BIOC*2580 - 2
Lipids: Overview
Discussion focuses on three types of lipids: fatty acids, triglycerides (triacylglycerols), and phospholipids (phosphoglycerides).
Phospholipids and Phosphate Groups
Phosphoric Acid (H₃PO₄):
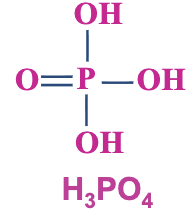
Not a carbon-containing compound.
Triprotic acid: has three dissociable protons, analogous to carboxylic acids with a COOH group.
Structure: Contains -POOH and three hydroxyl (OH) groups.
Important in biochemistry; often found in phosphorylated forms.
pKa Values: Each proton has a specific pKa value, impacting how it behaves at physiological pH (around 7).
at neutral pH it exists at equilibrium between its protonated and deprotonated forms
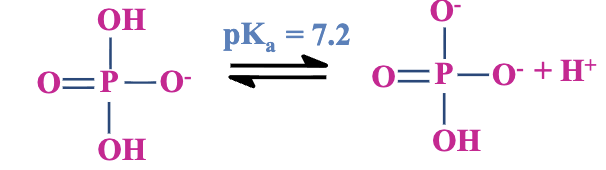
Phosphate Derivatives:
Found in various biological molecules including lipids, DNA, RNA, and proteins.
Phosphorylation increases water solubility due to the introduction of negative charges.
Phosphates contribute to molecular structure and function, enhancing solubility and playing a role in regulatory activities.
Reactions Involving Phosphoric Acid
Comparison with Carboxylic Acids:
Both can react with alcohols to form esters: Phosphoric acids yield phosphate esters (or phosphoesters).
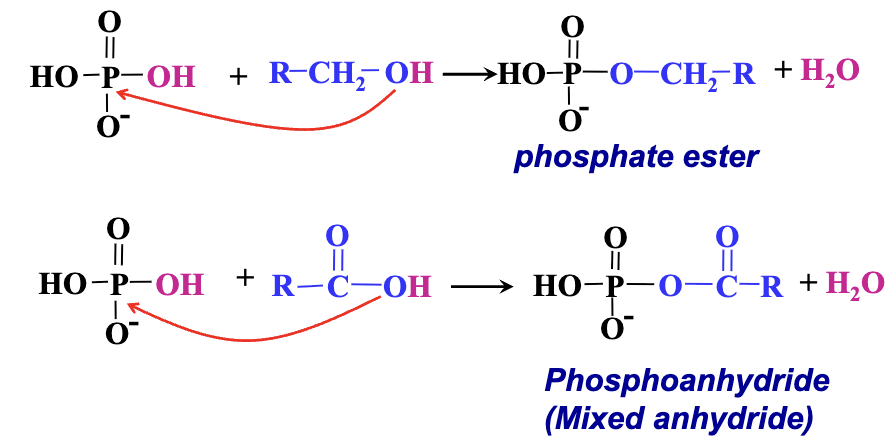
General Reaction Mechanism:
Nucleophile: Alcohol oxygen (O).
Electrophile: Phosphorus (P) in phosphoric acid.
Reaction leads to the formation of phosphate esters.
Anhydrides Formation:
Phosphoric acids can form anhydrides with other phosphoric acids or with carboxylic acids, leading to mixed anhydrides and phosphoanhydrides.
Glycerophospholipids (Phospholipids)
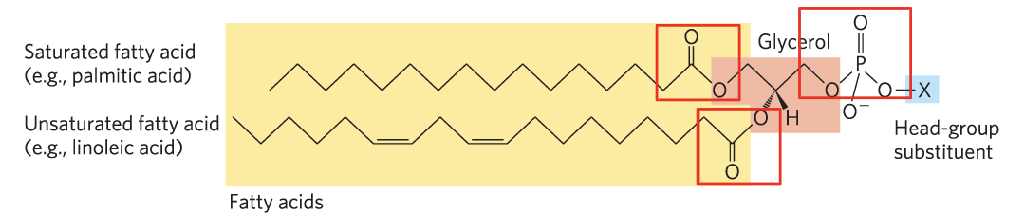
Definition: Basic components include glycerol, phosphate, and fatty acids.
Structure of Glycerophospholipids:
Glycerol backbone (three carbon atoms).
Esterified to fatty acids at carbons 1 and 2, while carbon 3 forms a bond with phosphoric acid.
Phosphate group can form phosphodiester bonds.
Amphipathic Nature:
Glycerophospholipids contain both hydrophilic (water-loving) and hydrophobic (water-repelling) regions (lipid bilator).
This characteristic is critical for biological membranes.
