M4: Proteins
Proteins
- basic unit of proteins: Amino Acids
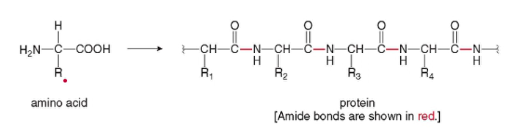
- biomolecules that contain many amide bond; formed by joining amino acids
- amino acids → peptide bonds → dipeptide (2) → oligopeptide (3-10) → polypeptide (>10) → protein
General features of Amino Acids
- Consist of the elements of life: carbon, hydrogen, oxygen, nitrogen, and a variable side chain or R group that renders chemical distinctiveness.
- Functional groups: Carboxylic acid (COOH), amino group (NH2)
* carbon adjacent to carboxylic group is alpha carbon -- carbon also attached to an H and R group ---making it a chirality center/chiral carbon 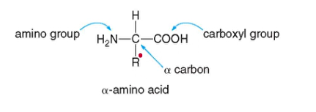
- if R is replaced by hydrogen -- glycine where R=H


- Chained together by amide (C-N) bonds known as peptide linkages.
- When hydrolyzed, bonds are broken and yield amino acids.
* ALL amino acids have an alpha-carbon (carbon next to the acid or COO- group)
○ Amino group (-NH3+) ammonium cation
○ Carboxylate anion (COO-)
○ Hydrogen
○ R group (side chain) – is unique and makes it asymmetric or chiral carbon.
* Amino acids are optically active.
- 20 amino acids (essential, nonessential, polar, nonpolar)
- \
- accounts 50% of dry weight
- must be consumed daily
- 0.8 grams per kg of body weight for adults (more for children)
- comes from meat and milk
Classification based on FUNCTIONAL GROUP
A. Non-polar Amino Acids
(electronegative present in group of atoms)
B. Polar Amino Acids
C. Acidic Amino Acids

D. Basic Amino Acids

Essential Amino Acids
Phenylalanine
Valine
Threonine
Tryptophan
Isoleucine
Methionine
Histidine
Arginine
Leucine
Lysine
- standard amino acid needed for protein synthesis
Nonessential Amino Acids

Classification based on NUTRITIONAL BASIS
Complete Proteins
- supplies all essential amino acids
- usually from animal sources (except gelatin - absence of tryptophan HC Test)
- contains all essential amino acids in the same relative amounts the body needs them.
* May or may not have all the nonessential amino acids
* Protein from animal sources
* Milk
* Soy
Incomplete Proteins
- deficient in one or more essential amino acids
- usually from plant sources (ex. rice - low in lysine and threonine)
- does not contain adequate amounts relative to the body’s needs.
* Protein from plant sources
Limiting Amino Acid:
- essential amino acid that is missing or present in inadequate amounts (in an incomplete protein)
Complementary Dietary Protein:
- two or more incomplete dietary proteins that combine to provide an adequate amount of all essential amino acids.
Functions of Proteins in the body
1. Provide structural framework
- keratin in hair and skin
- collagen in connective tissues
2. Direct and regulate metabolism in the body
- enzymes and polypeptide hormones
3. Permit muscle movement
- contractile proteins
4. Transport substances
- hemoglobin and plasma proteins
5. Defend an organism against infection
- antibodies (immunoglobulin)
Classification of Proteins according to SHAPE
1. Fibrous Proteins
- long, rod-shape molecules
- insoluble in water and physically tough
- have structural and protective functions
- ex. keratin, collagen, fibrin
2. Globular Protein
- coiled, compact, spherical molecules
- usually water soluble
- have dynamic function (transport protein)
- ex. enzymes, hemoglobin
Classification of Proteins according to COMPOSITION
1. Simple proteins
- contain only amino acids
- ex. albumin, globulin, glutalins, prolamines, histones
2. Conjugated proteins/complex proteins
- consist of a simple protein combined with a nonprotein component
- ex.
* nucleoprotein (nucleic acid)
* glycoprotein (carbohydrate),
* phosphoprotein (Phosphate),
* lipoprotein (lipid)
3. Derived proteins
- substances formed from simple and conjugated proteins
- ex.
