energy changes
Energy Transfer during reactions
when chemical reactions occur, energy is conserved
- if a reaction transfers energy to the surroundings, the product molecules must have less energy than the reactants, by the amount transferred
Exothermic Reactions
transfers energy to surroundings so temperature of surroundings increases; product molecules have less energy than the reactants
eg. combustion, neutralisation, self-heating cans, hand warmers
Endothermic Reactions
takes in energy from surroundings so temperature of surroundings decreases; product molecules have more energy than the reactants
eg. thermal decomposition, citric acid and sodium hydrogencarbonate. sports injury packs
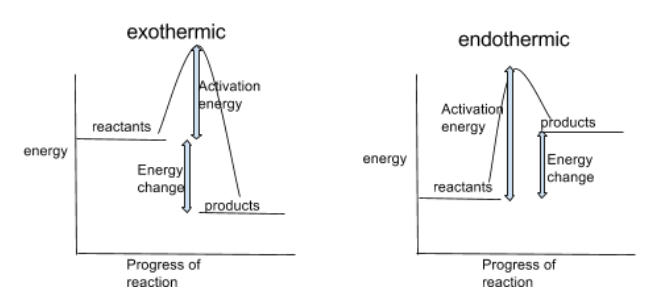
Reaction Profiles
can be used to show the relative energies of reactants and products, the activation energy and overall energy change of a reaction
activation energy = the minimum amount of energy colliding particles need to cause a reaction
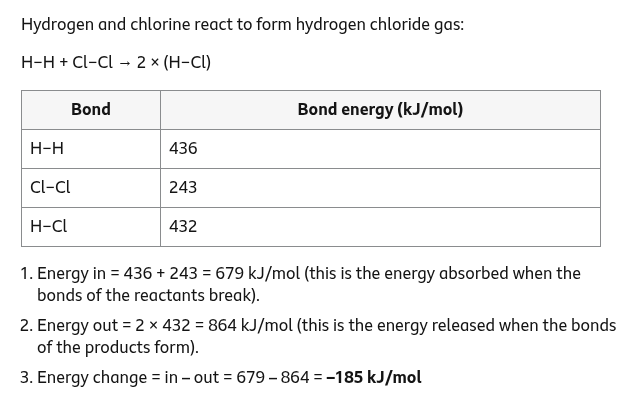
Energy Change of Reactions
breaking bonds in reactants requires energy
forming bonds in products releases energy
→ can be calculated from bond energies:
energy taken in to break < energy released when formed = exothermic
energy taken in to break > energy released when formed = endothermic
energy change = sum of bonds broken - sum of bonds made