Basic Cosmetic Chemistry
Introduction: Relevance of Chemistry to Dermal Therapies and Cosmetology
Chemistry provides a foundational knowledge for understanding cosmeceutical and pharmaceutical formulae and their interaction with the skin.
Helps in understanding cosmeceutical/pharmaceutical manufacturing.
Aids in interpreting literature and studies, especially in cosmetic science.
Topics covered include atoms and elements, atomic structure, electron configuration, chemical formulae, bonds, water in chemistry, acids and bases, and relevance to dermal therapies and cosmetic nursing.
Atoms and Elements
Atoms are the smallest units of matter; all matter is comprised of elements.
A chemical element is an atom or a pure chemical substance composed of atoms with the same number of protons (e.g., Hydrogen, Nitrogen, Carbon).
There are 118 known elements (94 occur naturally on earth)
Each element has a unique name and symbol.
Names historically originate from Greek or Latin; symbols are in Latin/short form.
All chemical matter consists of elements

Periodic Table and Atomic Structure
Contains all known elements, organised by chemical properties (families) and increasing atomic number/size.
Atomic number = number of protons in an atom’s nucleus.
Hydrogen: lightest element, atomic weight 1.008, atomic number 1.
Families/Groups: vertical columns where elements share similar properties.
Examples:
Alkali metals = 1st column
Alkaline earth metals = 2nd column
Halogens = 2nd last column
Noble gases = last column
Elements in the same family behave similarly → e.g., knowing Sodium helps predict properties of Lithium & Potassium.
Elements also classified as:
Metals → shiny, good conductors of heat & electricity
Non-metals → poor conductors
Semi-metals (metalloids) → semi-conductors
Simple Model of the Atom
Atom structure: made of a central nucleus with surrounding electrons.
Nucleus:
Small, dense, positively charged
Contains:
Protons → positive charge
Neutrons → neutral charge
Protons:
Positively charged particles
Found in the nucleus
Contribute to the overall positive charge of the atom
Neutrons:
Neutral (no charge)
Found in the nucleus
Add mass and stability to the atom
Electrons:
Negatively charged particles
Orbit the nucleus at fixed distances
Occupy energy levels called orbits/shells
Held in position by attraction to the positively charged nucleus, these electrons play a crucial role in forming chemical bonds and determining the reactivity of atoms in cosmetic formulations.
Atomic Structure: About Electrons
Electron levels (shells/orbits)
Electrons circulate the nucleus at different levels, also referred to as orbits/shells.
Electrons can spin in different directions, as long as they remain in their designated shell.
Electrons are kept in position by electric charges (electrostatic attraction to the positively charged nucleus).
Valence level & reactions
The outermost level is the valence level, where valence electrons reside.
Most chemical reactions occur via valence electrons.
Atoms are most stable when the valence shell is full or empty.
Group dependence of valence electrons
Valence electrons are group-dependent:
Group 1 elements (starting with Hydrogen) have 1 valence electron.
Noble gases have 8 valence electrons and are chemically stable.
Exception: Helium has 2 valence electrons due to its small size.
Representation
Valence electrons are typically shown in a group/outcome table for metals vs non-metals (as illustrated in your materials).
Valence Electrons
Elements in Group 1 (1st column) have 1 valence electron in their outer shell.
Elements in Group 2 (2nd column) have 2 valence electrons.
Elements in Group 3 and beyond continue after the transition metals, with valence electrons increasing in their outer shell as you move across the periods.
Elements in Group 8 (8th column) have 8 valence electrons, which is the maximum number for an outer shell. This makes them stable and less reactive, known as noble gases.
Exceptions:
Helium: Can only hold 2 electrons due to its small size.
Hydrogen: Has 1 electron in its outer shell and can hold a maximum of 2 electrons.
The Octet Rule and Ions
The octet rule: atoms tend to gain, lose, or share electrons to achieve a full valence shell of 8 electrons.
Exceptions: Hydrogen and Helium hold up to 2 valence electrons.
Ions form when atoms gain or lose electrons to satisfy the octet rule:
Ions that have lost electrons are CATIONS (+).
Ions that have gained electrons are ANIONS (-).
Key terms:
Atomic number: number of protons in the nucleus.
Atomic mass (daltons or amu): total number of protons and neutrons.
Molecule: two or more atoms bonded together.
Molecular weight: total mass of all atoms in a molecule.