* denatured proteins (add with acids/bases, protein precipitated)
* coagulated proteins (further heating)
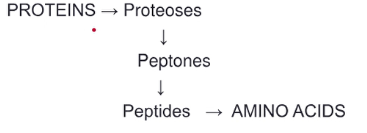
* proteoses (hydrolysis)
* peptones (hydrolysis)
* peptides (hydrolysis)
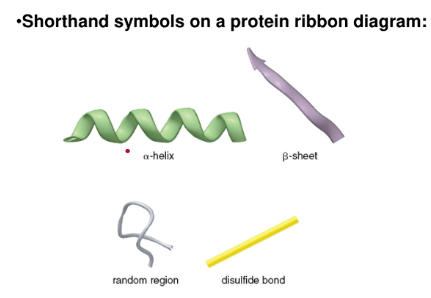
STRUCTURAL LEVEL of ORGANIZATION of PROTEINS
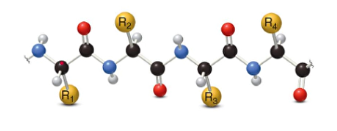
1. Primary
- sequence of amino acids joined together by peptide bonds(amide bond)
- all bonds are 120 degrees, giving the protein a zigzag arrangement
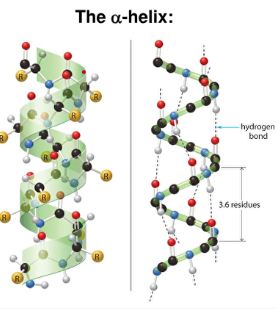
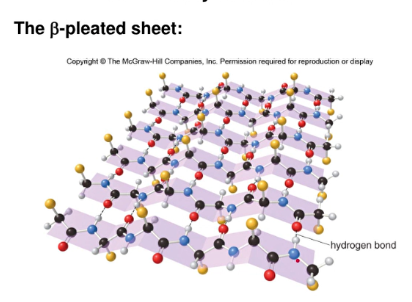
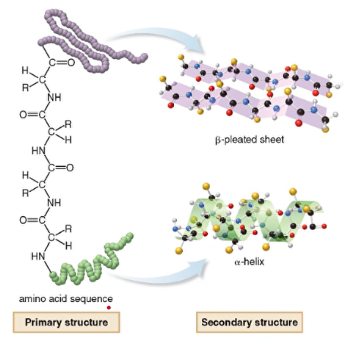
2. Secondary
- 3D arrangement of localized regions of protein
- these regions arise due to hydrogen bonding between the N-H group of one amide with C=O group to another
- two stable arrangements -- alpha helix and beta pleated sheet
* most have these regions but others are random arrangements
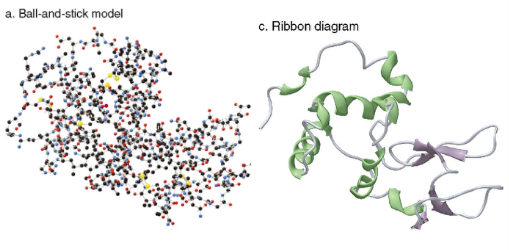
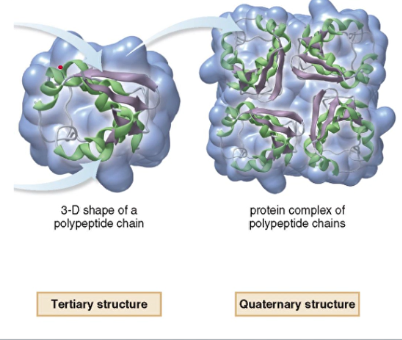
3. Tertiary
- 3D shape adopted by the entire peptide chain
- to maximize hydrogen bonding with water, the nonpolar side chains are stabilized by London dispersion forces in the interior of the structure
- Polar functional groups can hydrogen bond to each other
- Amino acids with charged side chains are attracted by electrostatic interactions
*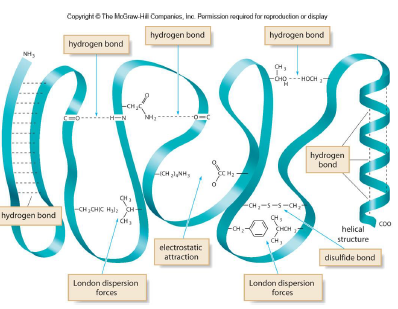
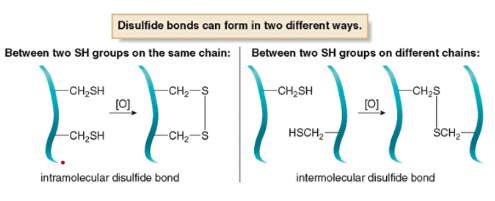
- Disulfide bonds form covalent bonds that stabilize the tertiary structure
*
4. Quaternary
- the shape adopted when two or more folded polypeptide chains come together into one complex
- Insulin
* consists of two separate polypeptide chains linked by intermolecular disulfide bonds - Hemoglobin
