Lecture 1_Part 1
Course Context and Real-World Relevance
Biotechnology and Society: a look at how modern biotech reshapes medicine, food, environment, and our concepts of life itself.
Course logistics mentioned:
Brightspace site hosts materials for the first lecture; read the syllabus.
Questions and responses forum and email for help; asynchronous office hours; contact info provided.
Emphasis: biotechnology is not just lab work; it has broad real-world impact and ethical dimensions.
Intracytoplasmic Sperm Injection (ICSI) and CRISPR in Embryos
Intracytoplasmic Sperm Injection (ICSI) procedure: fertilization of a human egg by injecting a single sperm directly into the cytoplasm with a fine glass pipette; egg held in place during injection.
Comparison to conventional IVF: IVF relies on sperm swimming to the egg; ICSI bypasses this step.
the needle delivering not only sperm but also a CRISPR construct into the embryo at fertilization.
CRISPR overview:
CRISPR-Cas9 is a gene editing tool, often called "genetic scissors," enabling precise genome editing.
cut and modify DNA with precision
DNA sequences found in prokaryotes like bacteria and archaea
include identical DNA repeats
Origin: derived from bacterial immune system that defends against viruses;
bacteriophages: bacterial viruses
CRISPR sequences include repeats and spacers;
Cas9 is an enzyme used for cutting DNA.
edit DNA in living cells
Applications: model human diseases, identify key genes, switch genes on/off, modify animals/plants.
Significance: CRISPR editing at the germline (embryo stage) raises profound ethical debates about human evolution and inheritance.
CRISPR: clustered regularly interspaced short palindromic repeats
play a crucial role in bacterial immunity against viruses
enables precise genome editing
Sickle Cell Disease (SCD) and CRISPR Therapeutics CTX001
Sickle Cell Disease (SCD) background:
Genetic disorder from a mutation in the beta-globin gene, causing abnormal hemoglobin S (HbS).
Mutation: Glutamic acid at position 6 replaced by valine (Glu6Val, E6V) in the beta-globin chain;
leads to sickling of red blood cells and impaired oxygen transport and circulation
this mutation disrupts the shape and functionality of the red cells causing severe pain, frequent infections, and increased risk of of stroke due to obstructive blood vessels
CRISPR-based therapeutic approaches:
Direct correction: edit hematopoietic stem cells to fix the beta-globin mutation so normal hemoglobin is produced.
HbF induction strategy: boost fetal hemoglobin (HbF) to substitute for defective adult hemoglobin.
HbF is naturally elevated at birth but declines at 3 months; a gene called BCL11A represses HbF after birth.
Disabling BCL11A lifts HbF production, which can compensate for HbS.
CTX001: a CRISPR Therapeutics drug that disables BCL11A to increase HbF; developed with Vertex Pharmaceuticals.
Reported result: in trials, over 90% of edited blood stem cells were successfully edited, leading to a significant rise in HbF.
Victoria Gray case (first in the United States):
Victoria Gray was a pioneering patient who received autologous hematopoietic stem cell therapy using her own CTX001-edited cells.
Autologous: the cells belong to the patient themselves, minimizing immune rejection risks.
Procedure: Her bone marrow cells were first removed. These diseased cells were then edited ex vivo (outside the body) using CRISPR to disable BCL11A. After a conditioning regimen to make space, the edited cells were reinfused into her body during a transplant procedure.
Outcome: The therapy resulted in high and stable levels of HbF production, providing profound relief from the debilitating painful crises and other severe symptoms associated with SCD, marking a functional cure for her condition.
Two main routes of CRISPR therapy for SCD:
Repair the mutation directly in beta-globin gene (gene correction).
Upregulate HbF by turning off BCL11A, allowing HbF to substitute for defective HbA, providing a functional cure.
Broader implication: this work demonstrates a potential functional cure for SCD and suggests the approach could extend to other genetic diseases.
