Paper 1
Atomic structure
The nucleus of an atom consists of protons and neutrons surrounded by electrons. The outer shell holds electrons
Proton or electron number = atomic number
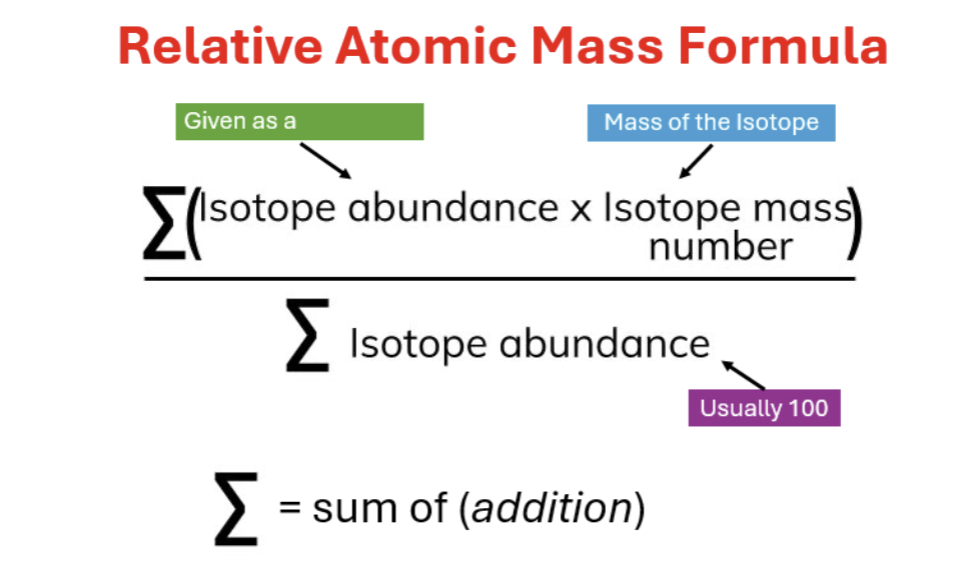
Nucleon number (proton + neutrons) = relative mass number
Number of neutrons = mass number - atomic number
Isotope = when an atom of the same element has the same number of protons and electrons but has different number of neutrons
Nuclear and Atomic diameters
The typical size of an atom is used to calculate the nuclear radius (nuclei radii) using R = R0 A^ (1/3). (r0 = is a constant and A= nucleon number) → average radius of a nucleus
The mass number (A), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus.
R0 = 1.2×10^-15
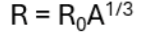
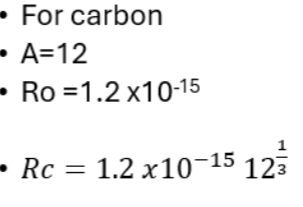
Electron configuration
Surrounding the nucleus are energy shells containing electrons.
The shells can be broken down into sub-shells - called orbitals.
These orbitals are called s,p,d,f and have different energy levels.
Electrons fills up these orbitals.
What is the max number of orbitals and electrons can they hold?
S - Orbital → 1, Electron → 2
P - Orbital → 3, Electron → 6
D - Orbital → 5, Electron → 10
F - Orbital → 7, Electron → 14
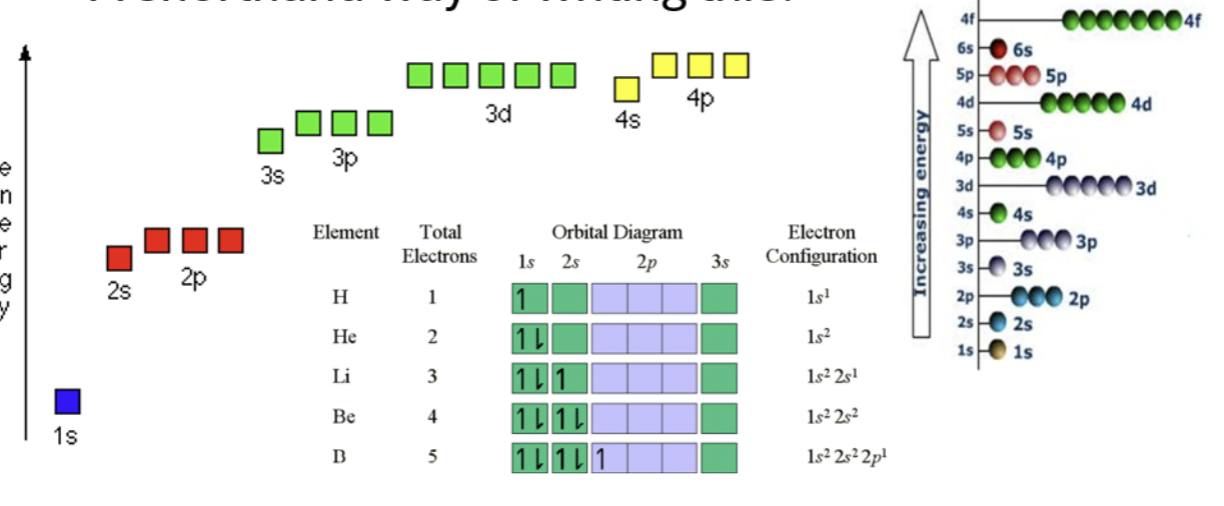
What is the max number of electrons that can be held on ELECTRON SHELLS?