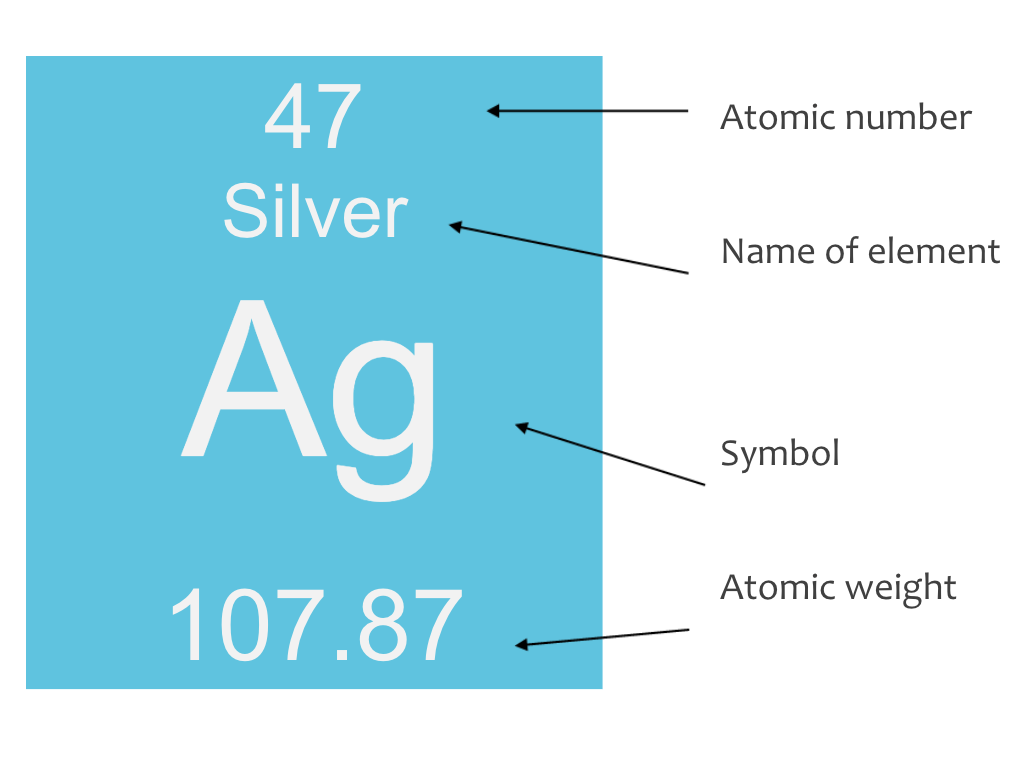
Reading Elements
Each element on the periodic table generally contains the same information in a standard format, including:
Name and Symbol: The element’s full name and its one- or two-letter symbol.
Atomic Number: Represents the number of protons in the nucleus of the element.
Atomic Weight: The average mass of the element’s atoms based on all naturally occurring isotopes.
Note: Atomic mass and atomic weight are not the same:
Atomic Mass: The sum of protons and neutrons in a single atom.
Example: Carbon-12 has an atomic mass of 12.
Atomic Weight: The weighted average of the atomic masses of all natural isotopes of an element.
Example: Carbon has an atomic weight of 12.017.
Cosmeceutical and Elements
Main metals used in cosmeceuticals as active ingredients include:
Zinc (Zn, Atomic Number: 30, Atomic Weight: 65.38, Group 12, Period 4, d-block)
Copper (Cu, Atomic Number: 29, Atomic Weight: 63.546, Group 11, Period 4, d-block)
Aluminium (Al, Atomic Number: 13, Atomic Weight: 26.982, Group 13, Period 3, p-block)
Selenium (Se, Atomic Number: 34, Atomic Weight: 78.971, Group 16, Period 4, p-block)
Strontium (Sr, Atomic Number: 38, Atomic Weight: 87.62, Group 2, Period 5, s-block)
Gold (Au, Atomic Number: 79, Atomic Weight: 196.967, Group 11, Period 6, d-block)
Metals have been used in skin care since ancient times (e.g., calamine lotion containing zinc; use of copper-based minerals).
Zinc
(Atomic Number: 30, Atomic Weight: 65.38, Group 12, Period 4, d-block)
Properties in Cosmeceuticals
Used in multiple topical forms across skincare and medications.
Functions depend on the specific form of zinc used.
Functions & Benefits
Antibiotic activity.
Antifungal activity.
Astringent effects.
Anti-inflammatory effects.
Skin-protective ointments.
Sunscreen: Zinc oxide acts as a physical sunblock by scattering and reflecting UVA/UVB radiation.
Supports skin repair and healing.
Mechanisms of Action
Accelerates wound healing by increasing re-epithelialisation.
Anti-inflammatory by inhibiting inflammatory mediators.
Antioxidant properties provide protective effects.
Enhances fibroblast resistance to UV-induced oxidative stress.

Copper
(Atomic Number: 29, Atomic Weight: 63.546, Group 11, Period 4, d-block)
Contains 29 protons in its nucleus.
Properties in Cosmeceuticals
Difficult to permeate through the skin when applied topically.
To improve penetration, copper is bound to a short peptide chain (a protein peptide of 3 amino acids).
