Chemistry Unit 6
States of Matter, Kinetic Molecular Theory (KMT)
Kinetic Theory
Kinetic = motion
Kinetic Energy = energy an object has because of its motions
Kinetic Molecular Theoru = tiny particles in all state of matter are in constant motion (solids vibrate, liquids slide, gas is random). Unless Kelvin is absolute 0, it is moving
Gases
Gases are composed of particles (atoms or molecules)
a. Empty space between particles
b. No forces between particles
c. Gases fill containers regardless of volume
d. if uncontained, gases diffuse into the atmosphere within gravitational limits
Gas Particles move rapidly,
a. gases move in straight paths, only change path when collide
Collisions are elastic meaning that kinetic energy is transferred from one particle to another but total KE remains constant.
Gas Pressure
Gas pressure is the result of particles colliding with objects
A vacuum is an area with no particles
Atmospheric pressure is the pressure that air exerts on earth because gravity holds air molecules into the earths atmosphere
Pressure increases closer to earth or lower elevation
Pressure decreases as elevation increases because there is less air
Gas volume decreases with increasing pressure
increase heat increases pressure
gases are compressable
Barrometers measure air pressure
Units: 1 atm = 760 mm Hg = 101.3kPa = sea level
Permanent gases- are gasses that cannot be turne into liquid with just pressure
Kinetic Energy and Kelvin Temperature
When a substance is heated, particles absorb the energy and store it. Stored energy does not raise the temperature.
When max energy capacity of particles is stored, remaining energy increases average Kinetic Energy and particles collide and move faster and increases the temperature
Kelvin temp is related to kinetic energy
Absolute 0 is 0 Kelvin and no movement, and never has been achived
Liquids
particles are in constant motion and slide pass eachother and flow
Has some intermolecular forces that hold liquid together
Liquids are more dense gases
Liquids and Solids are condensed meaning that pressure does not reduce volume
Evaporation/ Vaporization
Converstion of a liquid to gas below boiling point
When particles at the surface of a liquid have enough energy to break IMFs they escape as a gas and it is cooling process because particles with the highest KE or most temp escape and leave behind the liquids with less KE or cooler.
Boiling Point
tempature at which the vapor pressure of the liquid equals the external pressure
tempature that substance transitions from liquid to gas
tempature of boiling liquid will never exceed boiling point because it would have been a gas
normal boiling point = boiling point of liquid at STP
Presure changes boiling point. High elevations = lower boiling points because lower pressure
Boiling with low elevations or high pressure will need more energy, or higher temp
Solids
particles vibrate about fixed points
most particles are packed in organized patterns 3-d lattice (cystaline)
Dense and condensed (cant be compressed)
Applying heat causes more vibration of particles and breaking IMFs. Melting Points
Melting Point/ Freezing Points
Melting Point = temp are which solid turns into liquid
Freezing point = temp at which liquid turns into solid
Freezing point = melting point
Ionic solids have high melting point, molecular have low
Allotropes
different forms of the same physical element
Amorphous Solids
not crystal and has no lattice (pattern)
lacks organization
an example is glass which is also referred as a super liquid becayse it cools to a rigid state without cystalzying.
a super fuid is a liquid that cools to rigid state without crystallizing
rubber is also amorphorous
Sublimation
vapor pressure of some solids is high enough that they can go from solid to gas directy.
opposite of sublimation is depostion
ex: dry ice, moth balls
What is Vapor pressure?
Plasma
4th state of matter
when gas is heated even more, the molecules will seperate into atoms and then the atoms e- will strip off and positive ions are left with free e-
the sun is plasma, and partial plamas include lightning bolts, and neon lights
This moves faster than gas
Bose Einstin Condensate
5th state of matter
super cold atoms, and particles start to act like waves instead of particles
atoms can not be distinguished
slow
A Matter of state video
Energy is needed to change states of matter. Temperature and pressure
temperature and pressure of a gas are related. Increase of tempature increases pressure because it increases the speed of particles and they colide with the walls harder. Converse is true
Liquified Natural gas is produced with high pressue and cooling it.
Uses of liquid Nitrogen: it can fast freeze things, storage of biological specimens “organs, and refrigerant.
Why does perspiration (evaporation) cool a person. Liquid water is heated to gas and leaves cool water because water absorbs the heat.
Bromine 3 states gas is orange, liquid is red, yellow solid.
Particles in bromine when cooled lose kinetic energy, slow down and condense. Attractive forces increase
Crystal shape - organized lattice (pattern), the external shape is the same as the internal molecular shape.
amophorous solid - glass, not organied, no lattice (pattern).
