D1.3 Mutations and gene editing Notes
D1.3.1 gene mutations as structural changes to genes at the molecular level
Gene Mutations Alter The Instructions For Building Proteins
Definition
Gene mutationA gene mutation is a permanent change in the nucleotide sequence of DNA, affecting the structure and function of proteins.
Gene mutations can be classified into three main types:
Substitutions
Insertions
Deletions.
Analogy
Imagine reading a book where a single letter is changed, added, or removed.
The meaning of the sentence could stay the same, change slightly, or become completely nonsensical.
This is exactly how gene mutations work.
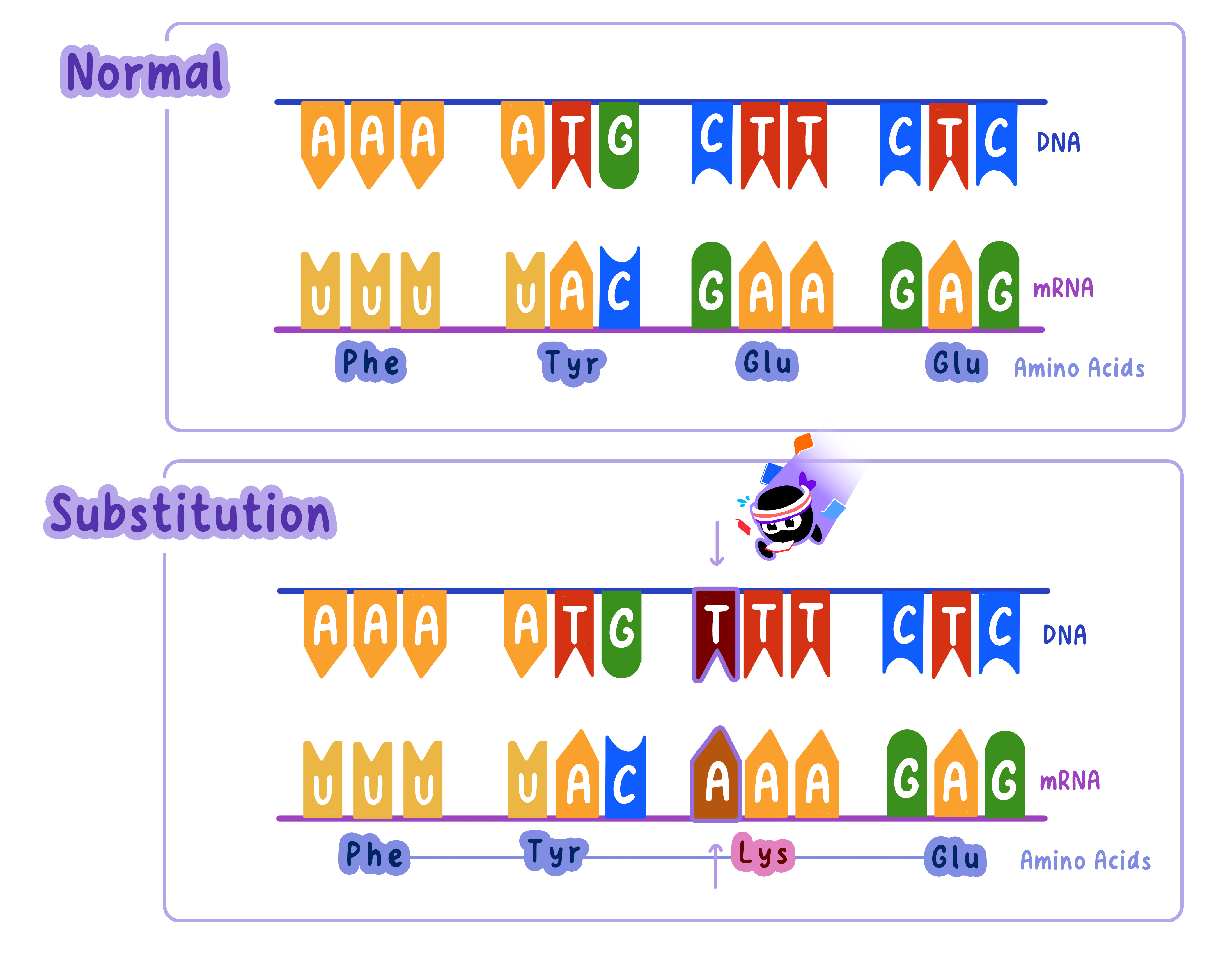
Substitution Mutations: Swapping One Base for Another
In substitution mutations, one nucleotide is replaced by another.
This is like swapping a letter in a word: "cat" becomes "bat."
Hint
Since substitution mutations affect only one codon, their impact is often less severethan insertions or deletions.
