Sulfur and Silica
Background
Nutrient: any element that an organism must take in to live, grow, and reproduce
Macronutrients: C, H, N, O, P, S
Hydrologic = C and H (water)
Gaseous = C and N (main nutrients in atmosphere)
Sedimentary = P and S (mostly in rocks and soil)
Liebig’s Law of Minimum
Composition of Silica (Si)
Biosphere = ~0
Lithosphere = 20.5
Hydrosphere = ~0
Atmosphere = ~0
Demand/Supply (in water) = 2000
Composition of Sulfur (S)
Biosphere = ~0.03
Lithosphere = ~0.06
Hydrosphere = ~0.01
Atmosphere = ~0
Forms of Sulfur
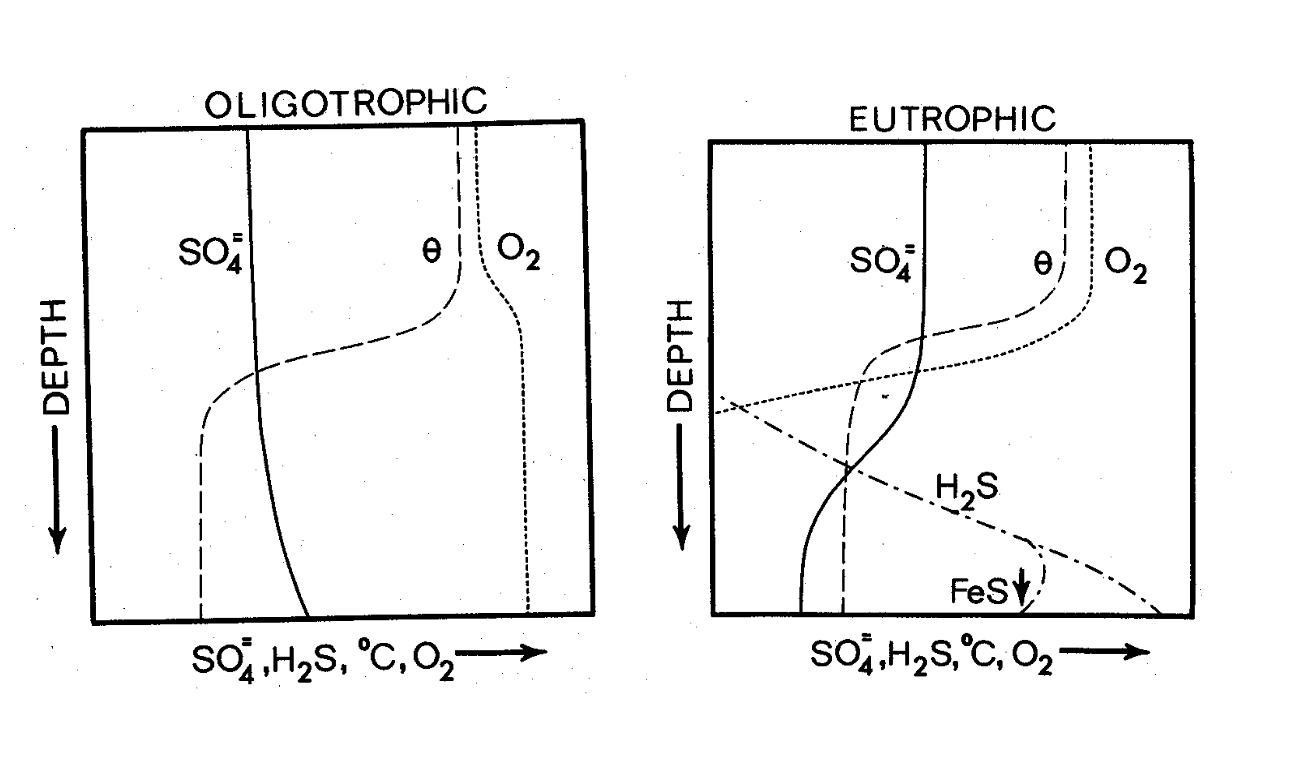
SO42- (sulfate) = most oxidized form
is 2nd only to carbonate as the principle anion in lakes
H2S (hydrogen sulfide)
during decomposition, S is reduced to H2S
oxidized rapidly in oxygenated waters
toxic to all eukaryotes (only some bacteria can survive)
Chemosynthetic bacteria
Chemosynthetic bacteria: get their energy from breaking down chemical bonds
Sulfur Cycle
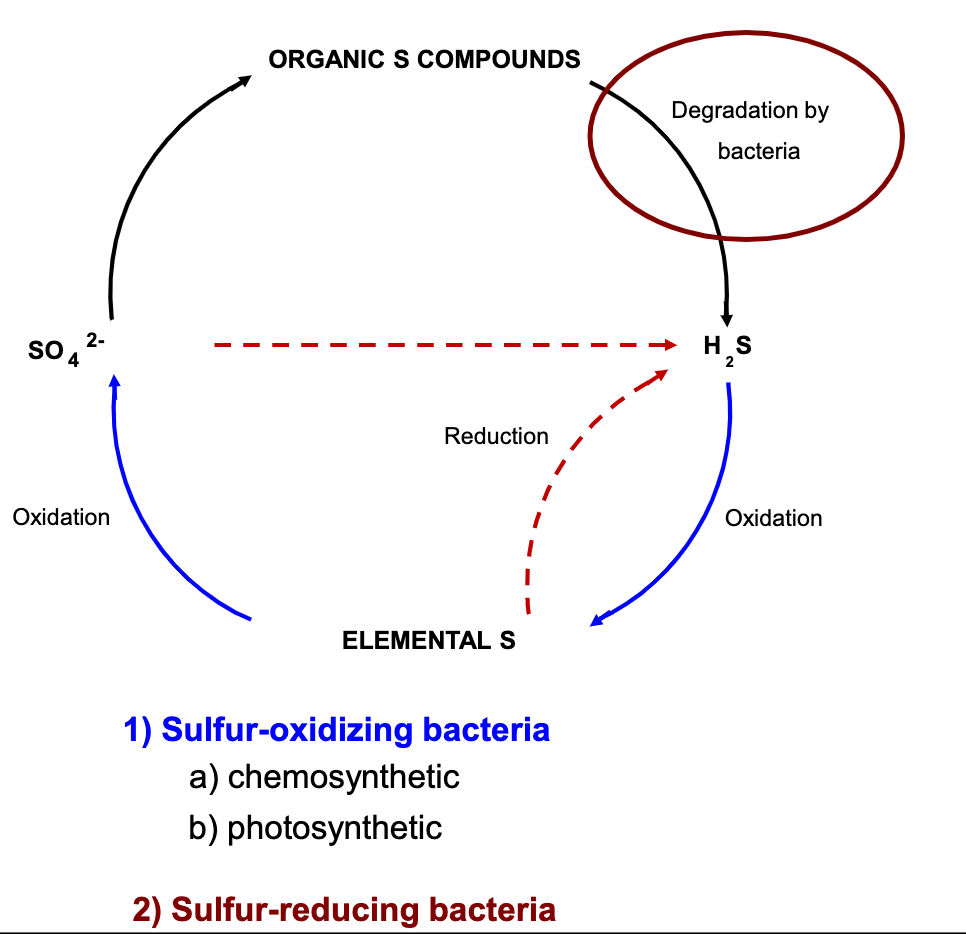
Bacteria in Lake
Bacteria can be important producers in lakes, as well as food sources
e.g. foor for zooplankton
Silica Cycle
