Electronegativity
electronegativity deescribes the tendecny of an atom to attract a shared pair of eelctrons towards its nucleus.
affected by its atomic number and the distance of the valence electrons from its nucleus (i.e atomic orbitial)
“how strong of a pull” a particular atom has on electrons from a neighbouring atom
more electronegative means it wil hog electrons more of the time while participating in a covalent bond
as we move from the left to the right, and top to bottom, the electronegativity increases because of atomic radius
0 non polar
0-0.5 slightly polar
0.5-1.7 polar
1.7 or greater ionic
a bond dipole
shows the direction in which electrons tend to spend more of the time
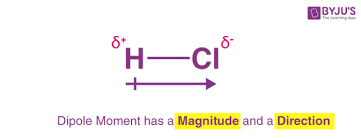
S+ represents a partial charge less than +1
S- represents a partial charge less than -1