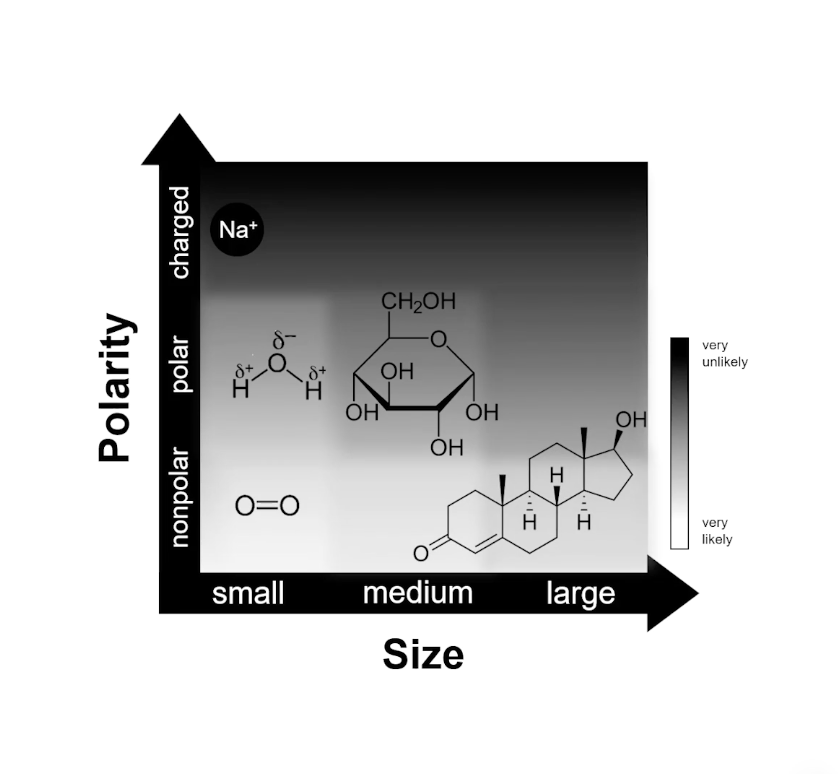
Diffusion Scale
Factors Influencing Membrane Permeability
The permeability of the phospholipid bilayer is selective, allowing some molecules to pass through while restricting others. This selectivity is primarily determined by two characteristics: Polarity and Size.
Polarity and Charge
Nonpolar Molecules: These are hydrophobic and can easily dissolve in the lipid interior of the bilayer. Even large nonpolar molecules have a higher likelihood of crossing than polar ones.
Example: Oxygen () and Steroid hormones.
Polar Molecules: These are hydrophilic and have difficulty crossing the hydrophobic core of the membrane.
Example: Water () with partial charges ( and ) and Glucose ().
Charged Ions: Molecules with a full electrical charge are effectively blocked by the membrane because they cannot interact with the nonpolar fatty acid tails.
Example: Sodium ().
Molecular Size
Small: Generally pass through more easily. Small nonpolar gases pass through the quickest.
Medium/Large: As the size of the molecule increases, the likelihood of simple diffusion decreases, often requiring specialized transport proteins.
Permeability Likelihood Scale
Based on the relationship between size and polarity, we can observe the following permeability trends:
Very Likely: Small, nonpolar molecules like Oxygen ().
Likely: Large, nonpolar molecules (such as steroids) and very small polar molecules like Water ().
Unlikely: Medium to large polar molecules like Glucose.
Very Unlikely: Charged particles and ions such as .

Summary Table
Molecule | Size | Polarity | Permeability |
|---|---|---|---|
Small | Nonpolar | High | |
Small | Polar | Moderate | |
Glucose | Medium | Polar | Low |
Steroids | Large | Nonpolar | Moderate/High |
Small | Charged | Very Low |