* consists of four subunits held together by intermolecular forces into a compact 3D shape
COMMON Proteins
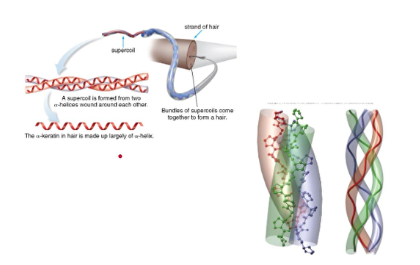
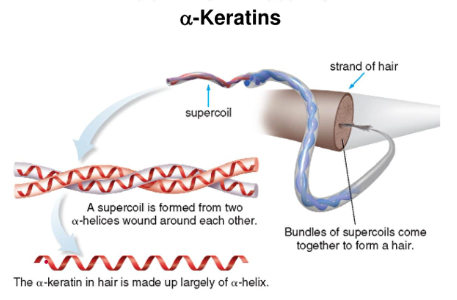
alpha-keratins
- found in hair, hooves, nails, skin, and wool
- made of two mainly alpha-helix chains coiled around each other in a superhelix
- these coils wind around other coils making larger and stronger structures (like hair)
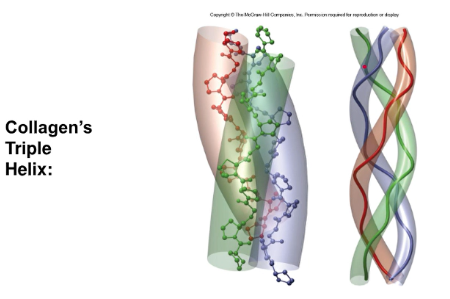
Collagen
- requires three chains in a superhelix
Vitamin C
- helps stabilize the chains
- when missing, poorly formed collagen fibers result
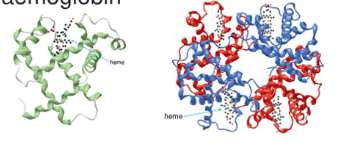
Hemoglobin and Myoglobin
- globular and conjugated proteins
* contain both a protein and non-protein component
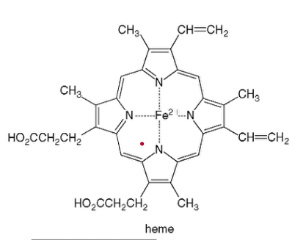
* non-protein is a heme, organic complex surrounding Fe^+2 ion (ferrous ion) which binds to O2 gas in the bloodstream
* then the hemoglobin protein transports the O2 to wherever it is needed in the body
*
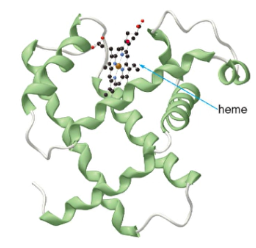
- or if needed, the myoglobin stores O2 in tissues
- Myoglobin has 153 amino acids in 1 polypeptide chain
*
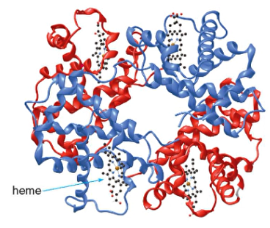
- Hemoglobin has 4 polypeptide chains, each carrying a heme unit
*
- Carbon monoxide poisoning (CO)
* poisonous because it binds 200x more strongly to ferrous ion than does oxygen
* Hemoglobin complexed with CO cannot carry oxygen and cells will die from lack of oxygen - Sickle cell anemia
* a single amino acid is different in two of the subunits of hemoglobin
* red blood cells containing these mutated hemoglobin units become elongated and crescent (sickle) shaped
