Outline of disease process 1-2
Learning Outcomes:
• Describe the principles of ‘staging’ in cancer.
• Explain the importance of genetic changes in cancer.
• Describe modalities of therapy currently available
-What is Cancer PT1
Disease of the genome
Cancer is a disease of the genome occurring as a result of unregulated cell growth
UK Statistics: •1 IN 2 PEOPLE BORN AFTER 1960 WILL DEVELOP CANCER IN THEIR LIFETIME
•A NEW DIAGNOSIS IS MADE EVERY 2 MINUTES
•CANCER CAUSES MORE THAN 1 IN 4 OF ALL DEATHS
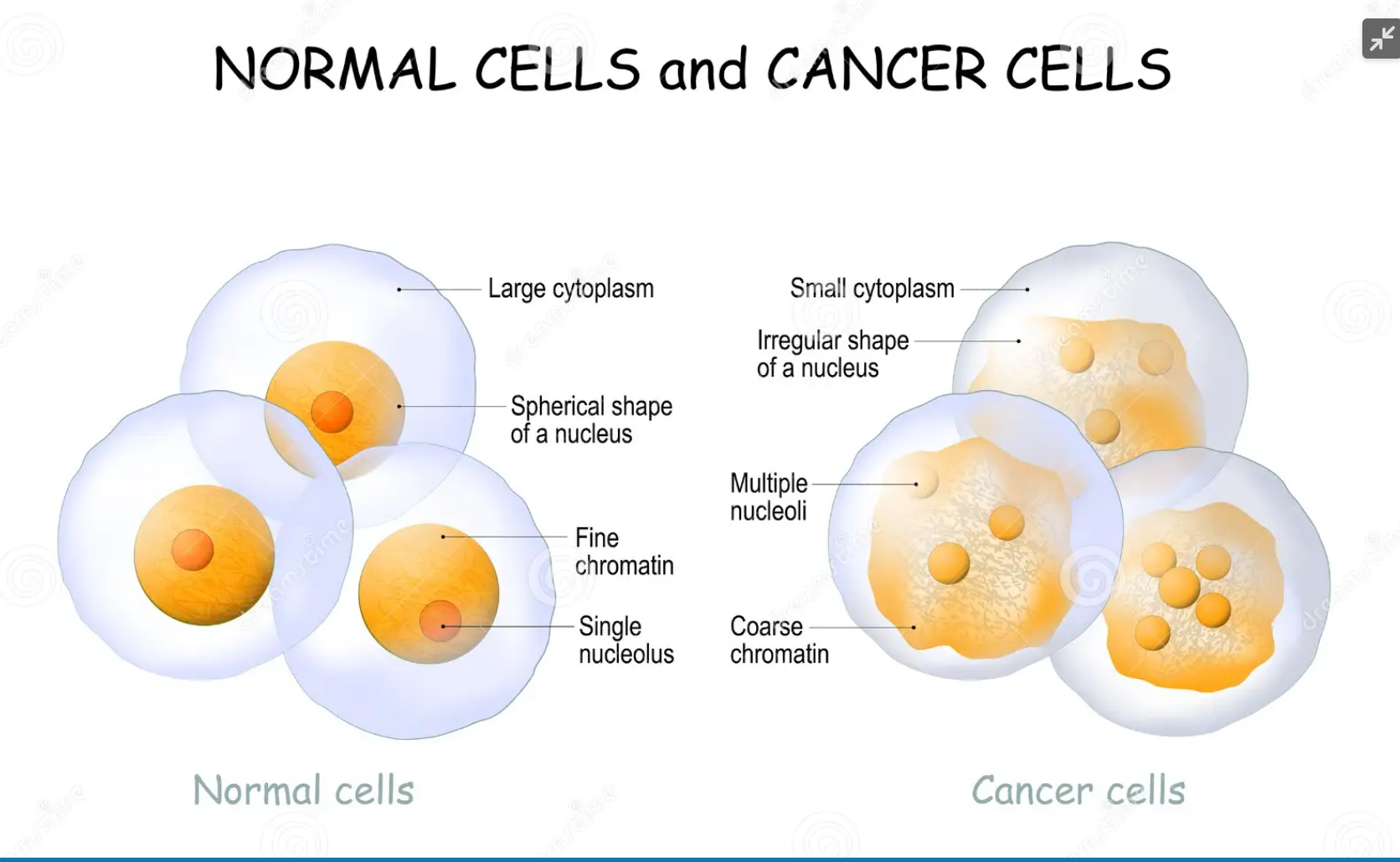
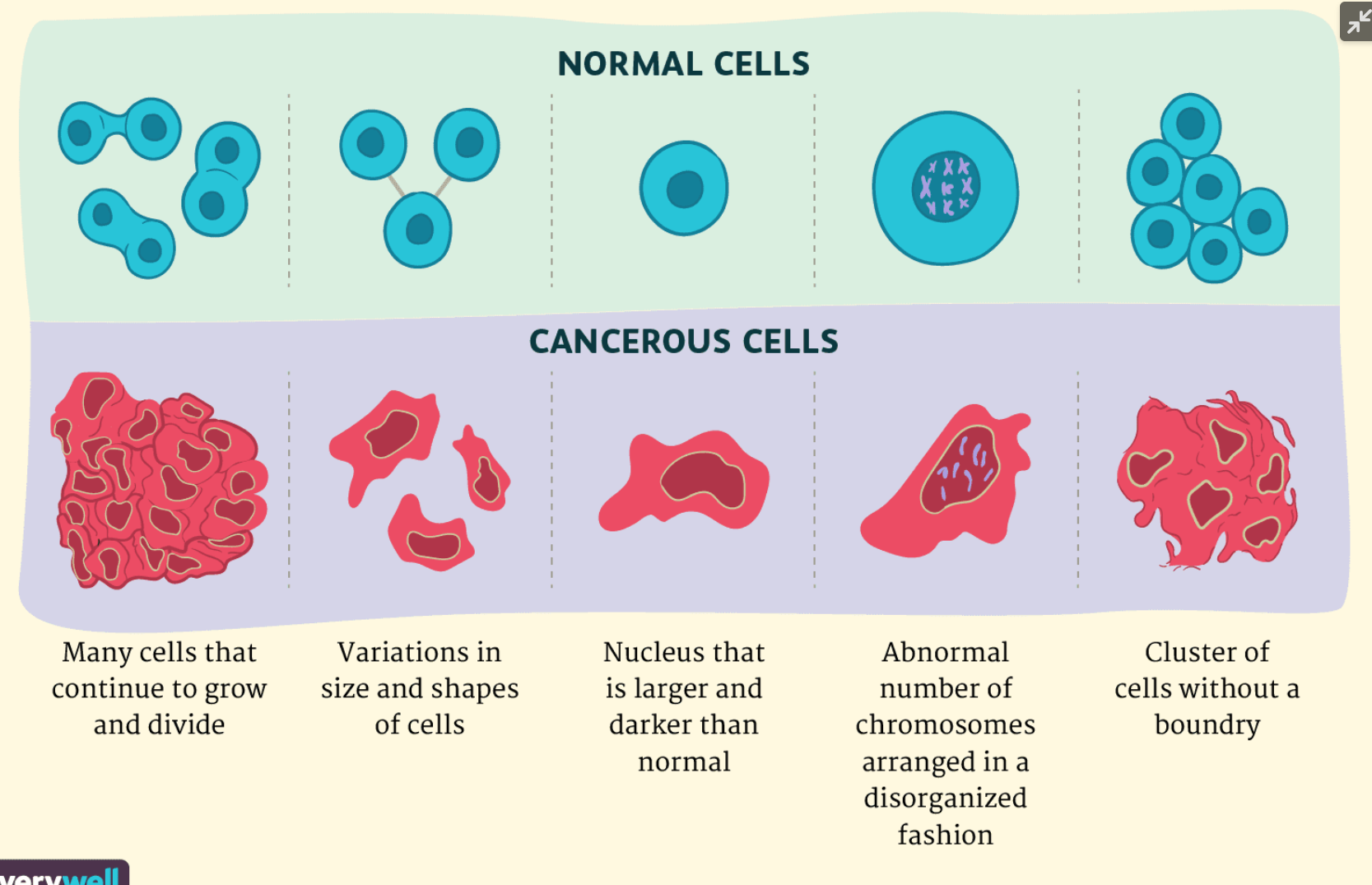
Types of cancer cells
-epithelial cells
squamous (flat)
cuboidal
columnar
85% of cancers
Carcinomas
-mesoderm cells
bone
muscle
Sarcomas
-glandular cells
glandular cells e.g. breast, oesophagus, lung
Adenocarcinomas
The hallmarks of cancer
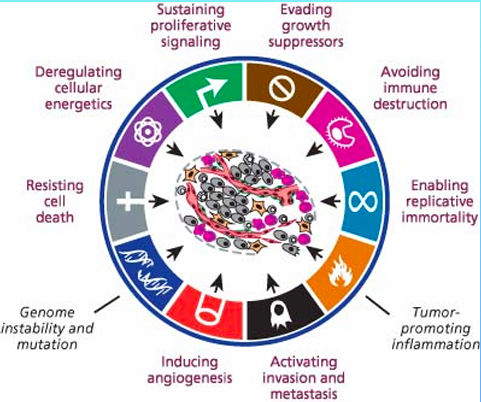
Sustaining proliferative signalling
GROWTH SIGNAL AUTONOMY = LACK OF REGULATION OF GROWTH FACTOR SIGNALLING
NORMAL CELLS REQUIRE AN EXTERNAL GROWTH SIGNAL TO DIVIDE
CANCER CELLS BYPASS NORMAL GROWTH FACTOR PATHWAYS LEADING TO UNREGULATED GROWTH
OCCURS AS RESULT OF ACQUIRED MUTATIONS
* MUTATION = ANY CHANGE IN DNA SEQUENCE OF A CELL
Evasion of inhibitory growth signals
Inhibitory growth signals maintain homeostasis within the tissue
Cells are not continuously dividing as a result
Cancer cells ignore these signals - enabled by acquired mutations and gene silencing
* Gene silencing = interruption or suppression of gene expression at transcriptional or translational level
Avoiding Immune Destruction