Electron shell = 1 → Electrons = 2
Electron shell = 2 → Electrons = 8
Electron shell = 3 → Electrons = 18
Electron shell = 4 → Electrons = 32
Electron shell = 5 → Electrons = 50

Atomic Radius
a trend in atomic radius in group 2 and 3 and down a group, in attraction of proton numbers (atomic number) and electron shell.
The atomic radius decreases from left to right across a period
The atomic radius increases from top to bottom within a group. As the atomic number increases as you go down the group, there is an increase in positive nuclear charge.
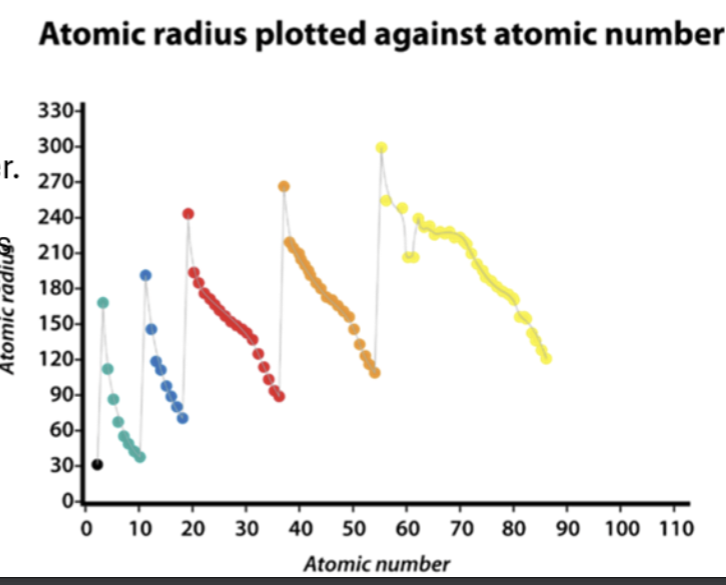
Each successive period is shown in a different period.
As the atomic number increases within a period, the atomic radius decreases.
Ionic bonding
Positive and negative ions form when a metal reacts with a non-metal, by transferring electrons. The oppositely charged ions are strongly attracted to each other, forming ionic bonds.
Structure of ionic compound
Giant lattice
neat row of alternately charged ions
strong electrostatic attractions act in all directions
Key properties of ionic compound
high melting and boiling point
no electrical conductivity as solid, conducts electricity when molten or in aqueous solution
high solubility in water, no soluable in organic (non polar) compounds
soluable → dissolve in a solvent to form a homogeneous mixture
Insoluable → does not dissolve in solvent therefore remains as seperate particles
Ionic bonding → the transfer of electrons
Covalent bonding → the sharing of electrons
Colavlent bonding
Covalent bonds form between non-metal molecules
A covalent bond is when two non-metal atoms share a pair of electrons
Dative Covalent bond
It has a lone pair of electrons (non binding pair of electrons)
Sharing of electrons but the electron come from the same element/atom
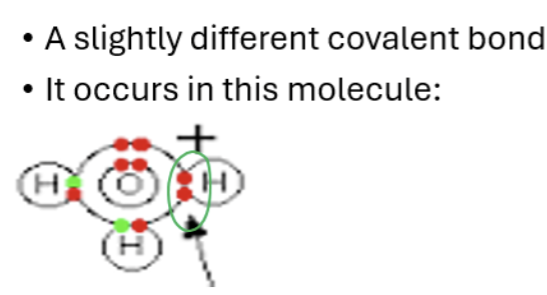
The ammonia molecule
Ammonia ions ,NH4+, are formed by the transfer of a hydrogen ion from the hydrogen chloride to lone pair of electrons on the ammonia molecule.
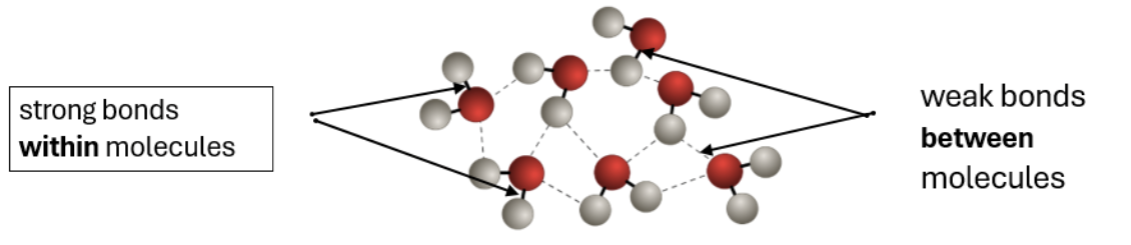
Intermolecular forces
The covalent bonds within molecules are STRONG
The forcs between molecules are weak and easy to break
Little energy is needed to overcome this forces therefore it has low melting and boiling points
Intermolecular forces are stronger between larger molecules
Metallic bonding
High melting and boiling points
conduct electricity and heat
malleable and ductile
Metallic molecules can be mixed together to make alloys
An alloy is a mixture of two or more elements, where at least one element is a metal. Many alloys are mixtures of two or more metals.