* these will rupture capillaries, causing pain and inflammation leading to organ damage and death
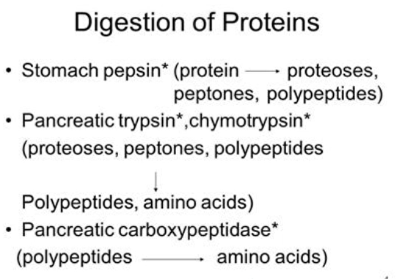
Protein HYDROLYSIS
- breaking the peptide bonds by treatment with aqueous acid, base, or certain enzymes
- in the body, enzyme pepsin in gastric juice cleaves some of the peptide bonds of large proteins to make smaller peptide chains
- in the intestines, enzymes trypsin and chymotrypsin hydrolyze the remainder of the amide bonds resulting to individual amino acids
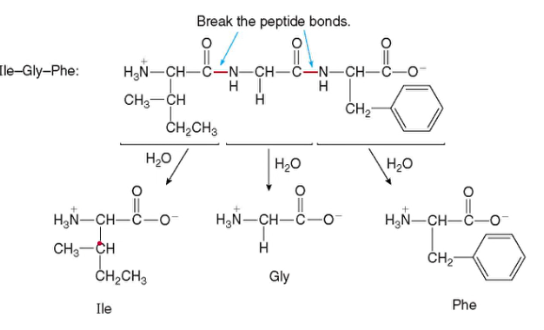
- heat and acid or base can completely hydrolyze proteins

Enzymatic hydrolysis
1. Acid Hydrolysis
- natural stereochemical configuration of amino acid is retained
- tryptophan is destroyed
- Serine and threonine are decomposed to some extent
2. Alkali hydrolysis
- racemization occurs
- arginine and cysteine are decomposed
3. Enzyme hydrolysis
- most gentle but seldom done in the lab
- in the digestive tract, hydrolysis is complete
Active Protein Enzymes
- add “sinogen”
- ex. pepsin - active ; pepsinogen - inactive
Protein DENATURATION
- process of altering the shape of a protein without breaking the amide bonds that form the primary structure
*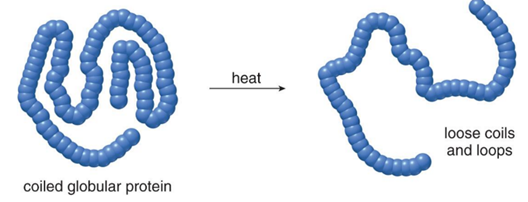
- high temp, acid, base, and agitation can disrupt the non-covalent interactions that hold a protein in a specific shape
- often makes globular proteins less water-soluble
- Ovalbumin
* major protein in egg white is denatured when an egg is cooked
* changing from clear and colorless to opaque and white
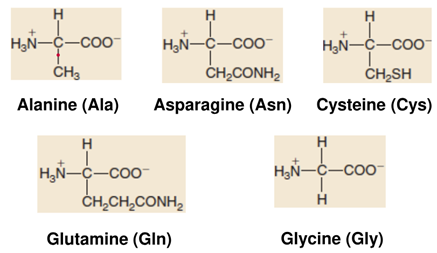
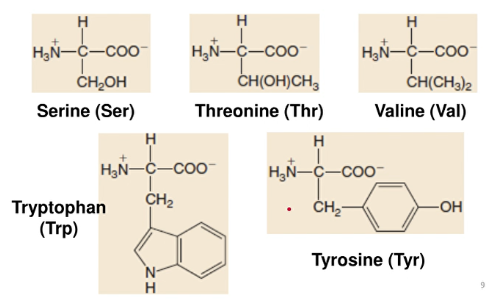
AMINO ACID STRUCTUREs
Neutral Common Amino Acids
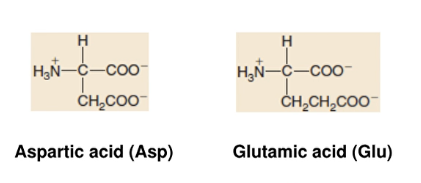
