23.3 Recycling within ecosystems
Energy has a linear flow through an ecosystem, it enters the ecosystem from the Sun and ultimately transferred to the atmosphere as heat. As long as the Sun continues to supply the Earth with energy, life will continue.
However nutrients have to be constantly recycled in order for plants and animals to grow, as they are used up by living organisms and there is no large external source constantly replenishing nutrients in the same way.
Decomposition
Decomposition - a chemical process in which a compound is broken down into smaller molecules, or its constituent elements. Often an essential element like nitrogen or carbon cannot be used directly used by an organism in the organic form it is in, in dead or waste matter.
This organic material must be processed into inorganic elements and compounds, which are a more usable form and returned to the environment.
Decomposer - an organism that feeds on and breaks down dead plant or animal matter, thus turning organic compounds into inorganic ones (nutrients) available to photosynthetic producers in the ecosystem. Decomposers are primarily microscopic fungi and bacteria but also include larger fungi like toadstools and bracket fungi.
Decomposers are saprotrophs because they obtain their energy from dead or waste organic material (saprobiotic nutrition) They digest their food externally by secreting enzymes onto dead organisms or organic waste matter. The enzymes break down complex organic molecules into simpler soluble molecules - the decomposers then absorb these molecules.
Through this process, decomposers release stored inorganic compounds and elements back into the environment.
Detritivores - another class of organisms involved in decomposition. They help to speed up the decay process by feeding on detritus - dead and decaying material.
They break it down into smaller pieces of organic material, which increases the surface area for the decomposers to work on. Examples of detritivores include woodlice that break down wood and earthworms that help break down dead leaves. Detritivores perform internal digestion.
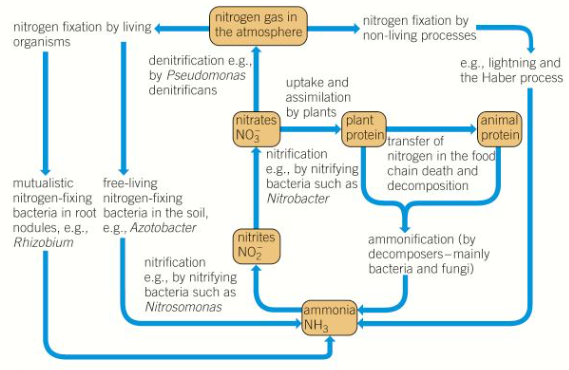
Nitrogen is abundant in the atmosphere (78%) of the air is nitrogen. However N2 cannot be taken up by plants in this form.
To be used by living organisms, nitrogen needs to be combined with other elements like oxygen and hydrogen.
Bacteria play a very important role in converting nitrogen into a form useable by plants, without bacteria, nitrogen would quickly become a limiting factor in ecosystems.
nitrates is needed for proteins, amino acids, DNA, chlorophyll in plants.
Nitrogen fixation
Nitrogen fixation bacteria like Azotobacter and Rhizobium contain the enzyme nitrogenase, which combines atmospheric nitrogen with hydrogen to produce ammonia or ammonium. - NITROGEN FIXATION
Azotobacter is an example of a free-living soil bacterium. However many nitrogen-fixing bacteria such as Rhizobium live inside root nodules. These are growth on the roots of leguminous plants such as peas, beans and clover.
They have a symbiotic mutualistic relationship with the plant as both organisms benefit
the plant gains amino acids from Rhizobium, which are produced by fixing nitrogen gas in the air into ammonia in the bacteria
the bacteria gain carbohydrates produced by the plant during photosynthesis, which they use as an energy source.
Other bacteria then convert the ammonia that is produced into other organic compounds that can be absorbed by plants.
 Nitrogen-fixing bacteria first and then nitrifying bacteria. (oxidised)
Nitrogen-fixing bacteria first and then nitrifying bacteria. (oxidised)
Nitrification
Nitrification - is the process by which ammonium compounds in the soil are converted into nitrogen-containing molecules that can be used by plants. Free-living bacteria in the soil called nitrifying bacteria are involved.