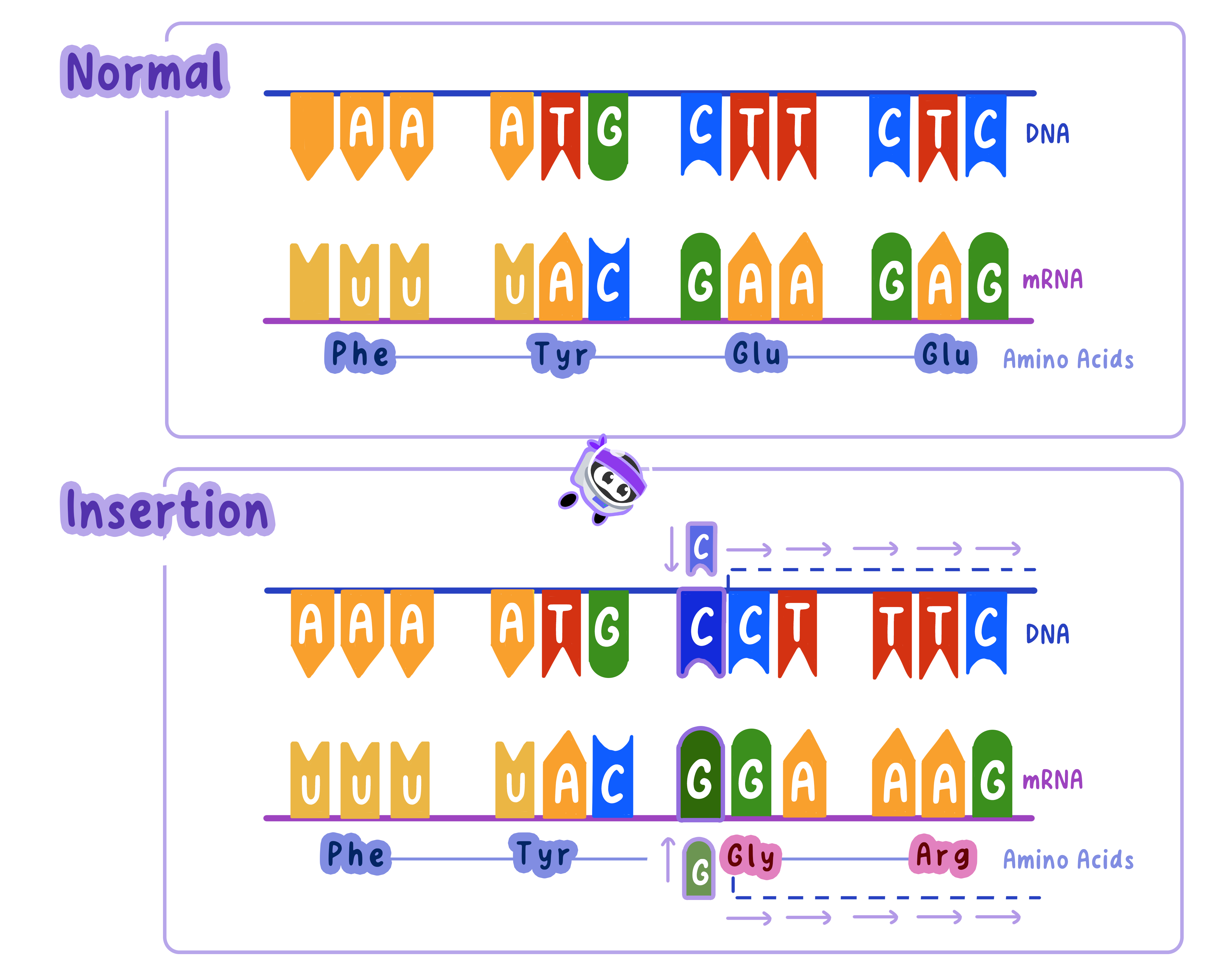
Insertion Mutations: Adding Extra Bases
In insertion mutations, one or more nucleotides are added to the DNA sequence.
This shifts the reading frame of the genetic code, altering every codon downstream of the mutation.
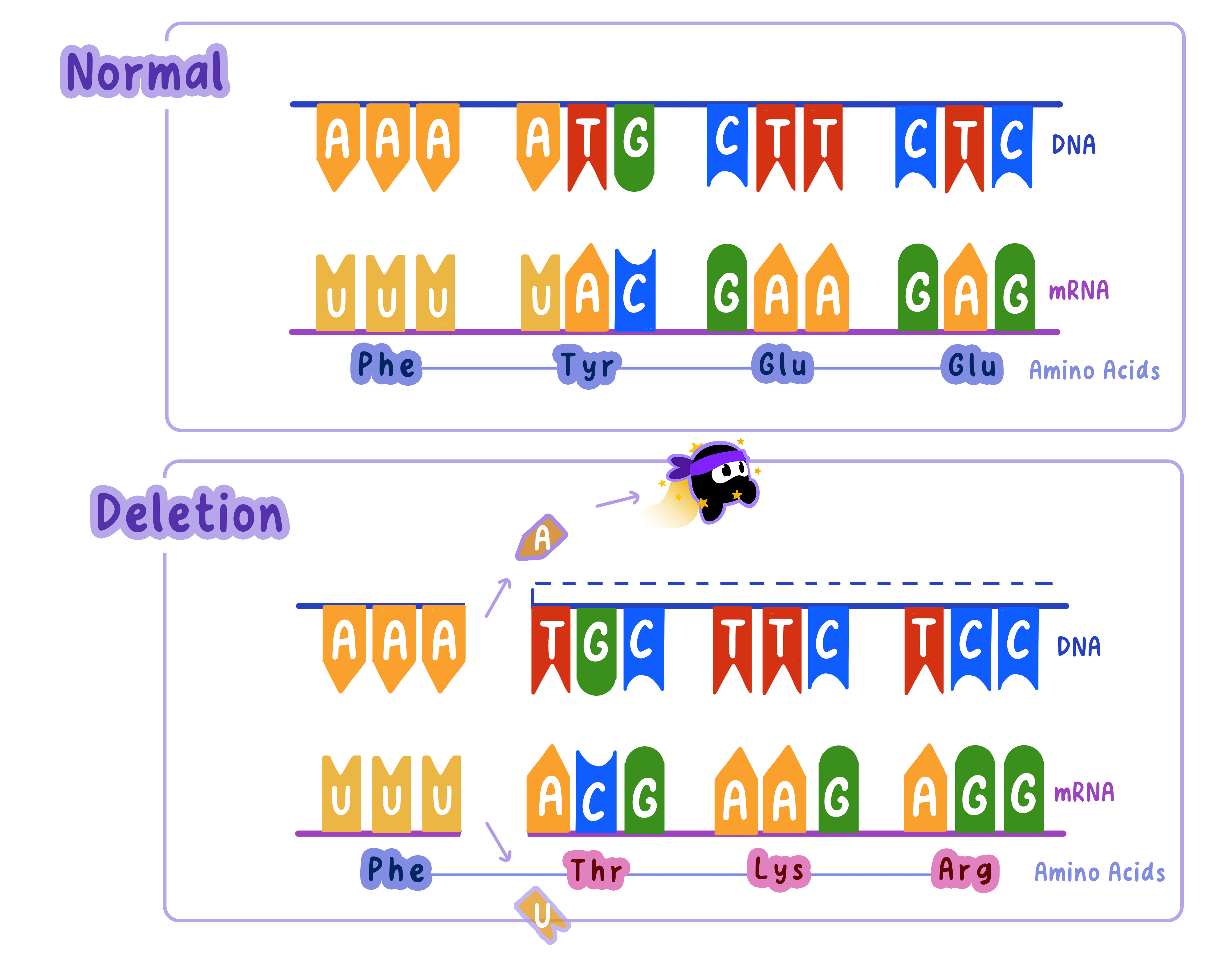
Deletion Mutations: Removing Bases
Deletion mutations occur when one or more nucleotides are removed from the DNA sequence.