CRISPR Milestones, Embryo Editing, and Ethical Debates
Early research milestones:
2013: A pivotal year where CRISPR-Cas9 was first successfully used to generate precise mutants in various organisms beyond bacteria, proving its broad applicability. A key proof-of-principle study demonstrated correction of a dominant CryGC mutation causing cataracts in mice, leading to corrected, fertile mice that could transmit the edits to their offspring. This showed the potential for germline editing in animals
By zygote injection of CRISPR/Cas9, mice carrying desired mutations can be generated in one step
Mice with mutations in the Crygc gene or dystrophin gene (Dmd) that cause cataracts or Duchenne muscular dystrophy (DMD) can be corrected by co-injection of CRISPR/Cas9 targeting the mutant alleles into zygotes
2015: Chinese scientists, led by Junjiu Huang, published the first report of gene editing in nonviable human embryos (specifically targeting the gene responsible for beta-thalassemia).
attempted to correct the beta thalassemia mutation using CRISPR
raised some major ethical concerns and revealed significant technical challenges highlighting that the technology
2017: An international team, including Shoukhrat Mitalipov, edited mutations in dozens of viable human embryos
targeting the MYBPC3 gene implicated in hypertrophic cardiomyopathy
mutation is dominant, meaning that the child just has to inherit only one copy of mutated gene either from the mother or the father to experience the effects
generated only one mosaic in an experiment, involving 58 embryos
This research, while also controversial, focused on addressing critical safety hurdles such as:
off-target mutations: unintended or unwanted genetic changes/edits in untargeted cells
mosaic: different embryonic cells contain different DNA sequence
the presence of both edited and unedited cells within the same embryo
showing improved precision but still demonstrating challenges.
The He Jiankui case (controversy):
Shenzhen team attempted to create gene-edited babies by disabling CCR5 using the gene editing technology CRISPR in an effort to prevent disease in newborns.
resistant to HIV, smallpox, and cholera
Claimed: his experiment aimed to disable the CCR five gene to make the twins resistant to HIV
Controversy: potential cognitive enhancement links to CCR5; ethical concerns about germline modification and heritability.
Legal outcome: He Jiankui was found guilty of illegal medical practices and sentenced to three years in prison; later returned to research in China.
returned to be his laboratory to work on the treatment of Alzheimer's and other genetic diseases
Ongoing ethical and regulatory landscape:
Germline editing for humans is illegal in at least 25 countries; Europe has long bans.
Proximity to Nazi-era history has shaped regulation and skepticism about improving the human species via genetic engineering.
UN (2015) called for a worldwide moratorium on germline modifications that could affect human dignity and future generations.
problem with modifying the germline is that this other line might not hold, and that's the line between therapy and enhancement
Public and scientific debate themes:
Safety and efficacy of germline edits; risk of unintended consequences and off-target effects.
Potential for unequal access and widening health disparities due to cost and availability.
Distinction between therapeutic (disease-correcting) edits and enhancement (traits beyond health).
Definitions and examples mentioned:
Unibaby: concept of creating sperm and eggs from somatic cells of the same person (germline derivation from somatic cells); not yet achieved in humans but demonstrated in concept via iPSCs.
The idea of using alternative sources for gametes (e.g., cow eggs with human DNA for early development studies) to study developmental biology; not a clinical practice.
Regulatory and ethical cautionary notes:
Germline editing remains highly controversial and tightly regulated; many countries ban it; the UN called for caution in 2015.
Debate continues about whether therapeutic benefits justify potential risks to future generations and society at large.
Somatic vs Germline Editing, Therapy vs Enhancement, and Involved Terminology
Key distinctions:
Somatic cells: body cells (skin, muscle, liver, etc.)
Edits affect only the treated individual; changes are not heritable.
the vast majority of cells in your body, blood brain, skin, muscle cells are all somatic cells
Germline cells: reproductive cells: sperm, egg, and very early embryonic cells
Edits are heritable and can affect future generations.
changes that ultimately will affect the human population and human evolution
Therapy: edits aimed at treating or curing a disease in the patient.
Everything that gets developed for therapeutic reasons almost always gets ends up getting used for enhancement
Enhancement: edits intended to provide advantages to healthy individuals (e.g., improved cognition or physical traits).
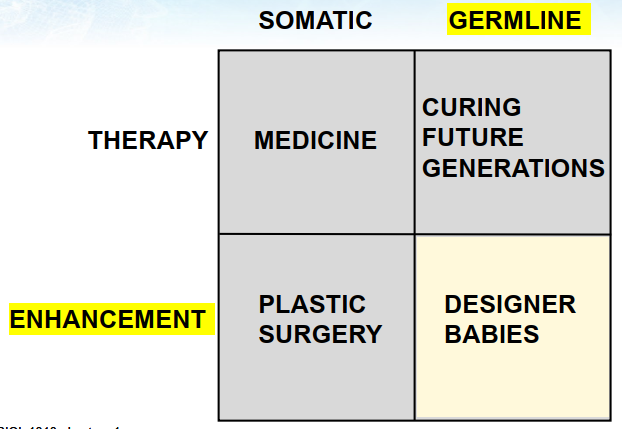
Somatic cell therapy: medicine
less controversial primarily because any change you make ends with that person
Somatic enhancement: plastic surgery
transhumanism: whole movement that supports this move to genetic enhancement
football players using steroids or human growth hormone or think of competitor cyclists using EPO to boost their red blood cell count or Michael Jackson before and Michael Jackson after
Germline cell therapy: curing future generations
editing to remove diseases in future generations
Germline enhancement: designer babies
change on the allele (eye colour, EPOE for Alzheimer, etc.)
allele: variant of a gene
Delivery challenges and target tissues:
Different diseases affect different tissues; some organs (like bone marrow) are more accessible for ex vivo edits.