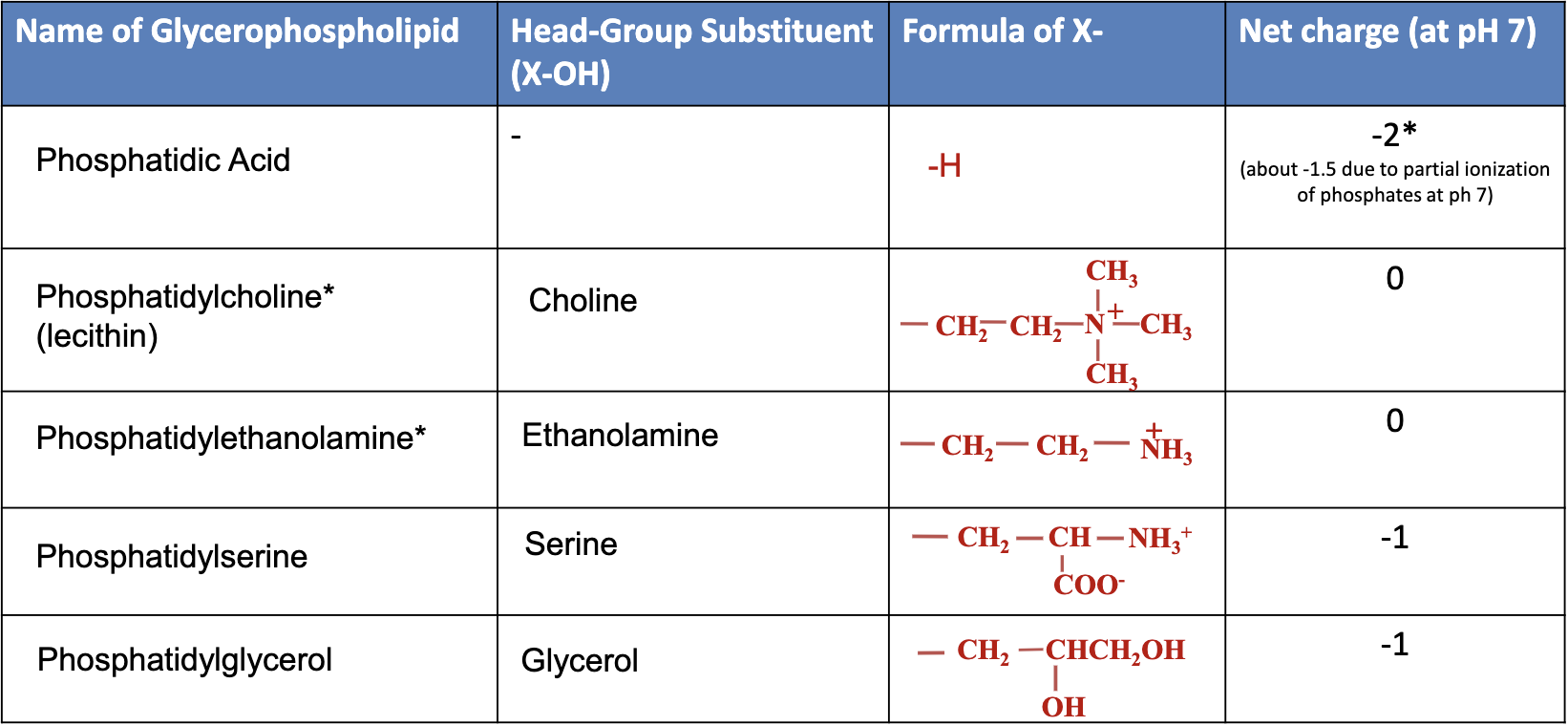
Common Types of Glycerophospholipids
Phosphatidic Acid: No additional head group, basic component.
Phosphatidylcholine: Contains choline as the head group.
Phosphatidylethanolamine: Head group is ethanolamine.
Phosphatidylserine: Head group is serine.
Phosphatidylglycerol: Head group consists of another glycerol molecule.
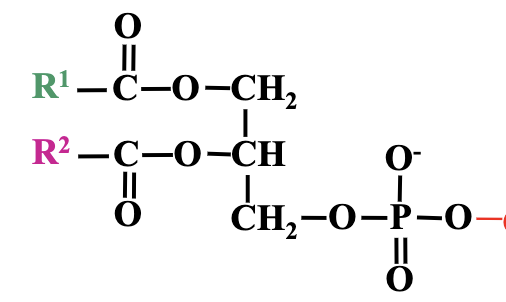
Structural Analysis and Relationships of Glycerophospholipids
Structures can vary based on the fatty acid chains (R1 and R2 groups) attached to the glycerol backbone.
Understanding these variations is essential for biochemical studies and applications in cellular membranes.
Functions of Glycerophospholipids
Major membrane constituents.
Amphipathic nature allows for membrane formation due to the hydrophobic regions facing inward and hydrophilic regions facing outward.
Lipid Aggregation in Aqueous Environments
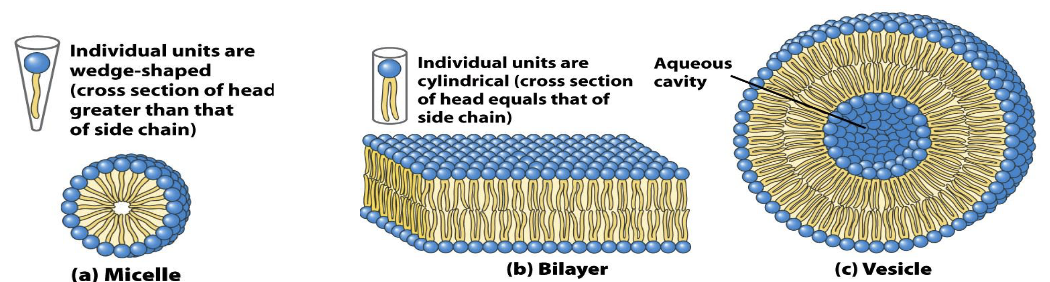
Micelles Formation:
Fatty acids in water aggregate into micelles, with hydrophilic heads outward and hydrophobic tails inward.