Like insertions, deletions can cause frameshifts unless the number of deleted bases is a multiple of three.
Hint
Frameshift mutations (caused by insertions or deletions) are usually more severe than substitutions because they affect the entire protein sequence downstream of the mutation.
Why Mutations Matter
Mutations can have a wide range of effects:
Neutral: Many mutations occur in non-coding regions of DNA or result in silent mutations that do not affect the protein.
Harmful: Mutations can disrupt protein function, leading to genetic disorders or diseases like cancer.
Beneficial: Rarely, mutations can improve a protein's function or confer an advantage, such as resistance to certain diseases.
Example
The CCR5-Î32 mutation provides resistance to HIV by altering a receptor on immune cells.
Tok
How do you think the randomness of mutations influences the balance between stability and change in living organisms?
Could this concept apply to other areas, such as technology or society?
Self Review
Distinguish between insertion, deletion, and substitution mutations.
What does a "point mutation" refer to?
Which type of mutation is most severe?
D1.3.2 consequences of base substitutions
Consequences of Base Substitutions
Imagine reading a sentence where a single letter is changed: "The dog ate the pie" becomes "The dog ate the pig."
The meaning shifts, but the overall structure remains intact.
Similarly, base substitutions in DNA can alter genetic instructions, with effects ranging from negligible to life-altering.
Definition
Base substitutionA base substitution is a gene mutation where one nucleotide in the DNA sequence is replaced by another.
Single-Nucleotide Polymorphisms (SNPs): Variations in the Genetic Code
Single-nucleotide polymorphisms (SNPs) are the most common type of genetic variation in humans.
They occur when a single nucleotide in the DNA sequence is altered.
SNPs can be found in both coding and non-coding regions of the genome.
Tip
The genetic code is like a safety net: its redundancy helps protect against mutations.
Types of Base Substitution Mutations
1. Same-Sense Mutations
These mutations change one codon into another that codes for the same amino acid.
Because of the degeneracy of the genetic code, they have no effect on the protein.
Note
Don't confuse mis-senseand nonsense mutations.
Mis-sense mutations change one amino acid, while nonsense mutations create a stop codon.
Example
A mutation changing CAG (glutamine) to TAG (stop codon) would truncate the protein.
Consequences of Base Substitutions
1. Neutral Mutations
Many base substitutions have no effect on the organism,
Especially if they occur in non-coding regions or result in same-sense mutations.
2. Harmful Mutations
Mis-sense and nonsense mutations can disrupt protein function, leading to genetic disorders or diseases.
Example
Cystic fibrosis is often caused by a nonsense mutation in the CFTR gene, resulting in a truncated, nonfunctional protein.
3. Beneficial Mutations
Rarely, base substitutions improve protein function or confer an advantage, such as resistance to disease.
Example
The CCR5-Î32 mutation provides resistance to HIV by altering a receptor on immune cells.
Single-Nucleotide Polymorphisms (SNPs) and Genetic Diversity
SNPs are a major source of genetic variation in populations.
They can influence traits, susceptibility to diseases, and responses to medications.
Example
SNPs in the BRCA1 gene are associated with an increased risk of breast and ovarian cancer.
Note
Not all SNPs have functional consequences.
Many are neutral and serve as markers for studying genetic variation.
The Role of SNPs in Evolution
While most mutations are neutral or harmful, beneficial mutations drive evolution by providing new traits that can be selected for in changing environments.
Tok
How do scientists determine whether a mutation is beneficial, neutral, or harmful?
Consider the role of the environment in shaping these outcomes.
Reflection and Review
Base substitutions are a key source of genetic variation, with effects ranging from silent to severe.
The degeneracy of the genetic code acts as a buffer, reducing the impact of many mutations.
SNPs contribute to genetic diversity and are essential for understanding evolution and human health.
D1.3.3 consequences of insertions and deletions
Consequences of Insertions and Deletions
Insertions add extra nucleotides into the DNA sequence.
Deletions remove nucleotides from the DNA sequence.
Both disrupt the genetic code, often with severe consequences.