Silica mainly important for one dominant algal group: the DIATOMS
silica is a major component of their cell walls (= frustules)
Chrysophyte algae also have some silica requirements
all chrysophytes form siliceous resting stages call stomatocytes
some chrysophyte genera are characterized by external siliceous scales
Silica utilization (or uptake) - Si that disappears from the water column as the result of production of biogenic Si
Silica limitation - [Si] where growth becomes limiting (<0.4 mg/L)
Silica depletion - long-term biogechemical decrease in [Si]
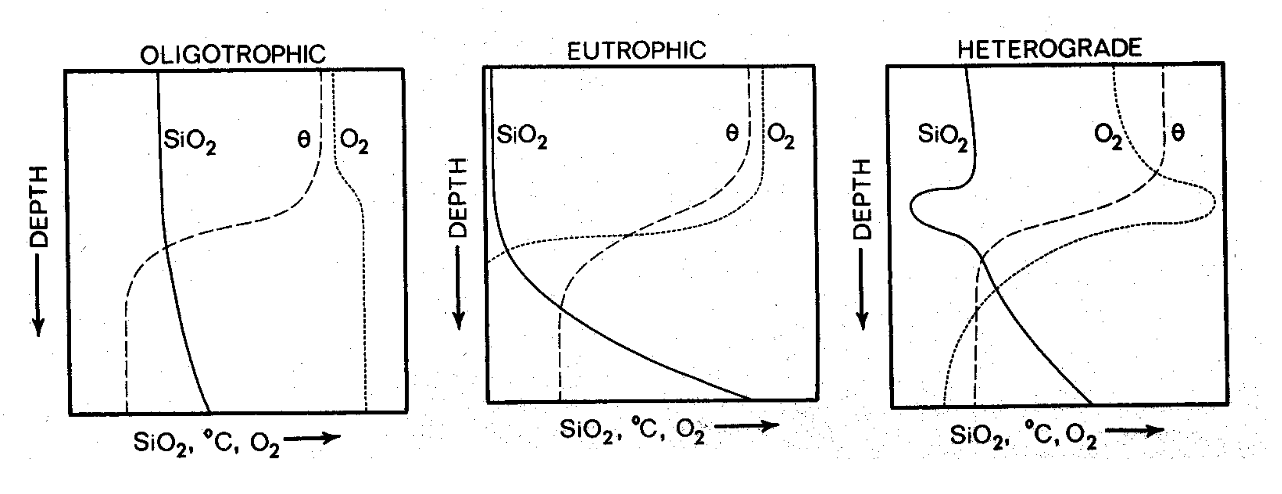
Oligotrophic lakes use a lot of silica and phosphorus is limiting
Eutrophic lakes use a lot of phosphorus which use up the very limiting silica
Heterograde lakes have algae blooms that photosynthesize and diatom blooms leading to an increase in both silica and phosphorus
Diatom changes in a lake can often be linked to SiO2 levels
Coupling of Silica and Phosphorus Cycles
Why are [Si] so low in lake Ontario and so high in lake Superior?
Lake Superior is an oligotrophic lake, so diatoms don’t get to high of a biomass, meaning they can’t and won’t use up all the silica because they don’t have enough phosphorus
Lake Ontario is a mesotrophic lake, so diatoms will use up more silica due to the higher availability of phosphorus because they have a greater biomass
Nutrients
Some nutrients can move “uphill” or against gravity in the water column
1) Aquatic insects (e.g. larval stages in water, remove nutrients, emerge as flying insects)
2) Aquatic invertebrates and some fish migrate up and down the water column (e.g. feed in hypolimnion, excrete in epilimnion)
3) Macrophytes (can take up nutrients from sediments, and then release them to water coloum)
4) Birds (e.g., Arctic seabirds) feed in the ocean, but return to land to nest
nutrient transport (via guano), as well as other sources (e.g., dead bodies)
5) Anadromous fish (like sockeye salmon), hatch in lakes (nursery lakes), then migrate to ocean and accumulate over 95% of biomass in sea, then return to nursery lakes to spawn and die
decaying carcasses are a major source of N and P and other nutrients to nursery lakes
6) Large animals such as grizzly bears or moose, who may feed in water, but excrete on land