It is an oxidation reaction, so only occurs in well-aerated soil
1) Nitrifying bacteria (nitrosomonas) oxidise ammonium compounds into nitrites (NO2-)
2) Nitrobacter (another genus) oxidise nitrites into nitrates (NO3-)
These nitrate ions are highly soluble, therefore the form in which most nitrogen enters a plant.
Denitrification
In absence of oxygen, like waterlogged soil, denitrifying bacteria convert nitrates in the soil back to nitrogen gas. This is denitrification - it only happens under anaerobic conditions, the bacteria uses nitrates as a source of energy for respiration and nitrogen gas is released.
Ammonification
This is the process by which decomposers convert nitrogen-containing molecules in dead organisms, faeces, and urine into ammonium compounds.
Nitrogen cycle
Process of nitrogen fixation, nitrification, denitrification and ammonification all form the nitrogen cycle
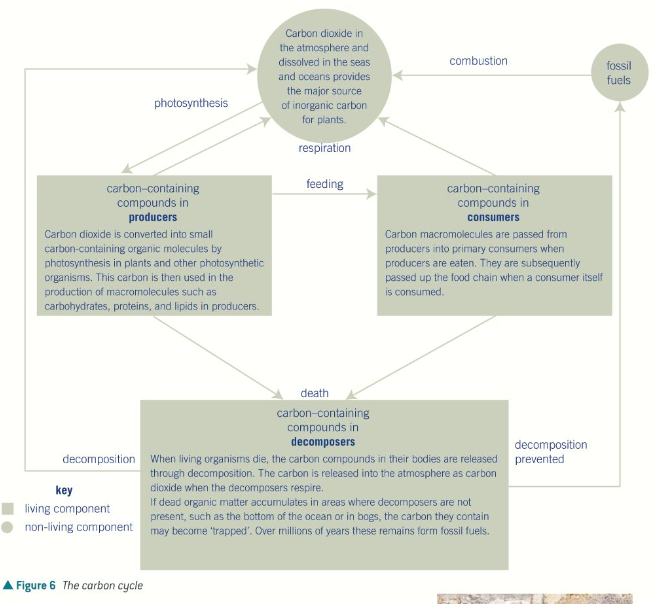
Recycling carbon
Carbon - component of all the major organic molecules present in living organisms such as fats, carbohydrates and proteins. The main source is the atmosphere. Despite only making 0.004% of the atmosphere, there is a constant cycling of carbon between the atmosphere, the land, and living organisms
Fluctuations in atmospheric carbon dioxide
Carbon dioxide levels fluctuate throughout the day.
Photosynthesis only takes place in the light, and so during the day photosynthesis removes carbon dioxide from the atmosphere. Respiration however is carried out by all living organisms throughout the day and night, releasing carbon dioxide at a relatively constant rate into the atmosphere, therefore the rates are higher at night than during the day.
Localised carbon dioxide levels also fluctuate seasonally. Carbon dioxide levels are lower in summer than winter, as photosynthesis rates are higher but also respiration is higher as endotherms need to maintain their internal body temperature.
Over the past 200 years, global atmospheric carbon dioxide levels have increased due to
the combustion of fossil fuels - which has released CO2 back into the atmosphere from carbon that had previously been trapped for millions of years below earth’s surface.
deforestation - which has removed significant quantities of photosynthesising biomass from earth, less CO2 is removed, often they are burnt releasing even more CO2 into the atmosphere.
Increased levels of CO2 in the atmosphere trap more thermal energy in the atmosphere, through human activities it has contributed to global warming.
Amount of CO2 dissolved in seas and ocean is affected by temperature - higher the temperature breaks the bonds forming between water and CO2 + less gas is then dissolved.
Global warming therefore reduces the carbon bank in the ocean and releases more CO2 into the atmosphere contributing to the process in a positive feedback.
Levels have varied over million of years, to gain information samples are taken from deep within glacier. (air bubbles reflect the composition of atmosphere at this point)