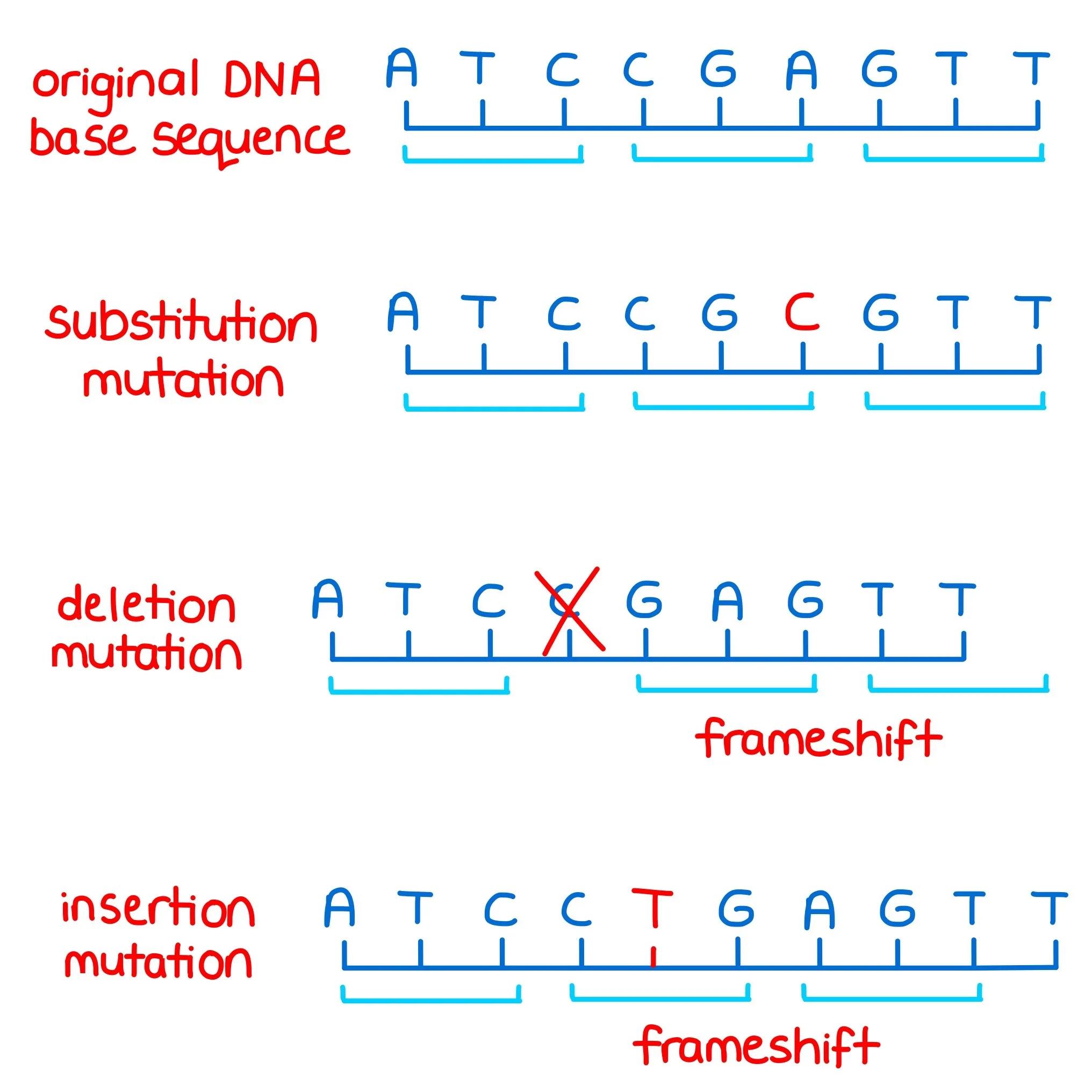
Frameshift Mutations: A Domino Effect
DNA is read in triplets called codons, each coding for a specific amino acid.
Inserting or deleting a nucleotide shifts the reading frame, altering every codon downstream.
Note
Frameshifts are not the same as substitutions.
Substitutions affect only one codon, while frameshifts disrupt the entire sequence.
Tip
If the insertion or deletion occurs near the start of the gene, the effects are usually more severe because most of the protein is altered.
Major Insertions and Deletions: Structural Chaos
Larger insertions or deletions (multiples of three) don't cause frame shifts but still disrupt protein function.
They add or remove entire amino acids, altering the protein's shape and stability.
Example
Imagine a zipper missing several teeth.
It might still work, but not as efficiently or reliably.
Consequences of Major Changes
Loss of Function: The protein may become unstable or unable to bind to other molecules.
Gain of Function: Rarely, these mutations create new functions, but they are usually harmful.
Note
Even if the reading frame is preserved, the addition or removal of amino acids can disrupt critical regions of the protein, such as active sites or binding domains.
Why Insertions and Deletions Are Rarely Beneficial
Most mutations are neutral or harmful because genes have evolved to perform specific functions.
Random changes are unlikely to improve these finely tuned systems.
Tok
How do we balance the risks and benefits of mutations in evolution?
Could a harmful mutation today lead to a beneficial adaptation in the future?
Exceptions: When Insertions or Deletions Don't Cause Frameshifts
If the number of nucleotides added or removed is a multiple of three, the reading frame is preserved.
However, the protein still gains or loses amino acids, which can disrupt its function.
Example
A deletion of three nucleotides might remove an amino acid essential for the protein's stability or activity.
Self Review
What is the difference between a frameshift mutation and a non-frameshift mutation? How do their effects on protein function differ?
Likelihood of Polypeptides Ceasing to Function
Frameshift Mutations: Highly likely to produce nonfunctional proteins due to widespread changes in the amino acid sequence.
Major Insertions/Deletions: Often result in loss of function by disrupting the protein's structure or removing critical regions.
Analogy
Think of a polypeptide as a carefully constructed building.
A frameshift mutation is like removing a foundation pillar, causing the entire structure to collapse.
A major insertion or deletion is like adding or removing a wall--it may not collapse the building, but it can severely weaken it.
Reflection and Broader Implications
Insertions and deletions highlight the precision required for genetic stability.
They also underscore the role of mutations in evolution, where even harmful changes can drive diversity over time.
Tok
How does our understanding of mutations influence ethical debates about gene editing?
Should we intervene to prevent harmful mutations, or is genetic variation too important for evolution?
Self Review
Why are frameshift mutations generally more harmful than base substitutions?
Can you think of a scenario where a deletion might be beneficial?
D1.3.4 causes of gene mutation
Causes of Gene Mutation
Gene mutations are changes in the DNA sequence that can alter the function of a gene.
These mutations can arise from:
Errors in DNA replication or repair.
External agents called mutagens.
Example
Adenine (A) is paired with cytosine (C) instead of thymine (T).
1. Errors in DNA Replication
During DNA replication, DNA polymerase occasionally incorporates the wrong nucleotide, resulting in mismatched base pairs.
Most replication errors are corrected by DNA polymerase's proofreading ability, but a small fraction escape repair, leading to permanent mutations.
Example
Instead of pairing adenine (A) with thymine (T), DNA polymerase might pair adenine with cytosine (C).
2. Errors in DNA Repair
DNA is constantly subjected to damage from metabolic byproducts and environmental factors.
Repair mechanisms like base excision repair and nucleotide excision repair fix most damage, but:
If the repair system fails or introduces errors, mutations can occur.
Example
Faulty repair of double-strand breaks can result in deletions, duplications, or chromosomal rearrangements.
