Patient Safety
1. Ways Medication Errors Can Occur
Medication errors can happen at any stage of the medication-use process:
Prescribing: Wrong drug, dose, or patient.
Order Communication: Misinterpretation of orders (e.g., poor handwriting, ambiguous abbreviations).
Product Labeling/Packaging: Look-alike/sound-alike drugs, confusing packaging.
Compounding/Dispensing: Incorrect preparation or labeling.
Distribution/Administration: Wrong route, dose, or patient.
Monitoring/Education: Inadequate patient counseling or failure to monitor for side effects.
System-based causes are the most common, not individual mistakes. Poor system design allows errors to reach patients.
Examples:
Drug binds to the IV bag (incompatibility).
Amoxicillin powder dispensed without reconstitution.
Anticoagulant IV not started or clamped.
2. Medication Error vs. Adverse Drug Reaction (ADR)
Medication Error: Preventable event leading to inappropriate medication use or patient harm while the medication is in the control of the healthcare professional,patient or consumer.
Example: Wrong drug dispensed, but caught before patient takes it (close call).Adverse Drug Reaction (ADR): Harm caused by a drug at normal doses, during normal use; not preventable.
Example: Vancomycin-induced acute kidney injury, despite correct dosing.
3. Key Definitions
Medication Error: Any preventable event that may cause or lead to inappropriate medication use or patient harm.
Adverse Drug Reaction (ADR): Harm caused by a drug at normal doses, not due to error.
Close Call/Near Miss: Error corrected before reaching the patient (e.g., patient notices wrong tablet).
Error of Omission: Failing to do something necessary for safety (e.g., not starting an infusion).
Error of Commission: Doing something incorrectly (e.g., dispensing wrong drug).
Sentinel Event: Unexpected event causing death or severe harm (e.g., wrong-site surgery, undiluted KCl injection).
4. Patient Safety Organizations and Efforts
Institute for Safe Medication Practices (ISMP): Non-profit, shares safety alerts, runs MERP (Medication Errors Reporting Program).
The Joint Commission (TJC): Accredits healthcare organizations and programs, sets National Patient Safety Goals (NPSG) for medication safety.
Centers for Medicare & Medicaid Services (CMS): Sets safety standards for reimbursement on behalf of US HHS
National Association of Boards of Pharmacy (NABP): Regulates pharmacy practice.
Utilization Review Accreditation Commission (URAC): Accredits healthcare organizations.
Efforts include: Reporting programs, safety alerts, setting standards, and regular accreditation surveys.
5. Implementing Error Reduction in Healthcare Systems
Tall Man Lettering: Highlights differences in look-alike drug names by using uppercase letters (e.g., hydrOXYzine vs. hydrOMORPHone).
Standardized Protocols: For high-alert medications (e.g., heparin dosing order sets).
Limiting Concentrations: Only certain strengths available to reduce dosing errors.
Barcoding: Ensures right drug, dose, route, and patient.
E-prescribing: Reduces prescription errors and increases efficiency.
Education: Staff and patient training, in-services.
Patient Profiles: Check allergies, interactions, polypharmacy.
"Five Rights": Correct patient, drug, dose, route, time.
6. High-Alert Medications (ISMP List)
Anticoagulants (heparin, warfarin)
Insulin
Oral hypoglycemics
Opioids
Antiarrhythmics
Anesthetics
Chemotherapeutics
Injectable Potassium Chloride
Hypertonic Saline
Strategies: Avoid storing in automated cabinets, use premixed products, monitor labs closely.
7. “Do Not Abbreviate” List (ISMP & TJC)
Common unsafe abbreviations:
U (unit) – write “unit”
IU (international unit) – write “international unit”
QD/QOD (daily/every other day) – write “daily” or “every other day”
Trailing zero (1.0 mg) – write “1 mg”
Lack of leading zero (.5 mg) – write “0.5 mg”
MS/MSO4/MgSO4 – write “morphine sulfate” or “magnesium sulfate”
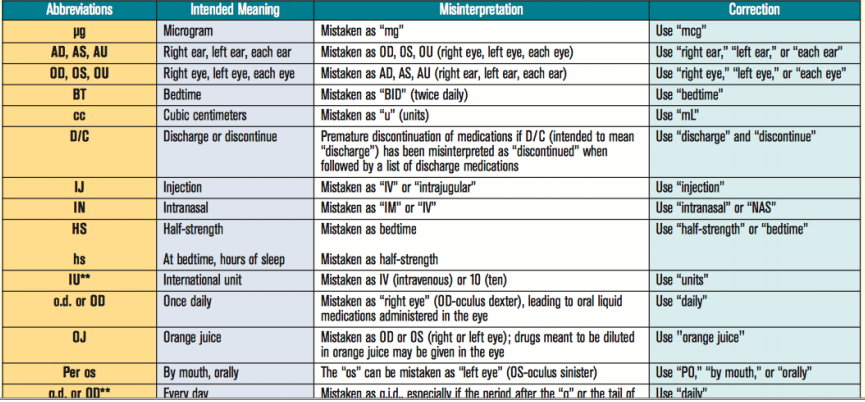
Example corrections:
“Nitroglycerin .4 mg PO Q5 min PRN” → “Nitroglycerin 0.4 mg PO every 5 minutes as needed”
8. REMS: Risk Evaluation and Mitigation Strategies
Definition: FDA-required programs for certain drugs to manage serious risks so that benefit outweighs risks.
Components: Prescriber/pharmacy certification, patient education, restricted distribution.
Examples:
iPLEDGE (isotretinoin)
ESA APPRISE (erythropoiesis-stimulating agents)
9. Evaluation and Quality Improvement Methods
Failure Mode and Effects Analysis (FMEA): Proactive, prospective assessment to identify and fix potential failures before they occur in design and medication delivery systems.
Root Cause Analysis (RCA): Retrospective, investigates errors after they happen to find and fix root causes that lead to error
Continuous Quality Improvement (CQI): Ongoing process to improve efficiency, quality, satisfaction, and reduce costs.
Lean: Minimize waste.
Six Sigma: Reduce defects using DMAIC (Define, Measure, Analyze, Improve, Control).
Summary Table: Learning Objectives & PDF Content
Learning Objective | Key Content/Examples from PDF |
|---|---|
Ways errors occur | System failures, look-alike drugs, poor communication |
Error vs. ADR | Preventable vs. not preventable, examples given |
Definitions | Medication error, ADR, close call, omission/commission, sentinel event |
Safety organizations | ISMP, TJC, CMS, NABP, URAC |
Reducing errors | Tall Man Lettering, protocols, barcoding, education, Five Rights |
High-alert meds | Anticoagulants, insulin, opioids, etc. |
Do not abbreviate | ISMP/TJC lists, example corrections |
REMS | iPLEDGE, ESA APPRISE, restricted distribution |
Quality improvement | FMEA, RCA, CQI, Lean, Six Sigma |