Immune system can recognise and remove cancer cells
However some cancer cells are able to avoid detection by not initiating an immune response
cancer cells hijack immune checkpoints and modulate immune response via Sting
What is an immune checkpoint? built in control mechanisms that maintain self tolerance during an immune response
Unlimited Replicative Potential
Normal cells have a counting device (telomeres) that monitor and adjust the number of cell doublings
Once cell numbers have reached this finite number they enter senescence
Cancer cells maintain telomere length - replication overdrive begins
Tumour Promoting Inflammation
All tumours have inflammatory immune cells
Inflammatory cells provide growth factors that promote angiogenesis and invasion
Cell death by necrosis gives rise to inflammation4
Necrotic cells release bioactive regulatory factors IL - 1
Inflammatory cells can release radical oxygen species that give rise to mutations
Invasion and Metastasis
Cancer cells develop the ability to migrate to other areas
Formation of metastasis is a major cause of death in cancer
Mutations within the genome may affect the enzymes involved in cell- cell adhesion e.g. E-cadherin
Angiogenesis
Creation of new blood vessels by the tumour
Provides supply of oxygen and nutrients
New blood vessels are friable leading to tumour cell escape
Many drugs have been developed to target angiogenesis
Genomic Instability
Alterations in DNA lead to instability
Faulty DNA repair pathways or hereditary predisposition contribute to the development of DNA alterations (mutations)