Phospholipid Bilayers:
Form due to the cylindrical shape of phospholipids, leading to bilayers with an aqueous cavity (vesicles/liposomes).
Lipid Analysis Techniques

Extraction Methods:
Using chloroform, methanol, and water to separate lipids from proteins and carbohydrates.
Two-phase extraction leads to the separation of lipids into chloroform (bottom layer) and polar molecules into the water layer (top).
Thin-Layer and Column Chromatography:
Used to separate triglycerides from phospholipids based on polarity.
Gas Chromatography:
Employed for analyzing fatty acid constituents after transesterification.
Carbohydrates: Introduction
Carbohydrates, also known as sugars or saccharides, are the most abundant biomolecules on Earth. They play key roles in energy storage and structural functions in organisms.
Classification:
Monosaccharides: Single sugar units such as glucose.
Disaccharides: Composed of two monosaccharides, e.g. sucrose.
Oligosaccharides: A few monosaccharide units.
Polysaccharides: Long chains of monosaccharide units, typically 20 or more.
Monosaccharide Structures and Characteristics
Monosaccharides must contain a carbonyl group (aldehyde or ketone) and at least two carbon atoms with hydroxyl groups.
Aldoses: Contain an aldehyde group.
Ketoses: Contain a ketone group.
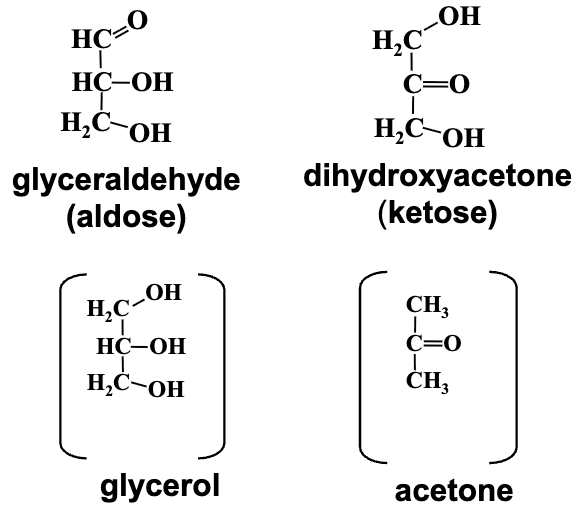
Simplest Sugars: The simplest monosaccharides consist of three carbon atoms (trioses), yielding glyceraldehyde (aldose) and dihydroxyacetone (ketose).
The most common monosaccharides include D-glucose, D-fructose, and D-galactose (hexoses)

Chemical Properties of Carbohydrates
Common herbaceous forms follow the general formula (CH₂O)n
Fischer Projection: Method for representing sugar structures in two dimensions.
D and L Designation: Based on the furthest chiral carbon from the carbonyl group; if -OH is on the right, it's a D sugar; if on the left, it's an L sugar.
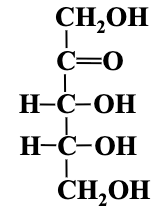
Chirality and Optical Activity
Chiral Carbons: Most monosaccharides are chiral except dihydroxyacetone.
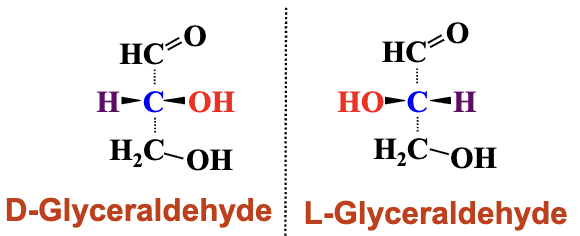
Enantiomers exhibit non-superimposable mirror images, e.g., D-glyceraldehyde vs. L-glyceraldehyde.
Have identical chemical properties but differ in their optical activities, rotating plane-polarized light in opposite directions.
Optical activity is an inherent property of chiral molecules; enantiomers rotate plane-polarized light in opposite directions.
Diastereomers, on the other hand, have more than one chiral carbon, are not mirror images of each other, and can have different physical properties, such as boiling points and solubilities.
A sugar is called a “D” sugar if the chiral carbon atom furthest away from the carbonyl group has the same configuration as D-glyceraldehyde.
Those with a configuration similar to L-glyceraldehyde are called “L” sugars.
Most (but not all) naturally occurring sugars are D-sugars
Biological Relevance and Applications
D-sugars predominate in nature, while L-amino acids are prevalent in proteins.
Understanding of carbohydrate structures is crucial for their roles in energy, metabolism, and interactions within living organisms.