Start of class review
Water and Its Molecular Properties
Reluctance to Share Answers
Acknowledgment that individuals may choose to keep thoughts to themselves.
Ethanol vs. Water
Presentation of ethanol structure.
Comparison of ethanol's hydroelectric oxygen to water molecules.
Question raised: What if life were ethanol-based instead of water-based?
Properties of Water
Polarity of Water
Water is a polar molecule.
Has a density greater than many other compounds.
Functions as a heat bank, absorbing and regulating temperature.
Biochemical Context of Water Properties
Importance of polar nature:
Cohesive (water molecules stick to each other).
Adhesive (water sticks to other substances).
High surface tension resulting from cohesive properties.
High Specific Heat Capacity
Water's ability to absorb heat without significant temperature change.
High Heat of Vaporization
Energy required for water to transition from liquid to gas.
Density and Structure of Water
Water is denser than many other molecules.
Solvent Properties: Water dissolves various molecules effectively, including ions and polar molecules.
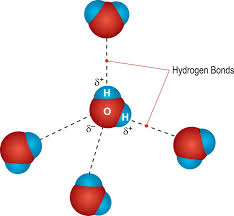
Hydrogen Bonds
Definition of Hydrogen Bond
Hydrogen bonds occur between hydrogen and oxygen atoms from different molecules.
Intermolecular Forces:
Such bonds are weak compared to covalent bonds, allowing for flexibility and interaction between different molecules.
Maximum of four hydrogen bonds can occur per water molecule.
Structure of Water
Illustration of water molecule bonds and capability to form four hydrogen bonds with neighboring water molecules.
Cohesion and Adhesion
Cohesion: Water's attraction to itself, crucial for concepts like surface tension.
Adhesion: Water's attraction to other charged surfaces, significantly aiding in processes such as water transportation in plants (meniscus formation in test tubes).
Implications for Plant Life
Capillary Action
Ability of water to move upward in plants against gravity due to cohesion and adhesion.
Water travels from roots to leaves, demonstrating the significance of these properties in biological contexts.
Thermal Properties of Water
Heat Absorption
Water can absorb immense heat (e.g., 100,000 units of energy) before temperature significantly increases.
Important for climate moderation (e.g., swimming pools remaining cooler than concrete on hot days).
Heat of Vaporization and Evaporative Cooling
Water's capacity to cool through sweat evaporation is a response to temperature changes. Evaporation requires breaking hydrogen bonds, leading to a cooling sensation on the skin.
Marine Life and Ice Density
Ice's structural properties (expanding when frozen) minimize freezing of oceans. This allows aquatic life to survive beneath the ice layer.
Summary of Water's Properties
Water is:
Polar
Cohesive and adhesive
Excellent solvent
High specific heat capacity
High heat of vaporization
Less dense in solid form than in liquid form (ice floats)
Intermolecular Forces
Covalent vs. Non-Covalent Interactions
Covalent bonds occur within a molecule; non-covalent (such as hydrogen bonds) occur between different molecules.
Water’s properties influenced by the presence of hydrogen bonding include high boiling temperatures and heat capacity.
Understanding Hydrogen Bonds in Context
Interaction of different charges (e.g., partial positive and negative from dipole moments).
Dipole Moment
Defined as a separation of charges in a molecule due to polarity (partial positive and negative regions).
Van der Waals Forces
Temporary interactions that arise from electron redistribution, contributing to molecular interactions such as base stacking in DNA.