Acidic Amino Acids
- more carboxylic group/amino group
- form acidic solution when dissolved in water
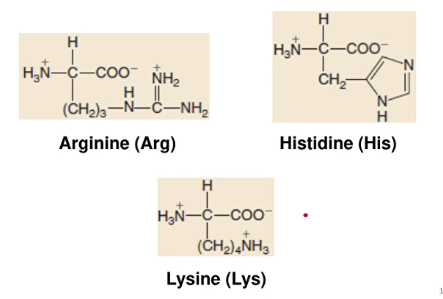
Basic Amino Acids
- form basic solutions when dissolved in water
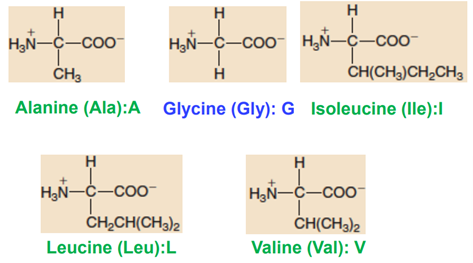
Aliphatic Non-polar Side Chains
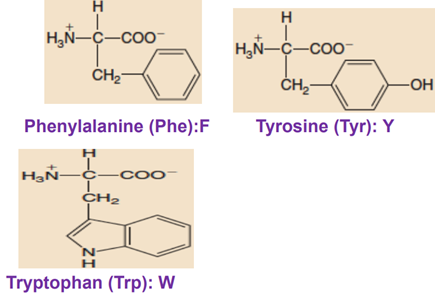
 Aromatic Polar Side Chains
Aromatic Polar Side Chains
 Hydroxyl-containing Side Chains
Hydroxyl-containing Side Chains
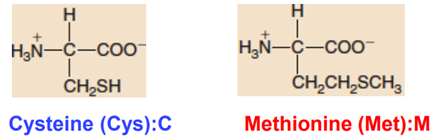
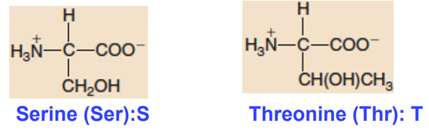 Sulfur-containing Side Chains
Sulfur-containing Side Chains
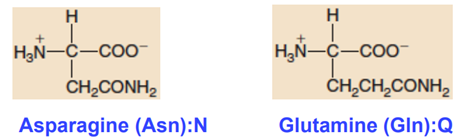
 Amidic
Amidic
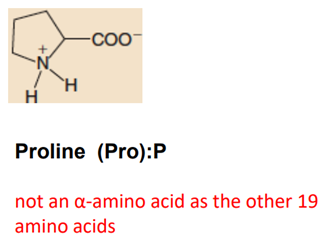
 Imino
Imino
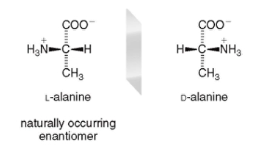
STEREOCHEMISTRY of Amino Acids
- all amino acids (save glycine) have a chirality center on the alpha carbon
* L amino acids (naturally occurring) have the -NH3+ on the left
* D amino acids (naturally occurring) have the -NH3+ on the right
*
ACID-BASE BEHAVIOR of Amino Acids
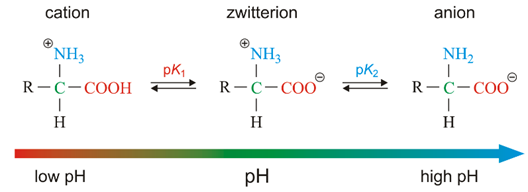
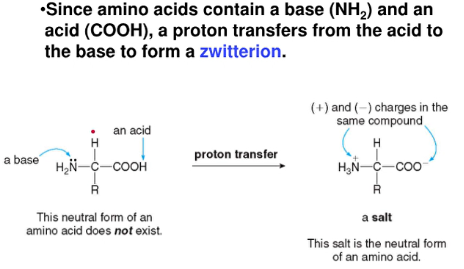
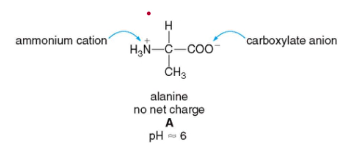
- amino acid exists as a neutrally charged zwitterion at a certain pH, the isoelectric pH
*
- amino acid can exist in different forms, depending on the pH of the aqueous environment
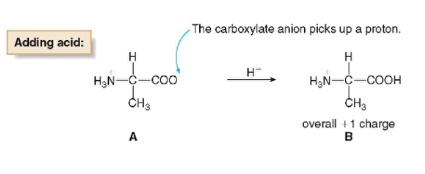
- when pH < isoelectric pH
* the carboxylate anion gains a proton, and the amino acid has a net positive charge
*
- when pH > isoelectric pH
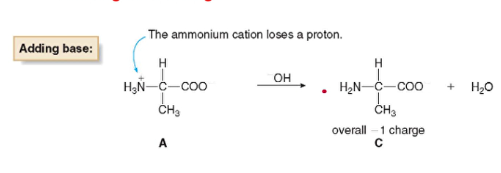
* the ammonium cation loses a proton, and the amino acid has a net negative charge
*
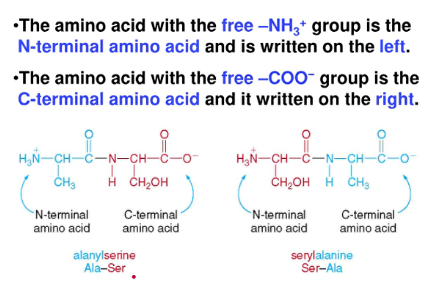
Peptides