These formulations are known as copper-peptide cosmeceuticals (CS).
Functions & Benefits
Anti-ageing: Enhances collagen synthesis and supports wound healing (particularly post-cosmetic surgery).
Antioxidant properties: Helps reduce oxidative stress.
Melanin production: Dietary copper is essential as it oxidises tyrosine, leading to melanin synthesis.
Common Uses in Skincare
Anti-ageing products.
Wound healing and repair support.
Antioxidant protection.

Aluminium in Cosmeceuticals
Aluminium salts are commonly used in deodorants for astringent and antiperspirant properties.
Mechanism: aluminium salt interacts with sweat components to form insoluble polymer gel plugs within the sweat duct, blocking sweat from reaching the surface.
Additional cosmetic uses: colorants in lipstick; mild abrasive in toothpaste.
Strontium
(Atomic Number: 38, Atomic Weight: 87.62, Group 2, Period 5, s-block)
Contains 38 protons in its nucleus.
Properties in Cosmeceuticals
Limited knowledge on skin-specific effects (relatively new to cosmeceutical industry).
Functions as an anti-irritant and anti-inflammatory when used in formulations with high concentrations of AHAs.
Example: Refinity Peel with Cosmederm-7 (70% glycolic acid, pH 0.6) → helps minimise redness, stinging, and irritation.
Mechanisms of Action (Unclear)
May act through topical effects on the skin barrier, or
Through alterations in the inflammatory cascade (Baumann, 2005).
More scientific evidence is needed to fully understand effects.
Gold
(Atomic Number: 79, Atomic Weight: 196.967, Group 11, Period 6, d-block)
Contains 79 protons in its nucleus (large atom).
Properties in Cosmeceuticals
Not commonly used in cosmeceuticals.
Reported to have anti-inflammatory and anti-bacterial properties.
Recent studies suggest gold nanoparticles may have anti-glycation properties.
Limitations & Cautions
No published evidence currently supports gold as an effective anti-ageing ingredient.
Marketing claims (e.g., “cures” or increased libido) linked to oral colloidal gold are unrealistic and may have discredited its potential topical benefits.
Much more research and evidence is required before its benefits can be confirmed.
Chemical Formulae and Molecular Representation
When two or more different atoms join, they form a compound.
Formulae are used to:
Represent the elements in a compound.
List the elements present.
Show how many of each element are present.
Indicate how elements are bonded.
Example: Ethyl alcohol (ethanol).
Ethanol representation:
Molecular formula: or
Composition: 2 carbons, 6 hydrogens, 1 oxygen.
Molecular representation: a 2D schematic is often used for simplicity; one molecule of ethanol contains the atoms shown above.
Chemical Bonds
Bonding is the force that holds atoms together; bonding occurs to satisfy the octet rule via valence electrons.
Major types of chemical bonds:
1) Ionic bonds
2) Covalent bonds
3) Hydrogen bondsHydrogen bonding can be viewed as hydrogen atom of one molecule attracted to an electronegative atom of another molecule, especially N, O, or F in second-row elements.
Ionic Bonds
Ionic bonds form between oppositely charged ions.
Electrons are transfered permanently from one atom to another (electrons are not shared).
Formation involves metals and non-metals exchanging electrons.
Examples of ionic compounds:
Sodium chloride:
Potassium iodide:
Magnesium oxide:
Conceptual summary: ions form cations and anions, which attract each other to form ionic lattices.
Visual cue: tendency to crystallize as crystals (e.g., aluminum oxide crystals in some contexts).
Common Ions in Cosmetic Chemistry
Initially listed ions and functional groups include:
Polyphosphates
Alpha Hydroxy Acids (AHA)
Beta Hydroxy Acids (BHA)
Thioglycolates
Ionic surfactants
Salts
Note: these ions and species can influence formulation properties, stability, and skin compatibility.
Covalent Bonds
Covalent bonds involve sharing a pair of electrons between two non-metal atoms.
Neither atom fully donates or accepts electrons; both share to achieve octet.
Bond types by number of electron pairs:
Single bond: one shared pair
Double bond: two shared pairs
Triple bond: three shared pairs
Examples of covalent compounds:
Methane: (one carbon, four hydrogens) with four single bonds
Carbon monoxide: (one C–O with a triple bond)
Covalent network concepts:
Large networks with many covalent bonds across atoms (e.g., Graphite, Diamond, Silica)
Covalent Bonding in Cosmetic Chemistry
Carbon chemistry is central: polymers of ethylene form emulsifying waxes and emollients.
Aromatic rings appear in essential oils.
Chains in carrier oils; silicon oils as emollients; silicon waxes as abrasives.
Hydrogen Bonding
Hydrogen bonding occurs when a hydrogen atom bound to an electronegative atom (like O or N) interacts with another electronegative atom in a different molecule.