Types of mixtures
Solutions, e.g. salt and water
-a homogeneous mixture of solute and solvent
-homogeneous is even distribution
Suspensions, e.g. oil and water
-a suspension in a mixture of water and not dissolved material
-not a solution - heterogeneous mixture is temporary parts do not mix evenly.
Colloid
A colloid is one of the three primary types of mixtures, with the other two being a solution and a suspension. A colloid is a mixture that has particles ranging between 1-1000 nanometers in diameter, yet are still able to remain evenly distributed throughout the solution.
These are known as colloidal dispersions because the substances remain dispersed and do not settle to the bottom on the container.
Classifying colloids
Sol → is a colloidal suspensions with solid particles in a liquid e.g. blood, pigmented ink, cell fluids, paint, antacids, mud
Emulsion → is between two liquids that usually do not mix together - immiscible e.g. oil and water
Foam → is formed when many gas particles are trapped in a liquid or solid e.g. shaving foam, whipped cream
Aerosol → contains small particles of liquid or solid dispearsed in a gas e.g. natural fog, mists, dust
Gels → the liquid has become viscous enough to behave more or less as a solid e.g. jelly
Solution
they are clear and transparent as light passes through with no scattering
Homogeneous so does not settle out
Molecule in size
Cannot be filtered due to size
Can be separated using the process of distillation
Colloid
is intermediate between a solution and a suspension
Cloudy but uniform
light passing through a colloidal dispersion will be reflected by the larger particles and the light beam will be visible
Homogeneous so does not settle out
Particleds are 10-1000 angstroms
Will not be separate out
Suspension
Cloudy
Heterogeneous, at least two substances visible
Particles are larger than 10,000 angstroms
size allows particles to be filtered
If allowed to stand the particles will separate out
Oxidation and reduction:
Oxidation and reduction (redox) reactions in terms of:
loss and gain of oxygen
loss and gain of hydrogen
loss and gain of electrons
to include overall balanced ionic equations
Oxidation is the loss of electrons. In terms of oxygen transfer, oxidation may be defined as the chemical process in which a substance gains oxygen or loses electrons and hydrogen. When one of the reactants is oxygen, then oxidation is the gain of oxygen. Reduction is a loss of oxygen and gain of electrons.
Oxidation
Is
Lose
Reduction
Is
Gain
Addition Reaction and full balanced equation
in an addition reaction, the double bond of the alkene partially breaks when the reactant molecule attacks and adds on across it
Substitution
Alkanes undergo a substitution reaction with halogens in the presence of light. I.e. in ultraviolet light, methane reacts with halogen molecules such as chlorine and bromine
This reaction is a substitution reaction because one of the hydrogen atoms from the methane is replaced by a bromine atom
Alkanes
consists of chains of carbon atoms surrounded by hydrogen atoms
Each carbon atom is bonded to 4 other atoms
Each hydrogen atom is bonded to 1 carbon atom
All atoms have formed bonds with as many atoms as they can - called saturated
Naming - all alkanes end with -ane
Alkenes
they are hydrocarobons
The alkenes form a homologous series (gradual variation in physical properties, such as their boiling points and have similar chemical properties)
They have one carbon-carbon double bond (C=C) - functional group
they are unsaturated
general formula: CnH2n
Alkynes
They are hydrocarbons
The alkenes form a homologous series (gradual variation in physical properties, such as their boiling points and have similar chemical properties)
They have one carbon-carbon triple bond (C≡C) - functional group
They are unsaturated
General formula CnH2n-2
Molecular formulae e.g. C4H10
Empirical formulae - the simplest ratio e.g. C2H5
General formula - simplest algebraic formula to represent all member of a homologous series e.g. CnH2n+2
Displayed Formula - shows the relative position of all the atoms in a molecule
e.g.
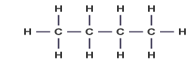
Structural formula - uses the smallest amount of detail necessary to show the arrangement in a molecule
e.g.
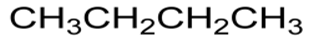
Skeletal formula - a simplidied organic formula with only carbons and functional groups
e.g.
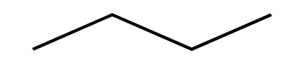
Aldehydes
Aldehydes and ketones as carbonyl compound. Aldehydes and ketones are simple compounds which contain a carbonyl group - a carbon-oxygen double bond.