- peptides and proteins are formed when amino acids are joined together by amide bonds
- dipeptide - two amino acids joined together by one amide bond
*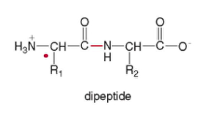
* the amide bond is called a peptide bond - tripeptide - three amino acids joined together by one amide bond
*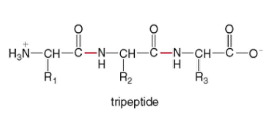
- Polypeptides have many amino acids;
- Proteins have more than 40 amino acids
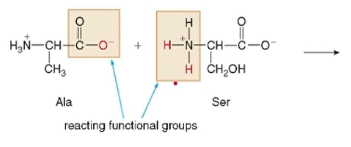
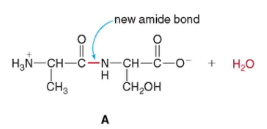
- amino acids Ala and Ser can be combined this way (vice versa):
* Condensation reaction forming water and combining
*
Biologically Active Peptides
Neuropeptides - Enkephalins and Pain Relief
Enkephalins
- pentapeptides made in the brain
- act as painkillers and sedatives by binding pain receptors
- addictive drugs morphine and heroin bind some of these pain receptors
* producing a similar physiological response, though longer-lasting - belong to the family of polypeptides called endorphins
* known for pain-reducing and mood-enhancing effects - example:
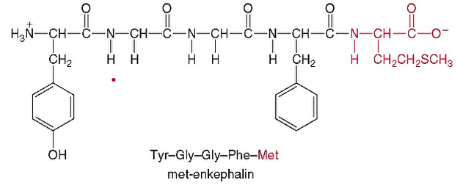
* met-enkephalin
*
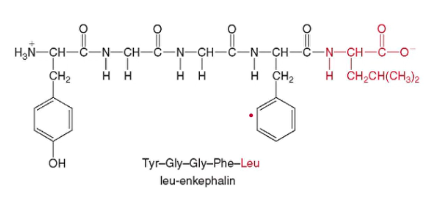
* leu-enkephalin
*
Peptide Hormones - Oxytocin and Vasopressin
Oxytocin and Vasopressin
- cyclic nonapeptide hormones
- have identical sequences except for two amino acids
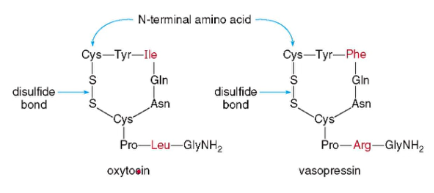
- slightly different sequence gives the two peptides vastly different effects on the body
Oxytocin
- stimulates the contraction of urine muscles, and signals for milk production
- often used in induced labor
Vasopressin
- antidiuretic hormone (ADH) targets the kidneys and helps to limit urine production
- to keep body fluids up during dehydration