Single point and large chromosomal abnormalities can be found in tumour DNA
Accumulation of mutations over a period time explains why cancer is more frequent in the ageing population
Evasion of cell death
Normal cells undergo cell death in response to extracellular factors or Physical damage
Cell death is either regulated (programmed)=Apoptosis or unregulated = Necrosis
Cancer cells evade death as a result of mutations within the Apoptosis pathway
Caspases play central role in apoptosis therefore mutations in this family will allow cancer cells to pass through unchecked
Cell death occurs in physiological conditions e.g. Menstruation/ embryogenesis and in pathological conditions e.g. DNA damage
Deregulating Cell Energetics
This is reprogramming energy metabolism
Aerobic glycolysis - used by cancer cells to redirect energy
Allows cancer cell to fuel cell growth and division
PET scanning was developed using FDG (glucose analogue) to trace the activity of cancer cells throughout the body
Genes
somatic mutations - most common and is acquired
Germline mutations - hereditary
Gene = composed of DNA, everyone has 2 copies of each gene
All cells have the ability to become oncogenic
Oncogene = mutated gene giving rise to tumour formation in a dominant fashion
Tumour suppressor gene = inhibits tumour formation
Mutations can occur within tumour suppressor genes - usually recessive
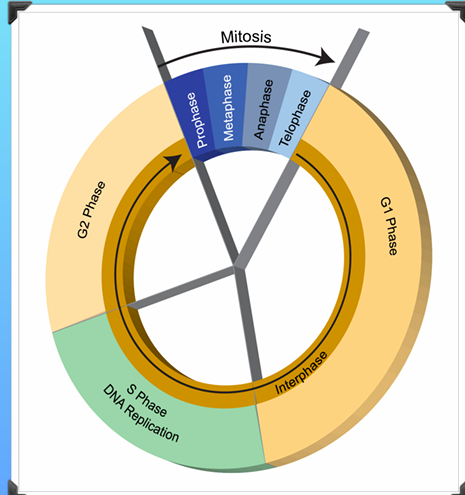
The Cell Cycle
G0- Resting phase
G1 - Cells grow larger and copy organelles
S - Cells make a complete copy of DNA