A Race for Absolute Zero
Tech - MRI, Refiferator, Conditions
Permanent Gases - oxygen, hydrogen, and nitrogen
they cant liquidity with just pressure
superconductivity - all resistance to he flow of electricity stops at cold temperatures
we will probably not reach 0k soon
Lewis Dot Structures
Covalent Bonds - 2 or more electrons are shared by 2 atoms to complete their octet
double bonds
triple bonds
Occurs between 2 non metals
Ionic Bonds - the metal will give up electrons to the non metal and the charges create a bond
more temp needed to break the bonds “stronger”
Steps
Draw the skeletal structure “most electronegative in the center”
Count the e-, consider the charges too
complete octect for all except hydrogen
too many electrons → use double or triple bonds
consider # of bonds
resonance structure - one multiple lewis structures can represent a molecule
Exceptions
Incomplete Octet:
Be only needs 4e-, or 2 pairs
B only needs 3 pairs because has 3 valence 3e-
odd Electron Molecules
Expanded Octet - can occur with central atoms with principal quantum number more than 2. third-row (or higher)
place extra pairs around the atom
use empty valence d orbitals
CNOF always follow, second row never exceed
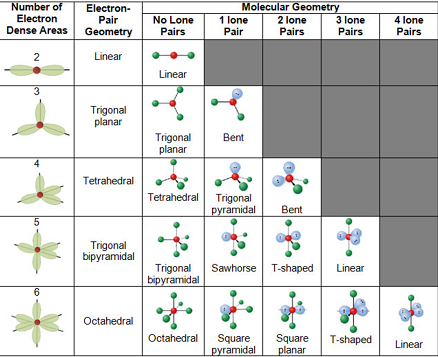
Shapes or Electronic Geometry VSEPR
VSEPR - theory that predicts the shape based on electron pairs repelling
Valence
Shell
Electron
Pair
Repulsion
Polarity
Electronegativity (EN) is an atom's tendency to attract electrons in chemical bonds.
EN increases to the right and up on the periodic table, excluding the noble gases.
side to side matters more than vertical
Bond Polarity
When two nonmetal atoms bond, they share electrons. They may or may not share electrons evenly.
If Same atom, then it is non polar because the non has more favor for electrons
highest electron density between atoms
if different atoms, one will be more electronegative and will pull electrons more thus its called a polar bond
creates partial charges
e- density is drawn towards the side of the more electronegative atom
Dipole Moments - is a measure of the polarity of bonds and is represented by an arrow which points to the more EN atom
Polar does not mean charges
Polarity in molecules of 3 or more Atoms
Polar if the central atom has lone pairs or the outer atoms are not all the same
nonpolar if the central atoms have no lone pairs and the outer atoms are identical
Properties
like disolve like
Polar molecules have a higher melting and boiling point than non polar because polar can have IMF - still far below than ionic
Intermolecular Forces - force between 2 molecules but not bonding
dipole dipole interaction - is when the partial charges attract → higher melting and boiling point
hydrogen bonding - is a stronger dipole interaction
occurs in molecules that have H bonded to ONF
hydrogen is small so it allows for the molecules to be close → stronger
london dispersion forces - the temporary attraction between dipoles
can happen for anything
more e- → stronger force
Radioactivity
General
Unstable isotopes try to gain stability by making changes
these changes release lots of energy
reactions are not affected by temperature, pressure, catalysis, or compound
cannot be slowed of speeded up
Becqueral, Marie and Pierre Curie
Terms
Radioisotope: radioactive isotope has too many neutrons
Radioactive decay: the process by which an unstable nucleus loses energy by emitting radiation “spontanous” → to a isotope of a different element
Types of Radiation
Alpha (α) 24H – a positively charged helium isotope
weakest
The mass number goes down by 4, the atomic number goes down by 2
Beta (β) – an electron – formed from decomposition of a neutron
medium strength
the mass number is unchanged and the mass number goes up by one
(one less neutron, one more proton)
Gamma (γ) – pure energy; called a ray rather than a particle
strongest, most dangerous and can affect DNA
Half Life - the time it takes for ½ of a sample to decompose
the rate of nuclear transformation depends on the reactant concentration
Mass and T table
start with the most mass and 0 time
divide the mass by 2 til the final value is reached
divide the time divided by the time
Transmutation: conversion of atoms of one element into another element
may be natural or manmade
all isotopes above #82 is radioactive
Transuranium element: man made and over #92
Nuclear Fission -
“spliting of atoms”
usually in large atoms that are not as stable
releases lots of energy
Ionizing Radiation - radiation strong enough to knock off electrons of the atoms that it strikes
Nuclear reactors use controlled fission to produce energy as heat, and this makes steam that turns turbines and generates electricity
Nuclear reactions are powered by bombarding neutrons and a chain reaction - the atoms will separate and produce neutrons which will bombard other atoms
Controlling Fission: to prevent too much heat or explosions → meltdowns
Neutron Moderation: slows neutrons with water and carbon
Neutron Absorbtion: traps neutrons in control rods
Cherynoble - lack of control
makes lots of energy
Nuclear Fusion
its the opposite of fission
Fusion - small nuclei combine “hydrogen combine" → helium”
This occurs in the sun and stars which created lots of energy
Exessive heat and hard to contain
Detecting Radiation
Can detect results of Radiation
Geiger Counter: tube with neutral gas
best for beta “e-”
it interacts with the gas inside, causing ionization.
Scintillation Counter: flasg of light occur when ions strike the phosphor
works for all radiation
best for alpha
Film Badge: is a photographic film encased in holder - used by workers
Research and Medicine
Neutron Activation: determine the age of a sample
Radioisotope Tracers: used to follow chemicals throughout a bioloifcal process
Cancer Treatment: since cancer cells divide more rapidly, they are susceptible to radiation
telethearapy
implantation
pharmacueticals