Dielectric properties at high frequencies
Learning Outcomes & Key Concepts
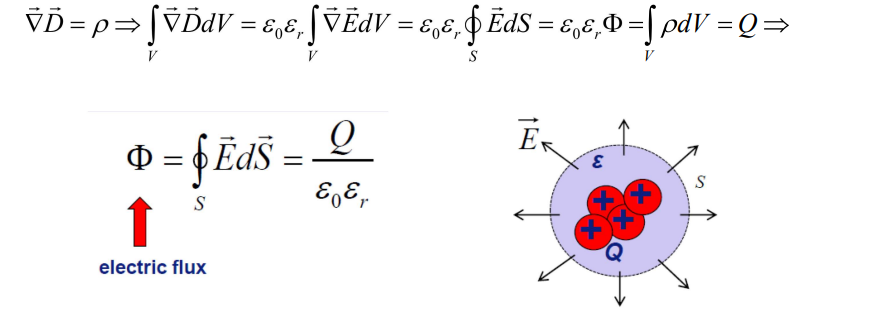
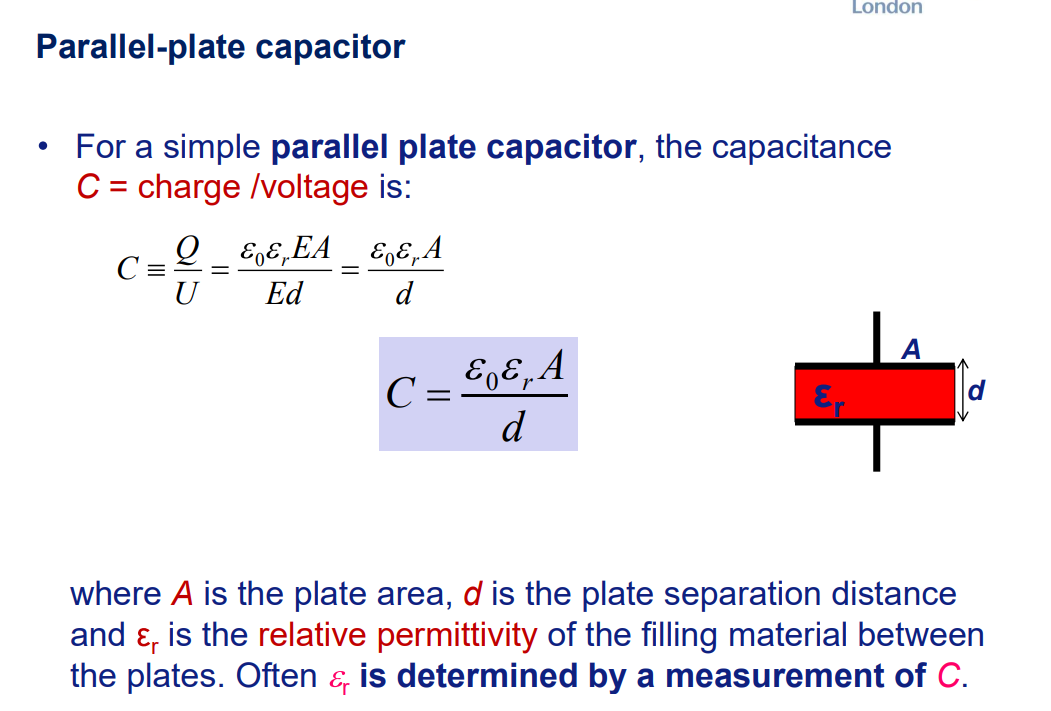
Dielectric Basics
Definition: Insulators with bound electrons (no free carriers), polarizable under electric fields.
Key Property: High resistivity (10^8–10^18 Ω·cm) vs. conductors (e.g., Cu: 10−6 Ω·cm).
Band Structure: Large bandgap (Eg≫kT) prevents electron conduction.
Polarization Mechanisms
Electronic: Electron cloud distortion (high frequencies, e.g., UV/visible light).
Ionic: Ion displacement (IR frequencies).
Orientation (Dipole): Alignment of permanent dipoles (microwave frequencies, e.g., water).
Frequency Dependence:
εr (permittivity) varies with frequency due to polarization lag (Debye relaxation).
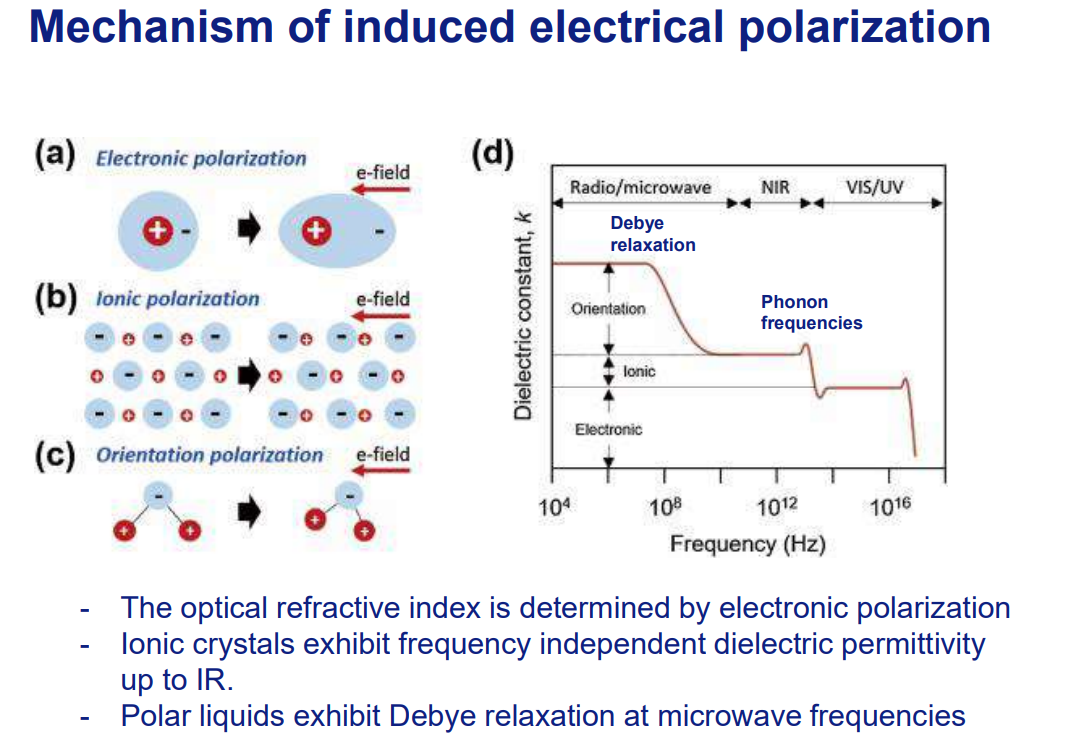
Low Frequencies (Radio/Microwave):
All polarization types (orientation, ionic, electronic) contribute.
Debye relaxation occurs in polar liquids (e.g., water) as dipoles align with the field.
Intermediate Frequencies (Infrared - IR):
Ionic polarization stops (ions can’t keep up with rapid field changes).
Only electronic polarization remains active.
High Frequencies (Optical/UV):
Only electronic polarization persists (electrons respond fastest).
The dielectric constant drops to match the refractive index squared (n²).