Mutagens: External Causes of Mutation
Mutagens are agents that increase the likelihood of mutations.
They can be classified into two main types:
Chemical Mutagens
Radiation
Chemical Mutagens
Chemical substances that interfere with DNA structure or function.
Base Analogues: Molecules resembling normal DNA bases but pair incorrectly during replication.
Alkylating Agents: Add alkyl groups to DNA bases, causing mispairing or strand breaks.
Deaminating Agents: Remove amino groups from bases, altering their pairing properties.
Intercalating Agents: Insert between DNA bases, distorting the helix and causing insertions or deletions.
Warning
Students often assume that all mutations are caused by external factors, such as radiation or chemicals.
They may overlook the fact that mutations can also arise naturally due to errors in DNA replication or repair.
Mutagenic Radiation
Forms of radiation that damage DNA, causing mutations.
Ionizing Radiation:
Includes X-rays, gamma rays, and alpha particles.
These high-energy forms cause double-strand breaks, leading to deletions, translocations, or chromosomal rearrangements.
Non-Ionizing Radiation:
Includes UV light.
UV radiation causes thymine dimers, where two adjacent thymine bases bond together. This disrupts normal base pairing and hinders DNA replication.
Example
Nitrosamines can convert cytosine (C) into uracil (U), which pairs with adenine (A) instead of guanine (G).
The Combined Impact of Errors and Mutagens
While replication errors and DNA repair failures are natural sources of mutations, exposure to mutagens amplifies the mutation rate.
Example
A single replication error might cause a base-pair substitution.
UV-induced thymine dimers further hinder replication, increasing the chance of additional errors.
Real-World Implications of Gene Mutations
Understanding the causes of gene mutations has profound implications for human health and environmental science.
Cancer: Mutations in genes controlling cell growth and division can lead to uncontrolled proliferation, resulting in cancer.
eg. UV-induced mutations in skin cells increase the risk of melanoma.
Genetic Disorders: Mutations in germline cells can lead to inherited diseases like cystic fibrosis or sickle-cell anemia.
Environmental Concerns: Pollutants like pesticides and industrial chemicals act as mutagens, increasing the mutation rate in wildlife and potentially affecting ecosystems.
Evolution: Mutations are the raw material for evolution, providing the genetic variation needed for natural selection.
D1.3.5 randomness in mutation
Randomness in Mutation
Imagine rolling a die.
You can't predict the outcome, but you know each number has a chance of appearing.
Mutations in DNA work similarly, they are unpredictable but follow certain patterns.
Definition
Mutation A mutation is a change in the DNA sequence, which can alter the instructions for building proteins or regulating cellular processes.
Why Are Mutations Random?
Mutations occur due to errors in DNA replication or exposure to mutagens (e.g., radiation, chemicals).
These events are unplanned and uncontrolled by the organism.
Analogy
Think of mutations as typos in a book.
They happen by accident, not because the writer intended to change the story.
Living Organisms Cannot Intentionally Change Specific DNA Bases to Achieve a Desired Trait.
Mutations are blind to their consequences they don't "know" if they will be beneficial, neutral, or harmful.
Note
Don't confuse natural mutations with gene editing techniques like CRISPR, which are human-engineered tools for making specific changes in DNA.
Why Some Bases Are More Likely to Mutate
While mutations are random, certain factors make some DNA regions more prone to change:
Chemical Instability: Bases like cytosine can undergo spontaneous changes, such as deamination, which converts cytosine to uracil.
Hotspots: Some DNA sequences, like repetitive regions, are more likely to experience errors during replication.
Exposure to Mutagens: Regions of DNA that are more exposed or less protected by proteins may be more susceptible to damage.
Example
The BRCA1 gene, involved in DNA repair, has specific regions that are more prone to mutations, increasing the risk of cancer.
The Role of Randomness in Evolution
Mutations are a key driver of genetic variation, which fuels evolution.
Most mutations are neutral or harmful, but occasionally, a mutation provides a benefitthat enhances survival or reproduction.
Example
The CCR5-Î32 mutation provides resistance to HIV by altering a receptor on immune cells.
This mutation likely arose randomly and spread in populations where it offered a survival advantage.
Note
Mutations in germ cells(sperm and eggs) can be passed to offspring, contributing to evolutionary change. Mutations in somatic cells(body cells) are not inherited.
Implications of Random Mutations
Unpredictability: Mutations cannot be predicted or controlled naturally.
Low Probability of Benefit: Most mutations disrupt well-adapted genes, making beneficial mutations rare.
Essential for Diversity: Despite their risks, mutations are vital for creating the genetic diversity needed for evolution.
Tok
How does the randomness of mutations challenge the idea of a perfectly designed organism?
Could this concept influence our understanding of evolution and natural selection?
Reflection and Review
Mutations are random changes in DNA that occur without direction or purpose.
Some DNA regions are more prone to mutations due to chemical instability or environmental exposure.
Random mutations are a double-edged sword: they can cause genetic diseases but also drive evolution and adaptation.