G2 - Further cell growth
M - 4 phases of mitosis
Invasive Cancer-PT2
Tumour Spread
• Organs are well defined by basement membranes
• Basement membrane is made up of extracellular matrix proteins
Theories of Spread- en route theory
-seed and soil
-pre- metastatic niche
Extracellular Matrix
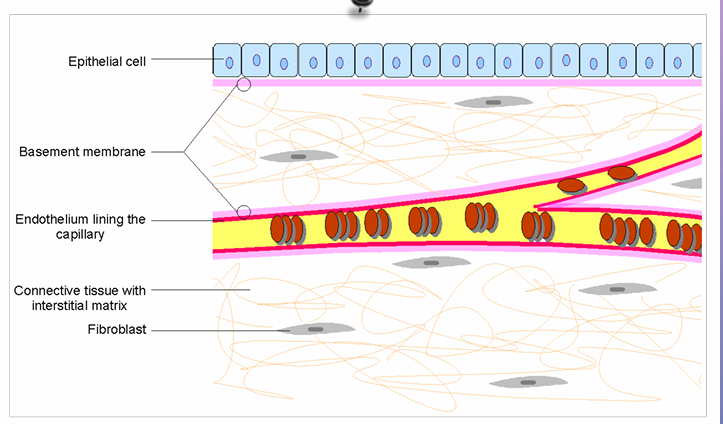
Extracellular Matrix (ECM) - complex meshwork of proteins and carbohydrates
Major component of ecm is collagen/proteoglycans - gives structural integrity to tissues
ECM is directly connected to the cells it surrounds- it is the interface between the cell and other surrounding structures like blood vessels
It is by penetration of this matrix that cancer cells can move into the blood stream and ultimately around the body
Cadherins are a type of cell adhesion molecule (cam) - these bind cells to each other and the ECM
E -cadherin is involved in cell-cell adhesion of epithelial cells
Epithelial cancers frequently show downregulation and mutation of e-cahderin
Mechanisms of Metastasis
Spread of tumour cells from the primary tumour is not clonal
Primary tumour is composed of cells that are subcloncal
2 Different Mechanisms >monoclonal >polyclonal
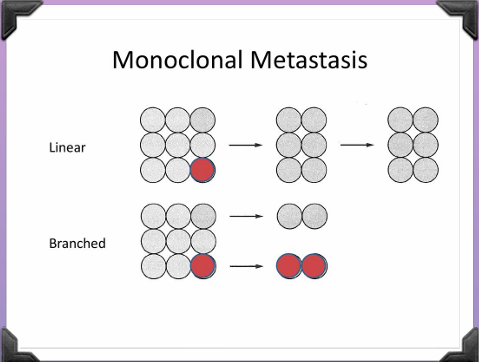
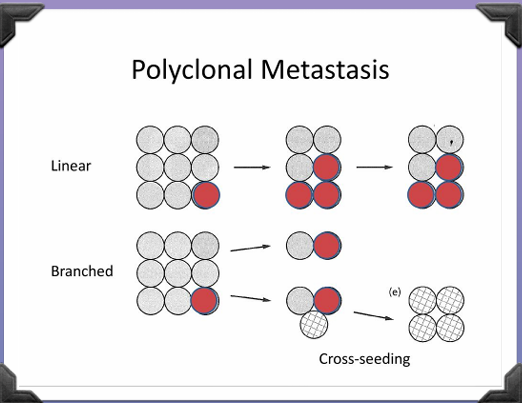
2 Different Patterns >Linear >Branched
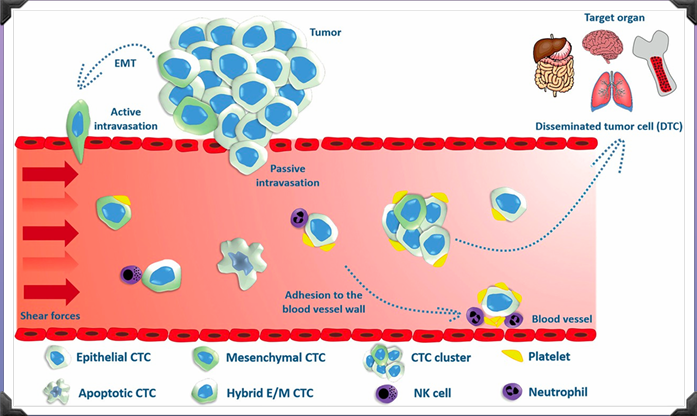
Epithelial Mesenchymal Transition (EMT)
Cells must acquire migratory characteristics
EMT is the conversion of closely connected epithelial cells becoming independent mesenchymal cells with the ability to move and invade their local environment
This is a reversible process
EMT usually occurs in embryogenesis however this also occurs in cancer metastasis
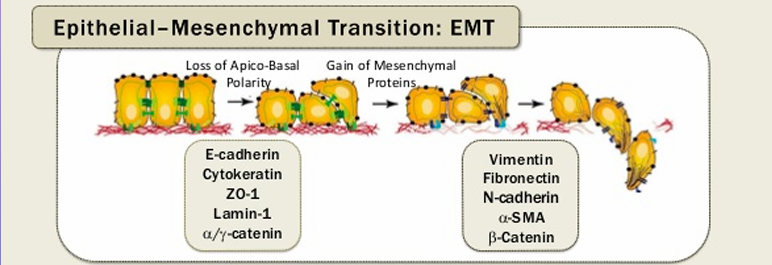
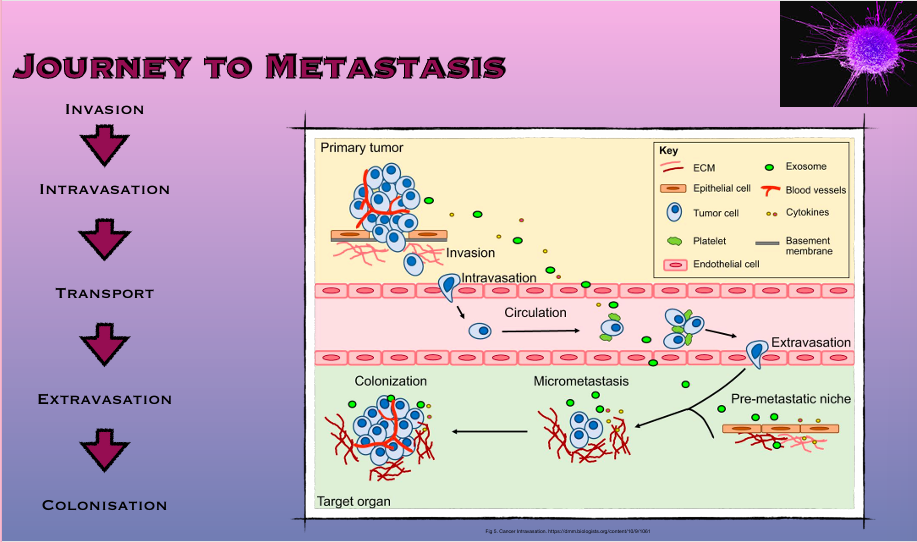
Journey to metastasis: invasion
EMT begins with signals from tumour stroma (HGF, TGF-beta) stimulate kinase receptors (EFGR) & trigger MAPK pathway
Multiple components involved in invasion
Cell Adhesion Molecules - Cadherins ( calcium dependent transmembrane proteins) & Catenins (protein inducing gene expression)
Integrins - enable cells to “break free” becoming mobile
Proteases - make the pathway through ECM, Matrix Metalloproteins contribute to loss of cell junctions
Journey to metastasis: intravasation
Intravasation = entry into blood or lymphatics
Tumour cell attaches to stromal side of basement membrane
MMPs and serin proteases help to degrade basement membrane
Tumour cell passes between the endothelial cells and off into the bloodstream (transendothelial migration)
Journey to metastasis: transport
Tumour cells in bloodstream = circulating tumour cells (CTCs)
Solo travellers vs. Clumps - unidirectional
Certain cancers have favoured metastatic sites - first pass organ
Journey to metastasis: extravasation
Exit of tumour cells from bloods vessels into distant tissues
Tumour cells become trapped in capillaries
Reverse of intravasation
Endothelial side of blood vessel - degrade basement membrane -migrate into stroma
E- Selectin is a calcium dependent receptor which enables attachment of the cancer cell to the endothelium surface of blood vessels and passage through the endothelium (transendothelial migration)