Common in covalently bonded molecules that contain hydrogen, notably water (H2O).
Examples of substances with hydrogen bonding potential in formulations: Ethanol (CH3CH2OH), Ammonia (NH3), Water (H2O), Hydrogen peroxide (H2O2), Acetic acid (CH3COOH), organic amines (e.g., Methanamine CH3NH2), and DNA.
Hydrogen Bonding in Water and the Importance of Water in Cosmetic Chemistry
Water exhibits hydrogen bonding: the hydrogen of one water molecule is attracted to the oxygen of another, creating a network and surface tension.
Hydrogen bonds are pivotal for many properties of water and its solvent capabilities.
Water plays two basic roles in cosmetic formulations:
Vehicle agents: essential ingredients in formulation and application (examples: water, oils, silicone) and often act as solvents.
Active agents: ingredients that actively affect the skin’s appearance (varied examples).
Some ingredients may play dual roles, such as water, which can act as both vehicle and active in certain contexts.
Properties and Characteristics of Water
Density: roughly 1000 kg/m³ for liquid water at 4°C; ice has a density around 917 kg/m³.
Phase at standard conditions: liquid around room temperature.
Melting point: 0°C; Boiling point: 100°C.
Triple point: 273.16 K, 611.73 Pa.
Critical point: 647 K, 22.1 MPa.
Specific heat capacity (liquid): ≈ 4186 J·kg⁻¹·K⁻¹.
Specific heat capacity (gas): ≈ 1970 J·kg⁻¹·K⁻¹; cv (gas) ≈ 1510 J·kg⁻¹·K⁻¹ at 300 K.
Viscosity (liquid water at 20°C): ≈ 0.001 Pa·s.
Surface tension (water at 20°C): ≈ 7.28 N·m⁻¹.
In cosmetic chemistry, water serves as:
Vehicle component and solvent.
Participant in reactions such as neutralisation with acids/bases to form salts.
Acids and bases require pH considerations in formulation to maintain skin compatibility.
Acids, Bases, and the pH Scale
Definitions:
Acid: anything that produces hydrogen ions (H⁺) in water.
Base: anything that produces hydroxide ions (OH⁻) in water.
pH scale describes hydrogen ion concentration; lower pH indicates acidity; higher pH indicates basicity/alkalinity.
Molecular formula cues:
Acids often have hydrogen written first (e.g., Carbonic acid: ).
Neutralisation and salts:
Acid + Base → Salt + Water.
A salt is an ionic compound; all ions are electrolytes.
In cosmeceuticals, acids are often neutralised with bases to form salts.
Some salts can cause irritation on the skin (as reported by Van Scott et al., 1996).
pH explainer concepts: the scale indicates acidity/alkalinity and informs formulation stability and skin compatibility.
References / Further Reading
Baumann, M.D. (2002). Cosmetic Dermatology. McGraw Hill.
Beauty Training Package Support Materials (2005).
Holford, P. (1997). The Optimum Nutrition Bible. Piatkus.
Marieb, E. (2004). Human Anatomy & Physiology (6th Ed).
Schwartz, J., Marsh, R., and Draelos, Z. (2005). Zinc and Skin Health: Overview and physiology and pharmacology. Journal of Dermatologic Surgery, 31: 837-847.
Scientific Committee on Consumer Safety (SCCS) (2020). Opinion on the safety of aluminium in cosmetic products (SCCS/1613/19).
Van Scott, E. & Yu, R. (1996). Bioavailability of Alpha-hydroxy acids in topical formulations. Journal of Cosmetic Dermatology, 9:54-62.
Windell, J. (2014). Cosmederm Redefines Peel Science Standard with Launch of Refinity® Skin Health System.
Key Formulas and Notation (Summary)
Ethanol (Ethyl alcohol):
Water:
Sodium chloride (NaCl):
Potassium iodide (KI):
Magnesium oxide (MgO):
Methane:
Carbon monoxide:
Covalent bonds: represented by shared electron pairs; e.g., methane and ethane{
Ethane: with a single bond framework (C–C and C–H bonds)
}
Ionic bond example: (electrons transferred to form cations and anions)
Acid + Base → Salt + Water:
Oxidation and Reduction concepts are implied in the context of electron transfer and free radicals; oxidation involves loss of electrons; antioxidants donate electrons to neutralise free radicals.
Free radicals examples cited: HO•, :CH_2, •O⁻₂
Polar vs Non-polar (dipole concept) is tied to electronegativity and dipole formation in covalent bonds.
Note on Terminology and Scope
This set of notes consolidates the material presented across the lecture slides, including definitions, mechanisms, and examples related to basic cosmetic chemistry as applied to dermal therapies and cosmeceuticals.
It integrates foundational chemistry concepts with practical cosmetic science applications, including metals used in cosmeceuticals, water properties, and pH considerations for formulation safety and efficacy.