Ex vivo approaches are currently the most promising for certain conditions (e.g., sickle cell, some cancers).
Germline editing regulation and debate:
Widespread bans in many countries; ongoing international debate about permissible uses and governance structures.
Public health considerations:
Potential to alter human evolution if germline edits become widespread; raises questions about consent of future generations.
Preimplantation Genetic Diagnosis (PGD) and the Unibaby Concept
Preimplantation Genetic Diagnosis (PGD) basics:
Embryos created via IVF are screened for disease-linked mutations before implantation.
only healthy embryos are implanted
PGD can test for analytical markers beyond disease (e.g., sex or disease risk).
As technology advances, full genetic profiling may become possible, akin to consumer DNA testing.
Distinguishing PGD from gene editing:
PGD screens or selects embryos but does not edit their genomes.
Doctors remove a cell from a three day old embryo to test for disease markers
doesn't cure disease, but it helps avoid disease
Polygenic risk scores and trait prediction:
Polygenic scores: they estimate they estimate traits based on genetic correlations across the genome
Predictions based on genetic correlations across the genome may estimate probabilities of traits (e.g., academic performance).
In the US/Canada, trait prediction in IVF is currently prohibited by law; future debates likely as capabilities improve
scientists may soon create human eggs from skin cells, a technique already successful in mice
Unibaby concept and IVF future:
Unibaby: envisions creating sperm and eggs from ordinary somatic cells (e.g., skin cells) to enable reproduction without traditional gametes.
iPSCs (induced pluripotent stem cells) can be reprogrammed to form gametes; progress shown in mice; human application remains experimental.
can potentially, form any tissue, including eggs and sperm
induced pluripotent stem cells: adult stem cells
Advances and challenges in gamete creation:
Developing eggs or sperm from skin cells could reduce invasive egg harvesting and enable large-scale embryo production, but raises ethical concerns about reproduction and selection.
Practical considerations:
IVF remains limited by egg harvesting difficulty and cost.
Advances in iPSC-derived gametes could transform reproductive options but require rigorous safety, ethical, and regulatory evaluation.
Ethical, Social, and Global Implications of Gene Editing
Equity and access:
Tools will likely be expensive and primarily available to the wealthy, potentially widening health and lifespan gaps.
Examples cited: sex-selective practices in some regions; Down syndrome prevalence reduction in Iceland due to prenatal testing.
Societal and philosophical questions:
Where to draw the line between therapy and enhancement? The line is often fuzzy due to polygenic traits, risk alleles, and context-specific values.
Potential for designer babies and consumer demand for non-medical enhancements.
Public trust and responsible science:
The stakes are high; premature or risky applications could erode public trust in biotechnology.
Balancing potential benefits (reduction of suffering) with risks and misuses requires careful governance and ongoing dialogue.
Notable scientists and recognitions:
CRISPR pioneers Jennifer Doudna and Emmanuelle Charpentier received the Nobel Prize in 2020 for CRISPR discovery;
raised fears about designer babies persist.
Practical guidance from the course perspective:
Critical thinking and humility are essential when evaluating new technologies.
The course aims to prepare students to discuss, analyze, and communicate biotechnology issues responsibly.
Course Learning Outcomes, Assessments, and Study Guidance
“Chance favors the prepared mind” by Louis Pasteur, the French microbiologist
emerged from his own work on vaccines
he developed vaccines for anthrax and rabies
It means that luck or chance opportunities are most useful to those who are ready to recognize and act on them
Learning outcomes (as defined for the course):
A learning outcome sets out what a learner is expected to know, understand and be able to do as the result of a process of learning
Describe key historical events and figures in biotechnology development (from fermentation to genetic engineering).
Define and explain fundamental biotechnology concepts underpinning the field.
Explain common biotechnology techniques and tools (e.g., PCR, cloning, CRISPR) in plain language.