D1.3.6 consequences of mutation in germ cells and somatic cells
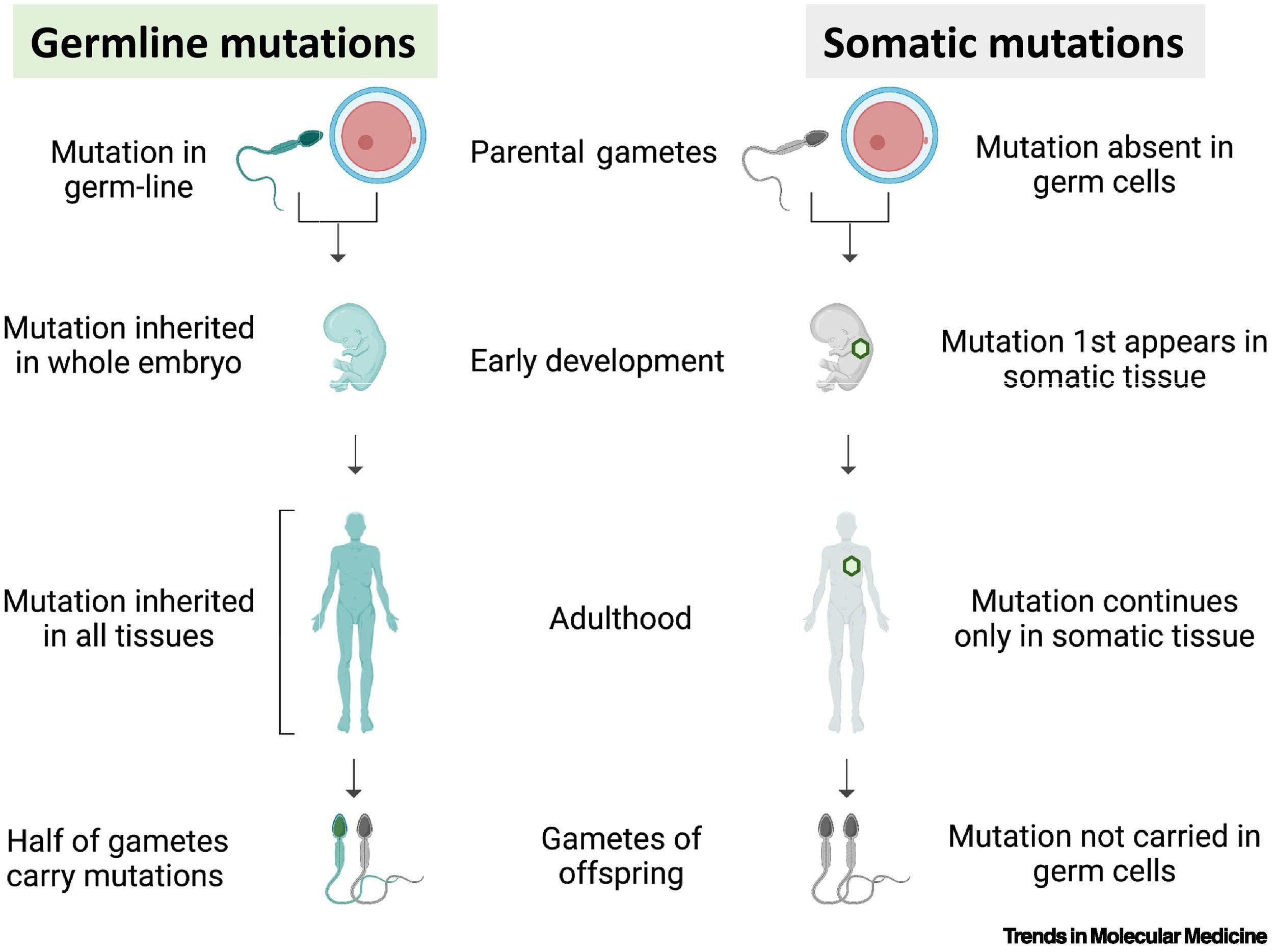
Mutations Occur in Two Primary Cell Types
Mutations can occur in two primary cell types:
Germ Cells: These give rise to gametes (sperm and eggs).
Somatic Cells: These make up the rest of the body.
The impact of a mutation depends on where it occurs.
Tip
Mutations in germ cells can be inherited, affecting future generations.
Mutations in somatic cellsaffect only the individual and are not passed on.
Mutations in Germ Cells: Inheritance and Evolution
Germ Cells and Gametes
Germ cells are specialized cells in the gonads (ovaries and testes) that undergo meiosisto produce gametes.
Mutations in these cells can create new alleles, which may be:
Neutral: No noticeable effect.
Harmful: Cause genetic diseases.
Beneficial: Enhance survival or reproduction.
Warning
It's a common misconception that mutations are always harmful. In reality, they are the source of all genetic diversity.
Mutations in Somatic Cells: Cancer and Other Effects
Somatic Cells and Limited Impact
Mutations in somatic cells affect only the individual and are not inherited.
These mutations are often neutral or cause localized effects, such as cell death.
Cancer: A Major Consequence
Some mutations disrupt genes that control the cell cycle, leading to uncontrolled cell division and tumor formation.
Key genes involved include:
Proto-oncogenes: Mutations can activate these as oncogenes, promoting excessive cell division.
Tumor Suppressor Genes: Mutations can inactivate these, removing checks on cell growth.
Note
Cancer often requires multiple mutations in different genes, which is why it typically develops over time.
Comparing Germ Cell and Somatic Cell Mutations
Germ Cell Mutations | Somatic Cell Mutations |
|---|---|
Affect future generations | Affect only the individual |
Can cause inherited genetic diseases | Cannot be inherited |
Drive evolution and genetic diversity | Do not contribute to evolution |
Examples: cystic fibrosis, sickle cell anemia | Examples: cancer, localized cell death |
Tok
How do our ethical perspectives influence decisions about using gene editing technologies to correct mutations in germ cells or somatic cells?
Reflection and Broader Implications
Consider the balance between the risks and benefits of mutations:
Risks: Genetic diseases, cancer.
Benefits: Evolutionary adaptation, biodiversity.
How might advances in gene editing change our ability to manage these risks and harness these benefits?
D1.3.7 mutation as a source of genetic variation
How Mutations Create Genetic Variation
Definition
Mutation A mutation is a change in the DNA sequence, which can alter the instructions for building proteins or regulating cellular processes.
Mutations alter the DNA sequence, creating new alleles.
These new alleles introduce variation within a population.
Without mutations, all individuals would be genetically identical, leaving no room for adaptation.
Example
Imagine a population of beetles living in a forest.
Most are green, blending perfectly with the leaves, but a few are brown due to a genetic change.
One day, a fire sweeps through, turning the forest floor dark.
Suddenly, the brown beetles are better camouflaged, making them less likely to be eaten by predators.
Over time, the brown beetles thrive, and their population grows.
Practical Implications: Genetic Testing
Modern technology allows us to identify mutations in our DNA through genetic testing.
These tests can reveal:
Disease Risk: Mutations in genes like \text{BRCA1} can increase the risk of cancer.
Ancestry: Single-nucleotide polymorphisms (SNPs) can trace genetic heritage.
