Atoms
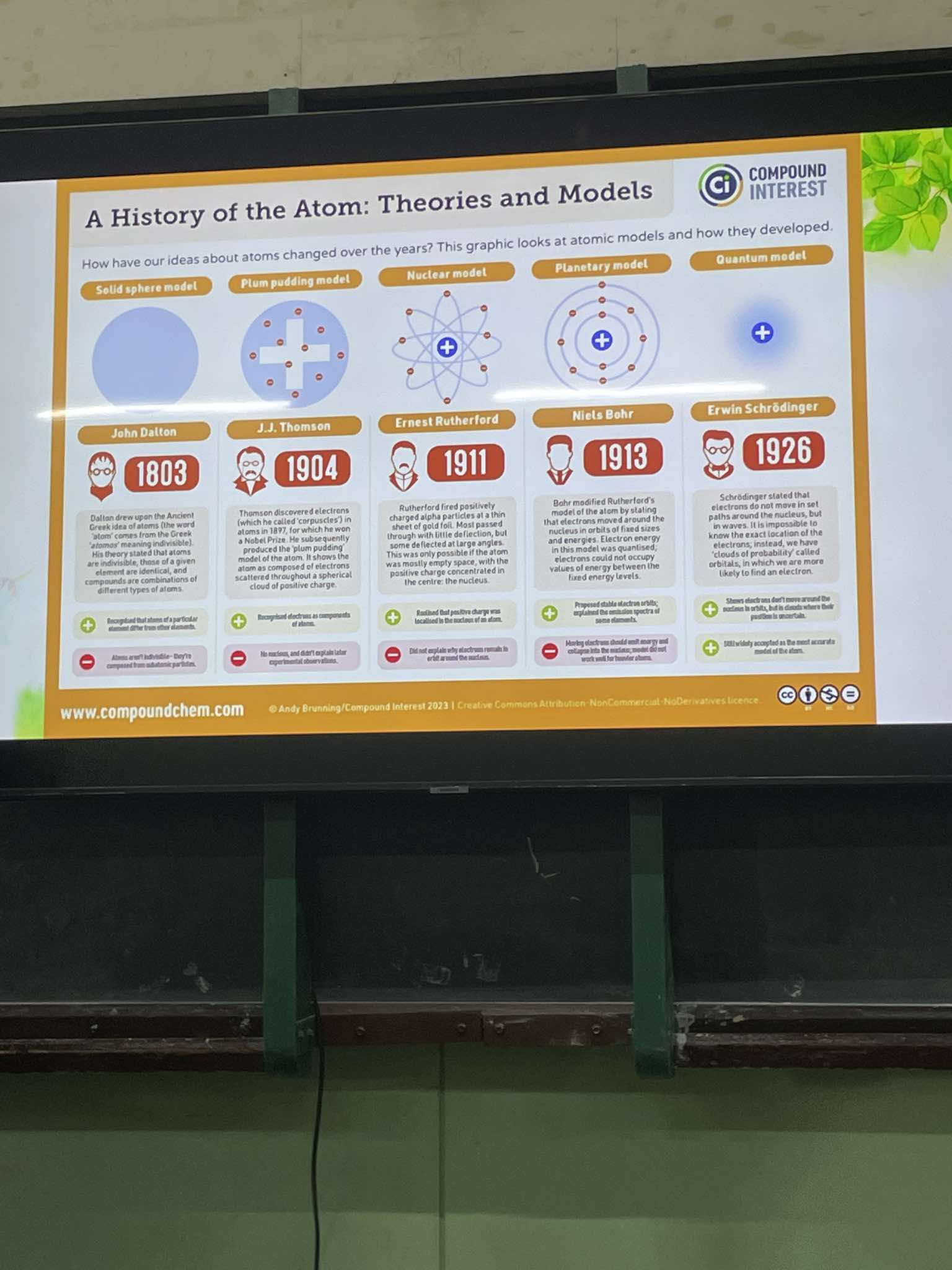
The lighter the color, the less likely the electron is there. Likewise, the brighter the color, the more likely the electron is there
Neutron is heaviest subatomic particle among the given subatomic particles with mass of 1.008 amu while proton have mass of 1 amu.
The atomic number is the identity of your element = number of protons and neutrons
Neutral: no charge - If you see a positive sign on the top right side, it is the sign of a charge
Al³+