Evaluate biotechnology's role in health, agriculture, and industry via real-world case studies (GMOs, gene therapies, industrial enzymes, biofuels).
Analyze ethical, legal, and social issues raised by biotechnology; discuss policy implications.
Develop skills to research, interpret, and communicate scientific information effectively.
Course organization and delivery:
Lectures and slides online; readings and videos online; self-assessments included.
Brightspace used for all materials; optional textbook (Introduction to Biotechnology by Femen and Paladino, 4th ed) with online companion site containing practice quizzes.
11 online tests (one per week over 12 weeks; best 10 counted). Tests: mostly multiple-choice, some matching; one attempt per test; 24-hour window (12:01 AM to 11:59 PM).
Test results and feedback released the day after the test period closes; no final exam in this course.
Assessment philosophy and evidence:
Assessment is the engine that drives student learning.
Motivation and Focus
Feedback and Improvement
Instructional Guidance
Learning Measurement
Formative assessments: assessment FOR learning
Purpose: to increase quality
low-stakes, monitor learning, guide improvement (e.g., self-check quizzes, homework forums)
gather feedback that that can be used by the instructor and the students to guide improvements in the ongoing teaching and learning context
When the cook tastes the soup
Summative assessment: assessment OF learning
Purpose: to measure quality
high-stakes tests to evaluate whether learning outcomes are met.
see if you have achieved the learning outcomes or not
When the guests in the restaurant taste the soup, they're assessing the quality of the soup
Retrieval practice (testing effect) evidence: tested groups recall better than those who only reread; e.g., a Purdue study showing higher scores with retrieval practice (67% vs 27–49% across groups).
Important organizational tips:
Read the syllabus carefully; explicit learning outcomes guide exam questions and expectations.
Pro tip: complete self-assessments after readings/videos; questions may reappear on tests.
Keep up with readings; plan to study roughly two hours outside the hour of lecture.
Study strategies and recommended practices:
Use the Feynman method to master concepts: explain a topic simply as if teaching a beginner; identify gaps and re-study.
Join or form study groups; compare answers to develop understanding.
Maintain scientific values: curiosity, humility, love, freedom, honesty, respect, cooperation, tolerance; science as a human, iterative, evidence-based enterprise.
Core ethical and intellectual anchors (per Bronowski and the course):
“Every judgment in science stands on the edge or error, and is personal” (Jacob Bronowski)
Science is a human form of knowledge; knowledge is personal, responsible, and unending adventure at the edge of uncertainty.
Accept that all judgments in science are tentative and subject to revision in light of new evidence.
Practical reminders and classroom culture:
Follow Wheaton’s Law for online conduct (treat others with respect in forums - “don’t be a dick”)
Sleep and balance: student well-being supports learning.
Snoop Dogg motif: read the syllabus; it’s essential for success.
Quick recap of essential definitions and terms:
Somatic cells vs germline cells: somatic edits affect only the individual; germline edits affect future generations.
ICSI: intracytoplasmic sperm injection.
PGD: preimplantation genetic diagnosis.
Unibaby: hypothetical creation of both sperm and eggs from a single person’s somatic cells (not yet realized clinically).
HbF: fetal hemoglobin; BCL11A gene suppresses HbF after birth; disabling BCL11A can elevate HbF to compensate for defective adult hemoglobin.
E6V: Glu6Val mutation in β-globin; responsible for HbS in sickle cell disease.
MYBPC3: gene mutation associated with hypertrophic cardiomyopathy (risk of sudden death in young athletes).
CCR5: gene edit proposed for HIV resistance in germline editing attempts; controversy and legal consequences.
Quick Reference: Key Dates, Terms, and Concepts
Timeline snapshots:
2013: CRISPR-Cas9 used to generate mutants in various organisms; mouse model of cataracts from CryGC mutation demonstrated curative editing.
2015: Chinese researchers edit nonviable human embryos for beta-thalassemia mutation; ethical concerns and technical limitations highlighted.
2017: Editing of dozens of viable human embryos to correct mutations (MYBPC3) reported; focus on off-target effects and mosaicism.
2015 (UN): Call for worldwide moratorium on germline modifications affecting human dignity.
2020: Nobel Prize awarded to CRISPR pioneers Doudna and Charpentier for CRISPR discovery.
Notable numeric references:
CTX001: over 90% of edited hematopoietic stem cells achieved in trials; HbF upregulation observed.
58 embryos mentioned in a study on mosaicism; 1 mosaic observed (no off-target mutations reported in that study).
11 tests in the course; best 10 count toward the grade; one missed test does not incur a penalty beyond the loss of that one attempt.
