larger M by the smaller M
- divide the little ones in empirical formula by the number that you get
- and these are the new little ones
- done :grinning: