Topic 5 - Respiratory system (respiratory pigments) [Part 1]
What are the four respiratory pigments
hemoglobin (red)
chlorocruorin (green)
hemerythrin (violet)
hemocyanin (blue)
which animals use it as part of their circulatory system
hemoglobin (red)
humans
most vertebrates
some invertebrates
chlorocruorin (green)
segmented worms - earthworms + leeches
marine worms (bristleworms)
hemerythrin (violet)
certain marine invertebrates - bottom dwelling worms
brachiopods
hemocyanin (blue)
many mollusks - chitons, many gastropods (snails + slugs)
cephalopods (octopus + squid)
arthropods (crustaceans - lobster, shrimp)
arachnids (spiders + scorpions)
horseshoe crab
determine if each pigment uses iron or copper when binding with oxygen
hemoglobin (red) → iron
chlorocruorin (green) → iron
hemerythrin (violet) → iron
hemocyanin (blue) → copper
determine colour of pigment when oxygenated
hemoglobin (red) → bright red
chlorocruorin (green) → darker green
hemerythrin (violet) → violet-pink
hemocyanin (blue) → blue
determine colour of pigment when deoxygenated
hemoglobin (red) → dark red
chlorocruorin (green) → light green
hemerythrin (violet) → colourless
hemocyanin (blue) → colourless
define porphyrin
a heterocyclic ring that holds an iron ion - part of the heme group → polypeptide subunit
list the six coordination bonds that help anchor iron in heme of one subunit
4 bonds total - from nitrogen atoms of porphyrin
1 bond total - from nitrogen atom associated with histidine amino0acid residue of hemoglobin
1 bond total - from oxygen when hemoglobin is oxygenated
differentiate between T-state and R-state hemoglobin
T-state (tense)
interactions between globulin subunits are stronger (compared to R-state)
oxygen affinity is lower in this state (is bonded less tightly - easier to release)
is deoxy-Hb
polypeptide subunits wrap tightly around the heme group - making it difficult for oxygem to gain access to iron → results in oxygen bind to be weaker
R-state (relaxed)
oxygen affinity is higher
oxy-Hb
polypeptide subunits are wrapped loosely around the heme group → easier for oxygen to gain access to iron (Fe2+)
Draw out a typical oxygen-adult hemoglobin equilibrium curve
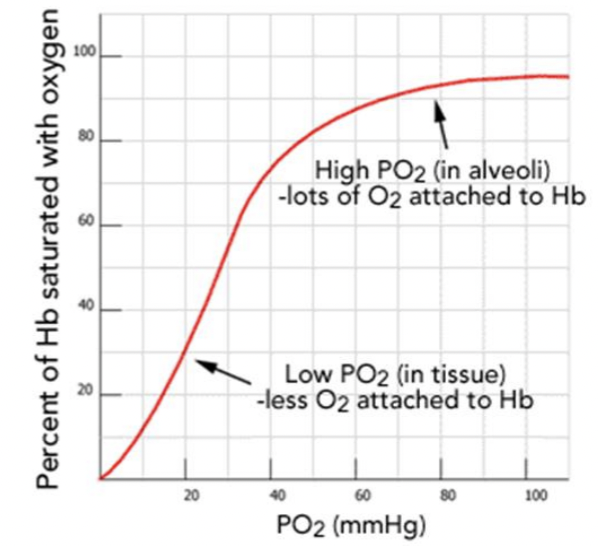
Cooperative Binding (Positive Cooperativity): Hemoglobin is a tetramer, consisting of four subunits, each containing a heme group that can bind one oxygen molecule.
Initial Low Affinity (T-State): Initially, hemoglobin is in a "tense" (T) state with low affinity for oxygen.
Conformational Shift (R-State): Once the first oxygen binds, the iron atom moves into the plane of the porphyrin ring, initiating a structural shift in the entire protein to a "relaxed" (R) state.
Increased Affinity: This transition increases the affinity of the remaining heme groups, making it easier for the second, third, and fourth oxygen molecules to bind
*make sure to label the axis correctly when recreating the graph
represents how a small change in partial pressure of oxygen can result in a large change in deliver of oxygen
identify the scientific name of the shape of curve and explain why this curve has this particular shape
sigmoidal shape
due to co-operative binding of oxygen to hemoglobin
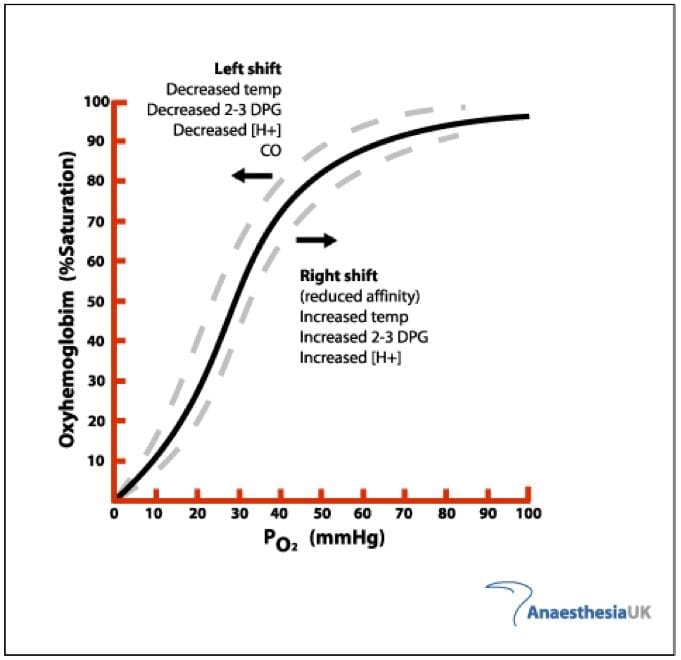
Explain what a left Bohr shift means in terms of oxygen binding capacity with hemoglobin
Bohr shift - a physiological phenomena in which factors can affect the loading or unloading of oxygen by hemoglobin
Bohr shifts to left → oxygen binds strongly to hemoglobin
Bohr shifts to right → oxygen is released readily by hemoglobin
Determine what causes a left Bohr shift in relation to pH, blood CO2 levels, temperature and 2,3-bisphosphoglycerate levels
increase in pH
decrease in blood CO2 levels
decrease in temperatures
decrease in 2,3-bisphophoglycerate levels
Draw out a typical oxygen-fetal hemoglobin equilibrium curve
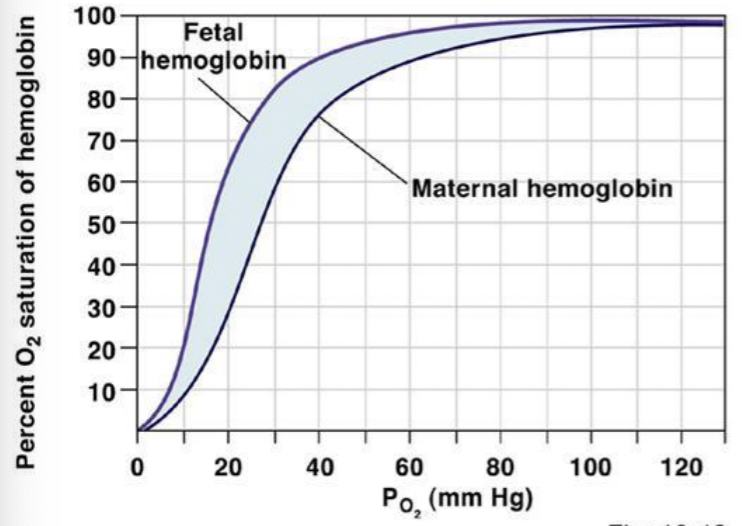
Differentiate between the structure of fetal and adult hemoglobin
fetal hemoglobin - 2 alpha chain + 2 gamma chains
adult hemoglobin - 2 alpha chain + 2 beta chains
Explain why fetal hemoglobin has a higher oxygen binding affinity compared to adult
hemoglobin
has 2 gamma chains that binds less strongly (compared to the beta chains) to 2,3-bisphosphoglycerate (2,3-BPG), a molecule that reduces oxygen affinity
Draw out a typical oxygen-myoglobin equilibrium curve
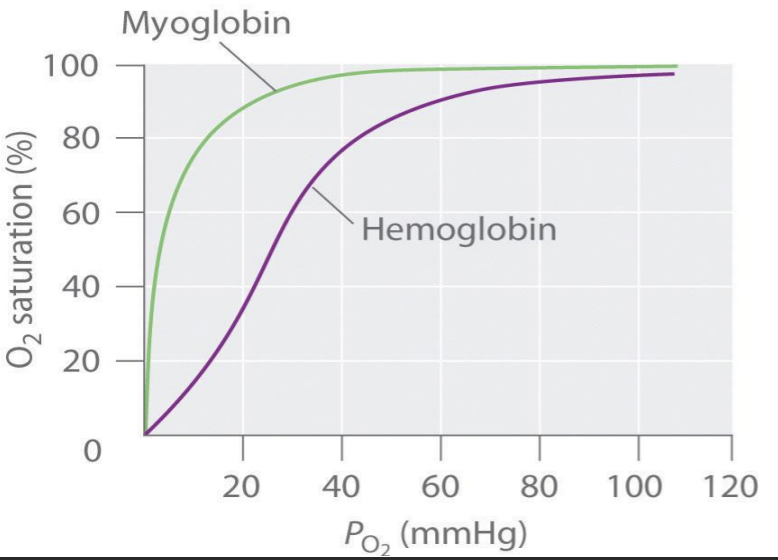
Be able to correctly identify the scientific name of the shape of curve, and why this curve has this particular shape
shape of myoglobin curve - hyperbolic
oxygen-myoglobin equilibrium is on the left - affinity of oxygen bound to myoglobin is greater than hemoglobin
Differentiate between the structure of myoglobin and adult hemoglobin
myoglobin
heme-containing respiratory pigment
monomeric protein
binds to oxygen more tightly than hemoglobin does
State the location of myoglobin
founded in striated muscle tissues (skeletal and cardiac)
plays a part in storing oxygen at high concentration inside muscle cells - needed for movement
Describe which types of animals has myoglobin (especially why diving animals
require higher levels of myoglobin
vertebrates
reptiles
amphibians
mammals
fish
higher concentrations of myoglobin allow organisms to hold their breath for a longer period of time
diving mammals (ie. whale + seals) have muscles with particularly high abundance of myoglobin
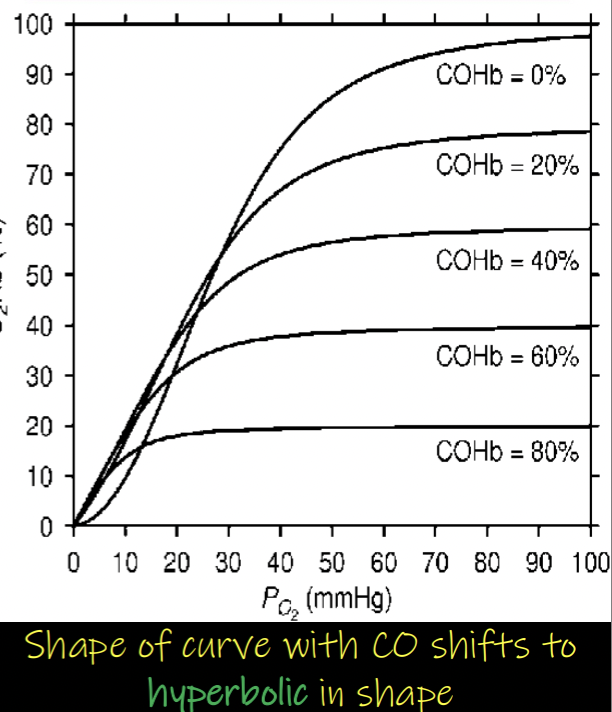
Describe how carbon monoxide affects binding of oxygen to adult hemoglobin
Carbon monoxide binds to hemoglobin with affinity that is about 240 times greater than that of oxygen
binds similarly to oxygen at the same site of hemoglobin - small quantities of CO effectively displace oxygen and stabilize the R-state
carbon monoxide binds at lower capacities of hemoglobin - by filling active sites
shifts equilibrium towards R-state → Bohr shift to left
oxygen carried by Hemoglobin is less likely to be released at the tissues - reduces the toxic effects of carbon monoxide
carbon monoxide (CO) drastically reduces oxygen binding to hemoglobin by binding to the sites with 240 times greater affinity - > decreases oxygen carrying capacity
when carbon monoxide binds to the hemoglobin → makes the remaining oxygen attached to the hemoglobin have higher affinity → harder to release