Journey to metastasis: colonisation
Site of metastasis is determined by the point of extravasation but also the microenvironment
Environment must be favourable - for the tumour to grow it must create new blood vessels (angiogenesis) for nutrients and oxygen
Cells can spread but not colonise - dormant (micrometastases)
Angiogenesis
Angiogenesis = formation of new blood vessels
Angiogenic switch - dependent on inhibitors and inducers
Anti - angiogenic factors:
Angiostatin
Endostatin
Prolactin
Protein 53 (p53)
Thrombospondin 1 & 2
Pro - angiogenic factors:
VEGF
Fibroblast growth factor
Hepatocyte growth factor
Epidermal growth factor
Platelet derived growth factor
Angiogenic Inducers
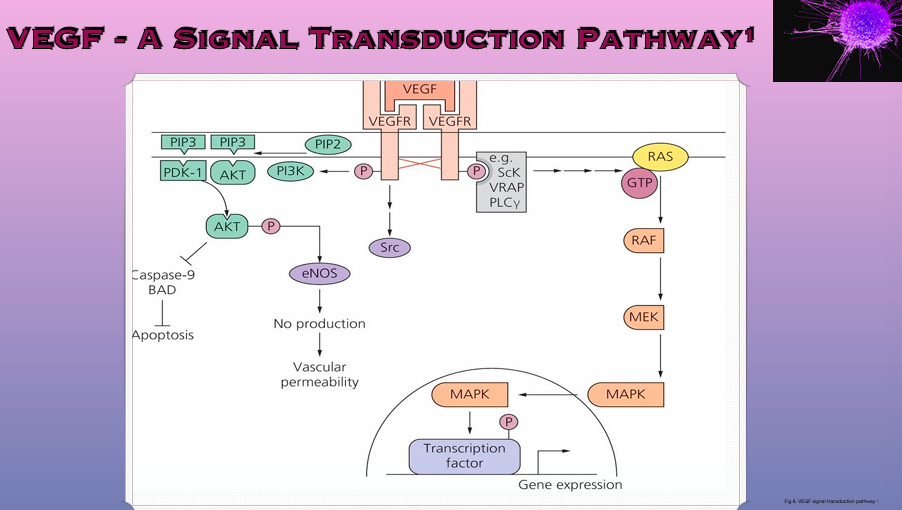
VEGF is the Star player ** (vascular endothelial growth factor)
VEGF family = A -D and placental growth factor
Signals are transmitted via VEGF Receptors 1-3
VEGFR must be phosphorylated to be become activated
Tumour cells can also stimulate nearby cells to produce VEGF and in turn promote angiogenesis
Angiogenic Inhibitors
Inhibitors help to regulate angiogenesis
Plasminogen is cleaved to form angiostatin
Endostatin blocks the MAPK pathway thus inhibiting gene expression
Concomitant resistance - enabling growth in distant metastases
Angiogenic switch is controlled by hypoxia
Tumours create a hypoxic environment activating HIF1 alpha & beta subunit triggering VEGF
Many drugs have been developed to inhibit angiogenesis e.g TKI Afatanib