Trait Prediction: Mutations can predict traits like lactose intolerance or eye color.
Tok
How should societies balance the benefits of genetic testing with the potential for misuse or misunderstanding of the information?
Self Review
Can you think of an example where a mutation has provided a clear survival advantage to a species?
D1.3.8 gene knockout (hl)
Gene Knockout: A Powerful Tool for Understanding Gene Function
Imagine trying to understand how a car works by removing one part at a time and observing the changes.
This is the essence of gene knockout, a technique that helps scientists uncover the roles of specific genes by making them inoperative.
Definition
Gene knockout A gene knockout is a genetic technique where a specific gene is intentionally inactivated or "knocked out" to study its function.
How Does Gene Knockout Work?
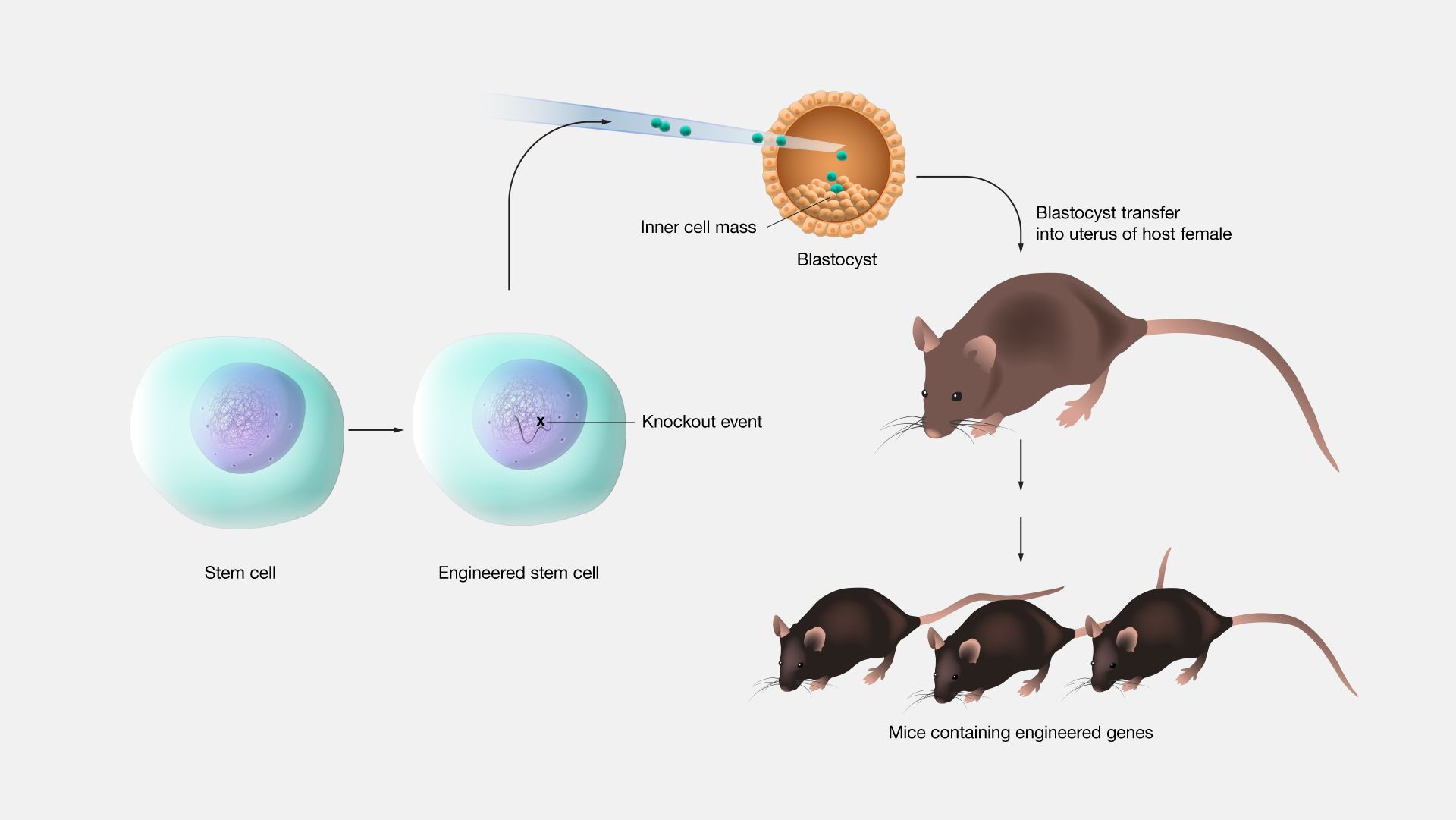
Identify the Target Gene: Scientists select a gene they want to study.
Disable the Gene: The gene is altered or removed so it can no longer produce its protein.
Observe the Effects: Researchers study the organism to see what changes occur, revealing the gene's role.
Tip
Gene knockout doesn't just delete a gene, it strategically disrupts it to ensure the organism survives for study.
Why Use Gene Knockout?
Understanding Biological Processes: By observing changes in knockout organisms, scientists can link genes to specific traits or functions.
Modeling Human Diseases: Knockout mice are often used to study genetic disorders, helping develop treatments.
Drug Development: Identifying genes critical for disease pathways can lead to targeted therapies.
Tip
When studying knockout organisms, always compare them to a wild-type (normal) organism to identify differences caused by the missing gene.
Limitations and Challenges
Compensatory Mechanisms: Other genes may compensate for the missing gene, masking its true function.
Lethality: Knocking out essential genes can kill the organism, making it difficult to study their roles.
Context-Dependent Functions: A gene's role might vary across tissues or environmental conditions.
Note
Don't assume that a gene's function in one species or context will be identical in another.
Always consider the broader biological context.
Applications of Gene Knockout
Medicine: Identifying genes involved in diseases like cancer or diabetes.
Agriculture: Developing crops resistant to pests or environmental stress.
Basic Research: Unraveling fundamental biological processes, such as development or metabolism.
Tok
How might the ethical considerations of gene knockout differ between studying human diseases and creating genetically modified crops?