Conceptual definitions:
Allele: a variant form of a gene; different alleles can influence disease risk or trait expression.
Polygenic scores: predictions of traits based on the combined effect of many genetic variants across the genome.
Final Takeaways
CRISPR and gene editing hold transformative potential for treating genetic diseases and shaping human biology, but carry profound ethical, social, and regulatory implications.
Somatic editing offers therapeutic potential with fewer ethical concerns than germline editing, which affects future generations and is widely restricted.
The field sits at the intersection of science, medicine, policy, and values such as equity, safety, and human rights; informed, critical discussion is essential.
Learning strategy emphasis in this course includes practicing retrieval, using the Feynman method to ensure understanding, and engaging with the material through ongoing discussion and reflection.
Homework
Define biotechnology.
Biotechnology is a set of tools that makes use of living things to solve a problem or develop a product.
Name and describe three ancient (B.C.E.) events that serve as examples of early Biotechnology.
Fermentation: This was used to create different products such as beer and wine
Selective breeding: Dogs were domesticated and bred in order to serve purpose for humans. The same could be said about certain crops that were grown.
Making of leavened bread: Unleavened bread was a common practice in bread making, until the discovery of yeast. This introduced a new way of making bread
Discuss the importance of Escherichia coli to biotechnology.
Escherichia coli contributed to one of the most ground breaking scientific discoveries and it is still being used in experiments in labs today. Due to its ability to reproduce quickly, it was perfect. It contributed in discoveries about DNA by allowing a strand of DNA to be cut at a specific point and make new DNA.
Discuss the contributions of certain scientists to biotechnology, namely:
Karl Ereky: He is known as the father of biotechnology, he coined the term in the early 1900s
Kary Mullis: He was the scientist that created PCR. This technology allows scientists to make multiple copies of specific DNA
Alexander Fleming: He discovered penicillin, an antibiotic that went on to save multiple lives
Stanley Cohen & Herbert Boyer: These scientists came up with recombinant DNA, otherwise known as rDNA. This allowed for the perfection of the cloning of DNA.
5. Define recombinant DNA technology and name 3 molecular applications and 3 agricultural applications.
Recombinant DNA is a sort of artificial DNA that has been cut, cloned and edited to have certain desirable characteristics for a certain purpose.
Molecular applications: Invention of insulin, medication diagnosis, invention of drugs for disease treatment
Agricultural applications: Invention of pest-resistant crops, making more nutritious crops, making faster growing and bigger crops to meet up food requirements
6. Describe the Human Genome Project (HGP), and explain its potential for biotechnology.
The Human Genome Project is an aim to fully understand and trace all the genes in humans, their individual functions and how they all work together. It has the potential to bring about personalised medicine. Every body is different and how they react to medicine is different as well. With the HGP, medicine can be tailored to each individual and this could potentially save countless lives.
7. Explain tissue engineering and gene therapy.
Tissue engineering: It is basically what it sounds like, engineering issues. This is a way of growing tissues outside the human body to meet a specific purpose. An example that was discussed was growing an ear on the back of a rat, in this case it has to do with somatic cells (skin).
Gene therapy: This is using genes to treat diseases and other things by editing specific cells at the root cause, genetic composition
8. Describe Humulin.
In the past, in order to get insulin it would have to be gotten from pigs. As a result several pigs had to be slaughtered to meet a minute need. As you can guess, it just wasn’t cutting it anymore. It was very practical to say the least. This is where humulin comes in. It is a form of artificial insulin that meets this specific need and it was made through rDNA.
9. Describe how stonewashing jeans has changed with the advent of biotechnology.
In the past, people would wash their jeans with literal stones and throw them on rocks repeatedly so they could get that “worn” look and overall wearable feel to them. If not, they would look and feel unbearably stiff. I know, not very comfortable looking or fashionable (this could also happen if you overstarch your jeans but that’s a topic for another day)
Soooo, with the advent of biotechnology, people no longer have to maltreat their jeans to make them comfy to wear and have a faded look. Instead, there are certain enzymes used when making the jeans to achieve this look.
10. Briefly describe the major ideas behind the following areas of biotechnology:
Microbial: The use of microbes for different purposes like waste disposal and the production of food
Agricultural: Using microbes to create pest resistant crops and higher yielding crops too
Animal: Cloning of animals
Forensic: DNA ancestry tracing and paternity tests
Environmental: Pollution clean up
Aquatic: Aquatic environment restoration
Medical: Tissue engineering, gene therapy, rDNA