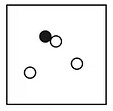
Elements, compounds and mixtures
Elements
made of only one type of atom
cannot be broken down into simpler substances by chemical methods
fixed melting and boiling points

Compounds
made of two or more elements chemically combined together in a fixed composition by mass
different properties from the elements that form it
form with an energy change
separated by chemical methods such as electrolysis or thermal decomposition
fixed melting and boiling point
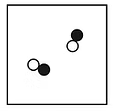
Mixtures
made up of two or more substances (elements and compounds) that can be separated by physical methods
not chemically combined together
have similar chemical properties as components
form with little or no energy change
can be separated by physical methods such a as filtration, distillation, evaporation, chromatography etc
melt and boil over a range of temperatures