Reflect on the broader societal implications.
Reflection and Broader Implications
Gene knockout has transformed our understanding of genetics and biology.
By systematically disabling genes, scientists can unravel the complex interactions that drive life.
D1.3.9 use of the crispr sequences and the enzyme cas9 in gene editing (hl)
How Does CRISPR-Cas9 Work?
Guide RNA (gRNA): A short RNA sequence guides Cas9 to the specific DNA target.
Cas9 Enzyme: Acts as molecular scissors, cutting the DNA at the target site.
DNA Repair: The cell repairs the break, allowing scientists to:
Disable a gene (knockout).
Insert a new gene.
Correct a mutation.
Analogy
Think of CRISPR-Cas9 as a GPS-guided pair of scissors.
The gRNA is the GPS, directing Cas9 to the exact location in the DNA where a cut is needed.
Steps of CRISPR-Cas9 Gene Editing
Design the gRNA: Scientists create a gRNA complementary to the target DNA sequence.
Introduce the System: The gRNA and Cas9 are delivered into the cell.
Target Recognition and Cutting:
The gRNA binds to the target DNA.
Cas9 cuts both DNA strands at the specified location.
DNA Repair:
Non-Homologous End Joining (NHEJ): Repairs the break but often introduces small errors, disabling the gene.
Homology-Directed Repair (HDR): Uses a template to insert or correct DNA sequences.
Tip
When designing gRNA, ensure it is highly specific to avoid off-target effects, which can cause unintended mutations.
Example: Treating Sickle Cell Disease
Sickle cell disease is caused by a single base mutation in the HBB gene, leading to abnormal hemoglobin.
CRISPR-Cas9 has been used to:
Cut the mutated gene in stem cells from a patient's bone marrow.
Introduce a corrected version of the gene via HDR.
Reintroduce the edited cells into the patient, producing healthy red blood cells.

Example
In 2020, a patient with sickle cell disease received CRISPR-edited stem cells.
After treatment, the patient showed significant improvement, with reduced symptoms and fewer hospitalizations.
Ethical Considerations of CRISPR
Human Germline Editing: Editing embryos could prevent genetic diseases but raises concerns about unintended consequences and "designer babies."
Biodiversity and Ecosystems: Using CRISPR to control pests (e.g., mosquitoes) might disrupt ecosystems.
Equity and Access: Will gene-editing therapies be accessible to all, or only to those who can afford them?
Tok
How should society balance the potential benefits of CRISPR with its ethical risks?
Consider the role of cultural and societal values in shaping these decisions.
International Regulation
Diverse Regulations: Countries have varying laws on gene editing.
Need for Harmonization: Scientists advocate for global guidelines to ensure safe and ethical use of CRISPR.
Example
Japan allows germline editing for research, while Canada prohibits it.
Reflection and Future Directions
CRISPR-Cas9 has transformed biology, offering solutions to genetic diseases and agricultural challenges.
However, its use must be guided by ethical considerations and robust regulations.
Self Review
Can you explain how CRISPR-Cas9 targets and edits specific DNA sequences?
What are the ethical implications of using this technology in humans?
D1.3.10 hypotheses to account for conserved or highly conserved sequences in genes (hl)
Hypotheses to Account for Conserved or Highly Conserved Sequences in Genes
Conserved sequences are identical or similar across species, while highly conservedsequences remain unchanged over millions of years.
These sequences are critical for life, and understanding why they persist offers insights into evolution and genetic stability.
Definition
Conserved sequencesConserved sequences are DNA or protein regions that remain unchanged across species or over time, indicating their essential functional roles.
Functional Requirements for Gene Products
Functional requirements are a key reason for conservation.
Mutations in these sequences often disrupt vital processes, leading to natural selectionagainst changes.
Why Are Functional Sequences Conserved?
Many conserved sequences code for proteins or RNA molecules essential for life.
Changes in these sequences can impair their function, reducing an organism's survival and reproductive success.
Example
The Hoxgene cluster, which regulates body plan development, contains conserved regulatory sequences.
Mutations in these regions can cause severe developmental defects.
Tip
When studying conserved sequences, consider both coding and non-coding regions.
Both play critical roles in maintaining biological function.
Slower Rates of Mutation
Another hypothesis is that conserved sequences experience slower mutation rates.
This could be due to enhanced DNA repair mechanisms or structural features that protect these regions.
Why Do Some Regions Mutate Less?
DNA Repair Mechanisms: Some regions may be more accessible to repair enzymes, reducing mutation rates.
Structural Stability: DNA sequences with stable structures, such as hairpins or G-quadruplexes, may be less prone to mutations.
Example
The HACNS1 gene is highly conserved across mammals but shows rapid changes in humans, linked to unique traits like thumb development.
This suggests that mutation rates can vary even in conserved regions.
Balancing Mutation and Conservation
While mutations drive evolution, conserved sequences highlight areas where change is detrimental.
This balance between stability and variation is crucial for adaptation and survival.
Warning
Don't assume all conserved sequences are coding regions.
Many are non-coding but still play critical roles in regulation and genome stability.
Implications of Conserved Sequences
Conserved sequences offer insights into evolutionary history and functional biology.
They help identify critical genes and regulatory elements across species.
Evolutionary Insights
Highly conserved sequences act as molecular fossils, revealing shared ancestry among species.
By comparing these sequences, scientists can trace evolutionary relationships and identify key functional elements.

Example
The ribosomal RNA (rRNA)genes are highly conserved and used to construct phylogenetic trees, mapping the evolutionary history of life.
Practical Applications
Conserved sequences are targets for gene editing and biomedical research.
Understanding these regions can lead to advances in medicine, agriculture, and conservation.
Tok
How does the conservation of genetic sequences challenge the idea of randomness in evolution?
Could this reflect a deeper order in biological systems?
Reflection and Broader Questions
Conserved sequences highlight the interplay between stability and change in evolution.
By studying these regions, we gain insights into the universal principles that govern life.
Tok
In what ways do conserved sequences reflect the interconnectedness of all life?
How might this influence our ethical considerations in gene editing?